Abstract
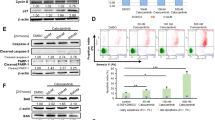
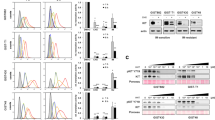
Activating mutations in the Kit receptor are frequently observed in various malignancies, pointing Kit as a molecule of interest for drug inhibition. When mutated on Asp 816 (corresponding to Asp 814 in the mouse), as preferentially found in human mastocytosis and acute myeloid leukemia, Kit became non-sensitive to imatinib mesylate (Gleevec). Erythroleukemic cells isolated from Spi-1/PU.1 transgenic mice express Kit mutated at codon 814 (KitD814Y or KitD814V) or codon 818 (KitD818Y). Using these cells in vitro, we demonstrate that the tyrosine kinase inhibitor SU5416 (Semaxinib) induces growth arrest and apoptosis independent of the mutation type by inhibiting the functions of Kit, including Kit autophosphorylation and activation of Akt, Erk1/Erk2 and Stat3 downstream signaling pathways. These findings indicate that SU5416 may be a promising tool to kill cancer cells driven by Kit oncogenic mutations that are resistant to treatment with imatinib mesylate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Akin C, Fumo G, Yavuz AS, Lipsky PE, Neckers L, Metcalfe DD . (2004). A novel form of mastocytosis associated with a transmembrane c-kit mutation and response to imatinib. Blood 103: 3222–3225.
Beghini A, Peterlongo P, Ripamonti CB, Larizza L, Cairoli R, Morra E et al. (2000). C-kit mutations in core binding factor leukemias. Blood 95: 726–727.
Chen H, Isozaki K, Kinoshita K, Ohashi A, Shinomura Y, Matsuzawa Y et al. (2003). Imatinib inhibits various types of activating mutant kit found in gastrointestinal stromal tumors. Int J Cancer 105: 130–135.
Fiedler W, Mesters R, Tinnefeld H, Loges S, Staib P, Duhrsen U et al. (2003). A phase 2 clinical study of SU5416 in patients with refractory acute myeloid leukemia. Blood 102: 2763–2767.
Fong TA, Shawver LK, Sun L, Tang C, App H, Powell TJ et al. (1999). SU5416 is a potent and selective inhibitor of the vascular endothelial growth factor receptor (Flk-1/KDR) that inhibits tyrosine kinase catalysis, tumor vascularization, and growth of multiple tumor types. Cancer Res 59: 99–106.
Giles FJ, Stopeck AT, Silverman LR, Lancet JE, Cooper MA, Hannah AL et al. (2003). SU5416, a small molecule tyrosine kinase receptor inhibitor, has biologic activity in patients with refractory acute myeloid leukemia or myelodysplastic syndromes. Blood 102: 795–801.
Heinrich MC, Griffith DJ, Druker BJ, Wait CL, Ott KA, Zigler AJ . (2000). Inhibition of c-kit receptor tyrosine kinase activity by STI 571, a selective tyrosine kinase inhibitor. Blood 96: 925–932.
Heymach JV, Desai J, Manola J, Davis DW, McConkey DJ, Harmon D et al. (2004). Phase II study of the antiangiogenic agent SU5416 in patients with advanced soft tissue sarcomas. Clin Cancer Res 10: 5732–5740.
Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S et al. (1998). Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science 279: 577–580.
Itokawa T, Nokihara H, Nishioka Y, Sone S, Iwamoto Y, Yamada Y et al. (2002). Antiangiogenic effect by SU5416 is partly attributable to inhibition of Flt-1 receptor signaling. Mol Cancer Ther 1: 295–302.
Kosmider O, Denis N, Lacout C, Vainchenker W, Dubreuil P, Moreau-Gachelin F . (2005). Kit-activating mutations cooperate with Spi-1/PU.1 overexpression to promote tumorigenic progression during erythroleukemia in mice. Cancer Cell 8: 467–478.
Krystal GW, Honsawek S, Kiewlich D, Liang C, Vasile S, Sun L et al. (2001). Indolinone tyrosine kinase inhibitors block Kit activation and growth of small cell lung cancer cells. Cancer Res 61: 3660–3668.
Lennartsson J, Jelacic T, Linnekin D, Shivakrupa R . (2005). Normal and oncogenic forms of the receptor tyrosine kinase kit. Stem Cells 23: 16–43.
