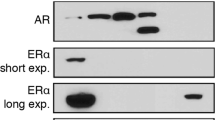
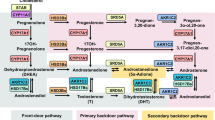
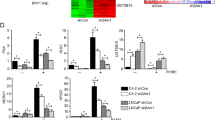
Abstract
Recent studies indicate that estrogen-related receptors (ERRs) are involved in similar estrogen receptor (ER) regulatory pathways and play roles in energy and lipid metabolism. Here, we analysed the functional role of ERRβ in prostate cancer cell growth regulation in an androgen-sensitive and androgen-insensitive prostate cancer cell lines. ERRβ was expressed in normal human prostates, but exhibited a reduced expression in prostate cancer lesions. Stable ERRβ expression suppressed significantly cell proliferation and tumorigenicity of LNCaP and DU145 cells, accompanied by an S-phase suppression and increased p21 expression. Reporter and chromatin immunoprecipitation assays showed that ERRβ could directly transactivate p21 gene promoter, which could be further enhanced by peroxisome proliferator-activated receptor-γ coactivator-1α. Truncation analysis showed that ERRβ-mediated p21 transactivation and prostate cancer cell growth inhibition required intact DNA-binding domain and AF2 domains in ERRβ. Interestingly, ERRβ displayed a cell cycle associated downregulated expression pattern in ERRβ-transduced and non-transduced cells. Finally, we showed that ERRβ-mediated growth inhibition could be potentiated by an ERRβ/γ agonist DY131. Knockdown of ERRβ by RNA interference could reduce the DY131-induced growth inhibition in prostate cancer cells. Taken together, our findings indicate that ERRβ performs a tumor suppressing function in prostate cancer cells, and targeting ERRβ could be a potential therapeutic strategy for prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Accession codes
References
Bookout AL, Jeong Y, Downes M, Yu RT, Evans RM, Mangelsdorf DJ . (2006). Anatomical profiling of nuclear receptor expression reveals a hierarchical transcriptional network. Cell 126: 789–799.
Castet A, Herledan A, Bonnet S, Jalaguier S, Vanacker JM, Cavaillès V . (2006). Receptor-interacting protein 140 differentially regulates estrogen receptor-related receptor transactivation depending on target genes. Mol Endocrinol 20: 1035–1047.
Chan PS, Chan LW, Xuan JW, Chin JL, Choi HL, Chan FL . (1999). In situ hybridization study of PSP94 (prostatic secretory protein of 94 amino acids) expression in human prostates. Prostate 41: 99–109.
Chen F, Zhang Q, McDonald T, Davidoff MJ, Bailey W, Bai C et al. (1999). Identification of two hERR2-related novel nuclear receptors utilizing bioinformatics and inverse PCR. Gene 228: 101–109.
Cheung CP, Yu S, Wong KB, Chan LW, Lai FM, Wang X et al. (2005). Expression and functional study of estrogen receptor-related receptors in human prostatic cells and tissues. J Clin Endocrinol Metab 90: 1830–1844.
Fujimoto J, Nakagawa Y, Toyoki H, Sakaguchi H, Sato E, Tamaya T . (2005). Estrogen-related receptor expression in placenta throughout gestation. J Steroid Biochem Mol Biol 94: 67–69.
Giguère V . (2002). To ERR in the estrogen pathway. Trends Endocrinol Metab 13: 220–225.
Giguère V, Yang N, Segui P, Evans RM . (1988). Identification of a new class of steroid hormone receptors. Nature 331: 91–94.
Griffiths K . (2000). Estrogens and prostatic disease. International Prostate Health Council Study Group. Prostate 45: 87–100.
Ho E, Boileau TW, Bray TM . (2004). Dietary influences on endocrine-inflammatory interactions in prostate cancer development. Arch Biochem Biophys 428: 109–117.
Ho SM . (2004). Estrogens and anti-estrogens: key mediators of prostate carcinogenesis and new therapeutic candidates. J Cell Biochem 91: 491–503.
Huang CY, Beliakoff J, Li X, Lee J, Li X, Sharma M et al. (2005). HZimp7, a novel PIAS-like protein, enhances androgen receptor-mediated transcription and interacts with SWI/SNF-like BAF complexes. Mol Endocrinol 19: 2915–2929.
Huss JM, Kopp RP, Kelly DP . (2002). Peroxisome proliferator-activated receptor coactivator-1α (PGC-1α) coactivates the cardiac-enriched nuclear receptors estrogen-related receptor-α and -γ. J Biol Chem 277: 40265–40274.
