Abstract
Objective:
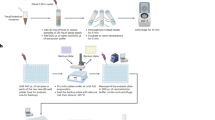
In the present study, Lactobacillus johnsonii (La1) doubly labelled with 15N and 13C (dlLa1) was used to follow the metabolic fate of orally administered dlLa1 in humans.
Design:
Experimental study.
Setting:
Research Laboratory, Children's Hospital, University of Rostock.
Subjects:
A total of 10 healthy adults aged 23–36 y.
Intervention:
The subjects received 87 mg/kg body weight viable dlLa1 and 10 g raffinose together with breakfast. Expired air samples were taken over 14 h, whereas urine and faeces were collected over 2 days. A blood sample was taken after 2 h. 13C- and 15N-enrichments were measured by isotope ratio mass spectrometry (SerCon, UK) and H2-concentrations were measured by electrochemical detection (Stimotron, Germany).
Results:
The orocaecal transit time (OCTT) was reached after 3.7 h. The 13CO2-exhalation amounted to 8.6% of ingested dose. The urinary excretion of 13C and 15N was 1.3 and 12.4% of ingested dose, respectively, whereas the faecal excretion was 39.9 and 37.6% of ingested dose, respectively. After 2 h, 13C- and 15N-enrichment of fibrinogen amounted to 70 and 90 ppm excess, respectively.
Conclusions:
In comparison to OCTT of 3.7 h, both stable isotopes appear after 30 min in breath and urine, indicating that dlLa1 is rapidly digested in the small bowel before reaching the caecum. This is confirmed by 13C-and 15N-enrichments of blood plasma fractions. The ingestion of dlLa1 led to an excretion of 50% of ingested dose of both stable isotopes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Alm L (1989): The therapeutic effects of various cultures – an overview. In Therapeutic Properties of Fermented Milks ed RK Robinson New York: Elsevier Applied Science.
Barrie A, Davies JE, Park AJ & Workmann CT (1989): Continuous-flow stable isotope analysis for biologists. Spectroscopy 4, 42–52.
Bernet MF, Brassat D, Neeser JR & Serving AL (1994): Lactobacillus acidophilus La1 binds to cultured human intestinal cell lines and inhibits cell attachment and cell invasion by enterovirulent bacteria. Gut 35, 483–489.
Brösicke H (1987): Bestimmung der Fettsäureoxidation Frühgeborener mit dem 13CO2 Atemtest während kontinuierlicher 13C-Triolein-Infusion. In Klinische Ernährung eds FW Ahnefeld, W Hartig, E Holm & G Kleinberger, pp 4–17. München, Bern, Wien, San Francisco: Zuckschwerdt.
Coplen TB (1996): New guidelines for reporting stable hydrogen, carbon, and oxygen isotope-ratio data. Geochim. Cosmochim. Acta 60, 3359–3360.
De Man JD, Rogosa M & Sharpe ME (1960): A Medium for the cultivation of Lactobacilli. J. Appl. Bacteriol. 23, 130–135.
Faust H, Bornhak H, Hirschberg K, Jung K, Junghans P, Krumbiegel P & Reinhardt R (1981): Klinisch-chemische und isotopenanalytische Methoden zur Untersuchung des Stickstoff-Stoffwechsels mit N-15 beim Menschen. Methodenkatalog pp 1–210. Leipzig: Akademie der Wissenschaften.
Finegold SM, Sutter VL & Mathisen GE (1983): Normal indigenous intestinal flora. In Human Intestinal Flora in Health and Disease ed DJ Hentges, pp 3–31. New York: Academic Press.
Fioramonti J, Theodorou V & Bueno L (2003): Probiotics: what are they? What are their effects on gut physiology? Best Pract. Res. Clin. Gastroenterol. 17, 711–724.
Fuller R (1989): Probiotics in man and animals. J. Appl. Bacteriol. 66, 365–378.
Haycock GB, Schwartz GJ & Wisotzki DH (1978): Geometric method for measuring body surface area: a hight-weight formula validated in infants, children and adults. J. Pediatr. 93, 62–66.
Heine WE, Mohr C & Wutzke KD (1992): Host-microflora correlations in infant nutrition. Prog. Food Nutr. Sci. 16, 181–197.
Heine WE, Berthold HK & Klein PD (1995): A novel stable isotope breath test: 13C-labelled glycosyl ureides used as noninvasive markers of intestinal transit time. Am. J. Gastroenterol. 90, 93–98.
Heine W, Mohr C & Münch C (1996): Zuckerverbindungen als Substrate für Wasserstoff-Atemgasteste: Korreleation zur Dosis, orozökalen Transitzeit, Mikroflora und zu Nebenwirkungen. Pädiatr. Grenzgeb. 34, 481–490.
Kandler O & Weiss N (1986): Genus Lactobacillus. In Bergey's Manual of Systematic Bacteriology, Vol. II eds PHA Sneath, NS Mair, ME Sharpe & JG Holt Baltimore: Williams & Wilkins.
Kimoto H, Nomura M, Kobayashi M, Mizumachi K & Okamoto T (2003): Survival of lactococci during passage through mouse digestive tract. Can. J. Microbiol. 49, 707–711.