Longley BJ, Tyrrell L, Lu SZ, Ma YS, Langley K, Ding TG et al. (1996). Somatic c-KIT activating mutation in urticaria pigmentosa and aggressive mastocytosis: establishment of clonality in a human mast cell neoplasm. Nat Genet 12: 312–314.
Ma Y, Zeng S, Metcalfe DD, Akin C, Dimitrijevic S, Butterfield JH et al. (2002). The c-KIT mutation causing human mastocytosis is resistant to STI571 and other KIT kinase inhibitors; kinases with enzymatic site mutations show different inhibitor sensitivity profiles than wild-type kinases and those with regulatory-type mutations. Blood 99: 1741–1744.
Mesters RM, Padro T, Bieker R, Steins M, Kreuter M, Goner M et al. (2001). Stable remission after administration of the receptor tyrosine kinase inhibitor SU5416 in a patient with refractory acute myeloid leukemia. Blood 98: 241–243.
Moreau-Gachelin F, Wendling F, Molina T, Denis N, Titeux M, Grimber G et al. (1996). Spi-1/PU.1 transgenic mice develop multistep erythroleukemias. Mol Cell Biol 16: 2453–2463.
Ning ZQ, Li J, Arceci RJ . (2001). Signal transducer and activator of transcription 3 activation is required for Asp(816) mutant c-Kit-mediated cytokine-independent survival and proliferation in human leukemia cells. Blood 97: 3559–3567.
O'Farrell AM, Yuen HA, Smolich B, Hannah AL, Louie SG, Hong W et al. (2004). Effects of SU5416, a small molecule tyrosine kinase receptor inhibitor, on FLT3 expression and phosphorylation in patients with refractory acute myeloid leukemia. Leuk Res 28: 679–689.
Pignon JM, Giraudier S, Duquesnoy P, Jouault H, Imbert M, Vainchenker W et al. (1997). A new c-kit mutation in a case of aggressive mast cell disease. Br J Haematol 96: 374–376.
Smolich BD, Yuen HA, West KA, Giles FJ, Albitar M, Cherrington JM . (2001). The antiangiogenic protein kinase inhibitors SU5416 and SU6668 inhibit the SCF receptor (c-kit) in a human myeloid leukemia cell line and in acute myeloid leukemia blasts. Blood 97: 1413–1421.
Wang YY, Zhou GB, Yin T, Chen B, Shi JY, Liang WX et al. (2005). AML1-ETO and C-KIT mutation/overexpression in t(8;21) leukemia: Implication in stepwise leukemogenesis and response to Gleevec. Proc Natl Acad Sci USA 102: 1104–1109.
Yee KW, O'Farrell AM, Smolich BD, Cherrington JM, McMahon G, Wait CL et al. (2002). SU5416 and SU5614 inhibit kinase activity of wild-type and mutant FLT3 receptor tyrosine kinase. Blood 100: 2941–2949.
Zermati Y, De Sepulveda P, Feger F, Letard S, Kersual J, Casteran N et al. (2003). Effect of tyrosine kinase inhibitor STI571 on the kinase activity of wild-type and various mutated c-kit receptors found in mast cell neoplasms. Oncogene 22: 660–664.
Acknowledgements
We thank C Guillouf and R Monni for many helpful discussions and F Wendling for comments on the manuscript. This work was supported by grants from the Institut National de la Santé Et de la Recherche Médicale, the Institut Curie (Paris), the Association pour la Recherche sur le Cancer, the Fondation de France and the Association Christelle Bouillot.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kosmider, O., Denis, N., Dubreuil, P. et al. Semaxinib (SU5416) as a therapeutic agent targeting oncogenic Kit mutants resistant to imatinib mesylate. Oncogene 26, 3904–3908 (2007). https://doi.org/10.1038/sj.onc.1210159
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210159
Keywords
This article is cited by
-
The activation loop tyrosine 823 is essential for the transforming capacity of the c-Kit oncogenic mutant D816V
Oncogene (2015)
-
Identification of molecular and cytogenetic risk factors for unfavorable core-binding factor-positive adult AML with post-remission treatment outcome analysis including transplantation
Bone Marrow Transplantation (2014)
-
Proteasome inhibition upregulates Bim and induces caspase-3-dependent apoptosis in human mast cells expressing the Kit D816V mutation
Cell Death & Disease (2012)
-
Pharmacological targeting of the KIT growth factor receptor: a therapeutic consideration for mast cell disorders
British Journal of Pharmacology (2008)