Huss JM, Torra IP, Staels B, Giguère V, Kelly DP . (2004). Estrogen-related receptor α directs peroxisome proliferator-activated receptor α signaling in the transcriptional control of energy metabolism in cardiac and skeletal muscle. Mol Cell Biol 24: 9079–9091.
Ichida M, Nemoto S, Finkel T . (2002). Identification of a specific molecular repressor of the peroxisome proliferator-activator receptor γ coactivator-1α (PGC-1α). J Biol Chem 277: 50991–50995.
Kamei Y, Ohizumi H, Fujitani Y, Nemoto T, Tanaka T, Takahashi N et al. (2003). PPARγ coactivator 1β/ERR ligand 1 is an ERR protein ligand, whose expression induces a high-energy expenditure and antagonizes obesity. Proc Natl Acad Sci USA 100: 12378–12383.
Litvinov IV, Griend DJ, Antony L, Dalrymple S, De Marzo AM, Drake CG et al. (2006). Androgen receptor as a licensing factor for DNA replication in androgen-sensitive prostate cancer cells. Proc Natl Acad Sci USA 103: 15085–15090.
Liu Y . (2006). Fatty acid oxidation is a dominant bioenergetic pathway in prostate cancer. Prostate Cancer Prostatic Dis 9: 230–234.
Lu D, Kiriyama Y, Lee KY, Giguère V . (2001). Transcriptional regulation of the estrogen-inducible pS2 breast cancer marker gene by the ERR family of orphan nuclear receptors. Cancer Res 61: 6755–6761.
Lui K, Huang Y, Choi HL, Yu S, Wong KB, Chen S et al. (2006). Molecular cloning and functional study of rat estrogen receptor-related receptor γ in rat prostatic cells. Prostate 66: 1600–1619.
Luo J, Sladek R, Bader JA, Matthyssen A, Rossant J, Giguère V . (1997). Placental abnormalities in mouse embryos lacking the orphan nuclear receptor ERR-β. Nature 388: 778–782.
Luo J, Sladek R, Carrier J, Bader JA, Richard D, Giguère V . (2003). Reduced fat mass in mice lacking orphan nuclear receptor estrogen-related receptor α. Mol Cell Biol 23: 7947–7956.
Mitsunaga K, Araki K, Mizusaki H, Morohashi K, Haruna K, Nakagata N et al. (2004). Loss of PGC-specific expression of the orphan nuclear receptor ERR-β results in reduction of germ cell number in mouse embryos. Mech Dev 121: 237–246.
Pettersson K, Svensson K, Mattsson R, Carlsson B, Ohlsson R, Berkenstam A . (1996). Expression of a novel member of estrogen response element-binding nuclear receptors is restricted to the early stages of chorion formation during mouse embryogenesis. Mech Dev 54: 211–223.
Prins GS, Huang L, Birch L, Pu Y . (2006). The role of estrogens in normal and abnormal development of the prostate gland. Ann N Y Acad Sci 1089: 1–13.
Puigserver P, Spiegelman BM . (2003). Peroxisome proliferator-activator receptor-γ coactivator 1α (PGC-1α): transcriptional coactivator and metabolic regulator. Endocr Rev 24: 78–90.
Schreiber SN, Emter R, Hock MB, Knutti D, Cardenas J, Podvinec M et al. (2004). The estrogen-related receptor α (ERRα) functions in PPARγ coactivator 1α (PGC-1α)-induced mitochondrial biogenesis. Proc Natl Acad Sci USA 101: 6472–6477.
Sharma M, Li X, Wang Y, Zarnegar M, Huang CY, Palvimo JJ et al. (2003). HZimp10 is an androgen receptor co-activator and forms a complex with SUMO-1 at replication foci. EMBL J 22: 6101–6114.
Sladek R, Bader JA, Giguère V . (1997). The orphan nuclear receptor estrogen-related receptor α is a transcriptional regulator of the human medium-chain acyl coenzyme A dehydrogenase gene. Mol Cell Biol 17: 5400–5409.
Soriano FX, Liesa M, Bach D, Chan DC, Palacín M, Zorzano A . (2006). Evidence for a mitochondrial regulatory pathway defined peroxisome proliferator-activated receptor-γ coactivator-1α, estrogen-related receptor-α, and mitofusin 2. Diabetes 55: 1783–1791.