King CE & Toskes PP (1986): Comparison of the 1-gram [14C]xylose, 10-gram lactulose-H2, and 80-gram glucose-H2 breath test in patients with small intestine bacterial overgrowth. Gastroenterology 91, 1447–1451.
Lee YK & Salminen S (1995): The coming age of probiotics. Trends Food Sci. Technol. 6, 241–245.
Leitzmann P, Heine W, Wutzke KD, von Bismarck P, Dorlöchter L, Miera O, Bührlen M, Corneließen BW & Höcker C (1998): Blood glucose, gastric emptying, and oro-coecal transit time after a conventional breakfast vs. a Kollath breakfast. Z. Ernährungswiss. 37, 31–37.
Meydani SN & Ha WK (2000): Immunologic effects of yogurt. Am. J. Clin. Nutr. 71, 861–872.
Pfeiffer A & Rosat JP (1999): Probiotics in alimentation: clinical evidence for their enhancement of the natural immunity of the gut. In Probiotics, Other Nutritional Factors and Intestinal Microflora Nestle Nutrition Workshop series, Vol. 42, eds LA Hanson, RH Yolken, pp 243–255. Philadelphia: Lippincott-Raven.
Radke M, Heine W, Wutzke KD, Leitzmann P & Walther F (1995): Tracer kinetic studies on a methionine-supplemented soy-based infant formula using 1-13C- and 15N-methionine as tracers. J. Pediatr. Gastroenterol. Nutr. 21, 209–214.
Robins-Browne RM & Levine MM (1981): The fate of ingested lactobacilli in the proximal small intestine. Am. J. Clin. Nutr. 34, 514–519.
Sarno S, Erasmus LP, Haslbeck M & Holzl R (1993): Orocaecal transit-time by the H2 method: effects of definitions of caecal entry and test meal. Ital. J. Gastroenterol. 25, 55–64.
Schoeller DA, Klein PD, Watkins JB, Heim T & MacLean Jr WC (1980): 13C abundances of nutrients and the effect of variations in 13C isotopic abundances of test meals formulated for 13CO2 breath tests. Am. J. Clin. Nutr. 33, 2375–2385.
Slater C, Preston T & Weaver LT (2001): Stable isotopes and the international system of units. Rapid Commun. Mass Spectrom. 15, 1270–1273.
Vantrappen GR, Rutgeerts PJ, Ghoos YF & Hiele MI (1989): Mixed triglyceride breath test: a noninvasive test of pancreatic lipase activity in the duodenum. Gastroenterology 96, 1126–1134.
Wutzke KD & Glasenapp B (2004): The use of 13C-labelled glycosyl ureides for evaluation of orocaecal transit time. Eur. J. Clin. Nutr. 58, 568–572.
Wutzke KD, Heine W, Drescher U, Richter I & Plath C (1983): 15N-labelled yeast protein – a valid tracer for calculating whole-body protein parameters in infants: a comparison between [15N]-yeast protein and [15N]-glycine. Hum. Nutr. Clin. Nutr. 37C, 317–327.
Wutzke KD, Heine W, Völker T, Kreyenbrink F & Krawielitzki K (1984): Präparative Darstellung von hochangereichertem 15N-markiertem Hefeprotein. Isotopenpraxis 20, 90–93.
Wutzke KD, Heine W, Plath C, Müller M & Uhlemann M (1992): Whole-body protein parameters in premature infants: a comparison of different 15N tracer substances and different methods. Pediatr. Res. 31, 95–101.
Wutzke KD, Heine WE, Plath C, Leitzmann P, Radke M, Mohr C, Richter I, Gülzow HU & Hobusch D (1997): Evaluation of oro-coecal transit time: a comparison of the lactose-[13C, 15N]ureide 13CO2- and the lactulose H2- breath test. Eur. J. Clin. Nutr. 51, 11–19.
Wutzke KD, Heine WE, Köster D, Muscheites J, Mix M, Mohr C & Wigger M (2001): Metabolic effects of Hay’ diet. Isotopes Environ. Health Stud. 37, 227–237.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guarantor: KD Wutzke.
Contributor: IO was principal investigator and doctoral candidate of KDW.
Rights and permissions
About this article
Cite this article
Wutzke, K., Oetjens, I. 13C- and 15N-incorporation of doubly stable isotope labelled Lactobacillus johnsonii in humans. Eur J Clin Nutr 59, 1167–1172 (2005). https://doi.org/10.1038/sj.ejcn.1602227
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602227
Keywords
This article is cited by
-
The effect of pre- and probiotics on the colonic ammonia metabolism in humans as measured by lactose-[15N2]ureide
European Journal of Clinical Nutrition (2010)
-
The metabolic fate of doubly stable isotope labelled heat-killed Lactobacillus johnsonii in humans
European Journal of Clinical Nutrition (2008)
-
The duration of enzyme induction in orocaecal transit time measurements
European Journal of Clinical Nutrition (2007)
-
15N-excretion of heat-killed Lactobacillus casei in humans
European Journal of Clinical Nutrition (2006)