Abstract
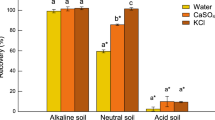
ON somewhat indirect evidence, the theory has been put forward1,2 that hydroxylamine is an intermediate in the first stage of soil nitrification, that is, the stage in which ammonium ions are oxidized to nitrite ions. I have now tested this theory directly by making use of the observation3 that fresh soil percolated with an ammonium salt and potassium chlorate produces no nitrate but only nitrite, because the chlorate ion selectively inhibits the proliferation of the nitrite-oxidizing micro-organisms. It therefore follows that if hydroxylamine is an intermediate in nitrite formation, a soil percolated with hydroxylamine and chlorate should produce nitrite in the percolate. Because the colorimetric estimation of very low concentrations of nitrite is simple and certain, the hydroxylamine may be tested at very low concentrations and its possible toxic effect4 minimized.
Similar content being viewed by others
Article PDF
References
Martin, W. P., Buehrer, T. F., and Caster, A. B., Proc. Soil Sci. Soc. Amer., 7, 223 (1942).
Kluyver, A. J., and Donker, H. J. L., Chem. d. Zelle u. Gewebe., 13, 134 (1926).
Lees, H., and Quastel, J. H., Nature, 155, 276 (1945).
Lees, H., and Quastel, J. H., Biochem. J., 40, 824 (1946).
Lees, H., J. Agric. Sci., 37, 27 (1947).
Corbet, A. S., Biochem. J., 29, 1086 (1935).
Bonazzi, A., J. Bact., 6, 479 (1921).
Godlewski, E., Cent. Bact., II, 2, 458 (1896).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LEES, H. Intermediates in Soil Nitrification. Nature 162, 702 (1948). https://doi.org/10.1038/162702a0
Issue Date:
DOI: https://doi.org/10.1038/162702a0
This article is cited by
-
Hydroxylamine as an Intermediate in Nitrification
Nature (1952)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.