Abstract
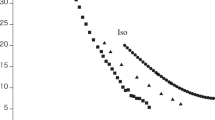
SINCE the crystal structure of hexamethyl benzene at room temperatures has been accurately determined by Robertson and Brockway1, it seemed of interest to investigate the intra- and inter-molecular interactions of the methyl groups. Pauling2 has suggested that a lower transition point at — 165° C. marks the beginning of rotation of methyl groups. It is then desirable to elucidate the mechanism of an upper transition found at 110° C. The thermal data of these transitions as well as those of the melting point have been reported by Parks and his co-workers3. We have made a preliminary dilatometric investigation of this substance from room temperature to the liquid temperature. The results are shown in the accompanying graph.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Brockway and Robertson, J. Chem. Soc., 1324 (1939).
Pauling, Phys. Rev., 36, 430 (1930).
Spaght, Thomas and Parks, J. Amer. Chem. Soc., 36, 882 (1932).
Smits and Muller, Z. phys. Chem., B, 36, 140 (1938).
Smits, Pollender and Kröger, Z. phys. Chem., B, 41, 215 (1938).
Bridgman, "Physics of High Pressure".
White and Bishop, J. Amer. Chem. Soc., 62, 16 (1940).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
CHIHARA, H., SEKI, S. Volume Change of Hexamethyl Benzene. Nature 162, 773–774 (1948). https://doi.org/10.1038/162773a0
Issue Date:
DOI: https://doi.org/10.1038/162773a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.