Stewart SA, Dykxhoorn DM, Palliser D, Mizuno H, Yu EY, An DS et al. (2003). Lentivirus-delivered stable gene silencing by RNAi in primary cells. RNA 9: 493–501.
Tanaka S, Kunath T, Hadjantonakis AK, Nagy A, Rossant J . (1998). Promotion of trophoblast stem cell proliferation by FGF4. Science 282: 2072–2075.
Trapp T, Holsboer F . (1996). Nuclear orphan receptor as a repressor of glucocorticoid receptor transcriptional activity. J Biol Chem 271: 9879–9882.
Vanacker JM, Pettersson K, Gustafsson JÅ, Laudet V . (1999). Transcriptional targets shared by estrogen receptor-related receptors (ERRs) and estrogen receptor (ER) α, but not by ERβ. EMBO J 18: 4270–4279.
Vankoningsloo S, Piens M, Lecocq C, Gilson A, De Pauw A, Renard P et al. (2005). Mitochondrial dysfunction induces triglyceride accumulation in 3T3-L1 cells: role of fatty acid β-oxidation and glucose. J Lipid Res 46: 1133–1149.
Villena JA, Hock MB, Chang WY, Barcas JE, Giguère V, Kralli A . (2007). Orphan nuclear receptor estrogen-related receptor α is essential for adaptive thermogenesis. Proc Natl Acad Sci USA 104: 1418–1423.
Waga S, Hannon GJ, Beach D, Stillman B . (1994). The p21 inhibitor of cyclin-dependent kinases controls DNA replication by interaction with PCNA. Nature 369: 574–578.
Wende AR, Huss JM, Schaeffer PJ, Giguère V, Kelly DP . (2005). PGC-1α coactivates PDK4 gene expression via the orphan nuclear receptor ERRα: a mechanism for transcriptional control of muscle glucose metabolism. Mol Cell Biol 25: 10684–10694.
Weihua Z, Warner M, Gustafsson JA . (2002). Estrogen receptor beta in the prostate. Mol Cell Endocrinol 193: 1–5.
Wright ME, Tsai MJ, Aebersold R . (2003). Androgen receptor represses the neuroendocrine transdifferentiation process in prostate cancer cells. Mol Endocrinol 17: 1726–1737.
Xie W, Hong H, Yang NN, Lin RJ, Simon CM, Stallcup MR et al. (1999). Constitutive activation of transcription and binding of coactivator by estrogen-related receptors 1 and 2. Mol Endocrinol 13: 2151–2162.
Yang X, Downes M, Yu RT, Bookout AL, He W, Straume M et al. (2006). Nuclear receptor expression links the circadian clock to metabolism. Cell 126: 801–810.
Yu DD, Forman BM . (2005). Identification of an agonist ligand for estrogen-related receptors ERRβ/γ. Bioorg Med Chem Lett 15: 1311–1313.
Yu S, Wang X, Ng CF, Chen S, Chan FL . (2007). ERRγ suppresses cell proliferation and tumor growth androgen-sensitive and androgen-insensitive prostate cancer cells and its implication as a therapeutic target for prostate cancer. Cancer Res 67: 4904–4914.
Zhou W, Liu Z, Wu J, Liu J, Hyder SM, Antoniou E et al. (2006). Identification and characterization of two novel splicing isoforms of human estrogen-related receptor β. J Clin Endocrinol Metab 91: 569–579.
Acknowledgements
This research was supported by a Competitive Earmarked Research Grant from Research Grants Council (CUHK4411/06M) and Li Ka-Shing Institute of Health Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, S., Wong, Y., Wang, X. et al. Orphan nuclear receptor estrogen-related receptor-β suppresses in vitro and in vivo growth of prostate cancer cells via p21WAF1/CIP1 induction and as a potential therapeutic target in prostate cancer. Oncogene 27, 3313–3328 (2008). https://doi.org/10.1038/sj.onc.1210986
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210986
Keywords
This article is cited by
-
SIRT1 mediates breast cancer development and tumorigenesis controlled by estrogen-related receptor β
Breast Cancer (2024)
-
Expression of estrogen-related receptors in ovarian cancer and impact on survival
Journal of Cancer Research and Clinical Oncology (2021)
-
Nuclear receptor HNF4α performs a tumor suppressor function in prostate cancer via its induction of p21-driven cellular senescence
Oncogene (2020)
-
Identification of transcription factor co-regulators that drive prostate cancer progression
Scientific Reports (2020)
-
NEDDylation negatively regulates ERRβ expression to promote breast cancer tumorigenesis and progression
Cell Death & Disease (2020)