Abstract
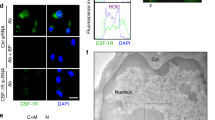
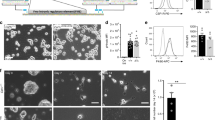
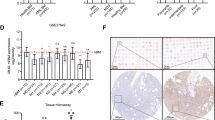
Gene translocations that repress the function of the Runx1 transcription factor play a critical role in the development of myeloid leukemia. In this report, we demonstrate that Runx1 precisely regulates c-fms (CSF-1 receptor) gene expression. Runx1 controlled expression by binding to multiple sites within the mouse c-fms gene, allowing interaction between promoter and downstream enhancer elements. The runx1 and c-fms genes showed an identical pattern of expression in mature macrophages. Runx1 expression was repressed in CSF-1 stimulated, proliferating bone marrow-derived macrophages (BMM) and significantly increased in quiescent, CSF-1 starved cells. The RAW264.7 and Mono-Mac-6, macrophage-like cell lines expressed low levels of Runx1 and both showed growth arrest and cell death with ectopic expression of Runx1. The EM-3 cell line, which represents an early myeloid progenitor cell line, showed growth arrest with Runx1 expression in the absence of any detectable changes in cell differentiation. These findings suggest that Runx1 regulates growth and survival of myeloid cells and provide a novel insight into the role of Runx family gene translocations in leukemogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Adya N, Stacy T, Speck NA and Liu PP . (1998). Mol. Cell. Biol., 18, 7432–7443.
Castilla LH, Garrett L, Adya N, Orlic D, Dutra A, Anderson S, Owens J, Eckhaus M, Bodine D and Liu PP . (1999). Nat. Genet, 23, 144–146.
Cecchini MG, Hofstetter W, Halasy J, Wetterwald A and Felix R . (1997). Mol. Reprod. Dev., 46, 75–83; discussion 83–84.
Cochet O, Kenigsberg M, Delumeau I, Duchesne M, Schweighoffer F, Tocque B and Teillaud JL . (1998). Mol. Immunol., 35, 1097–1110.
Dai XM, Ryan GR, Hapel AJ, Dominguez MG, Russell RG, Kapp S, Sylvestre V and Stanley ER . (2002). Blood, 99, 111–120.
Downing JR, Head DR, Curcio-Brint AM, Hulshof MG, Motroni TA, Raimondi SC, Carroll AJ, Drabkin HA, Willman C and Theil KS et al. (1993). Blood, 81, 2860–2865.
Follows GA, Tagoh H, Lefevre P, Hodge D, Morgan GJ and Bonifer C . (2003). Embo. J., 22, 2798–2809.
Fukamachi H and Ito K . (2004). Oncogene, 23, 4330–4335.
Heibert SW, Lutterbach B, Durst K, Wang L, Linggi B, Wu S, Wood L, Amann J, King D and Hou Y . (2001). Cancer Chemother. Pharmacol., 48 (Suppl 1), S31–S34.
Himes SR, Tagoh H, Goonetilleke N, Sasmono T, Oceandy D, Clark R, Bonifer C and Hume DA . (2001). J. Leukoc. Biol., 70, 812–820.
Hume DA, Yue X, Ross IL, Favot P, Lichanska A and Ostrowski MC . (1997). Mol. Reprod. Dev., 46, 46–52; discussion 52–53.
Ichikawa M, Asai T, Saito T, Yamamoto G, Seo S, Yamazaki I, Yamagata T, Mitani K, Chiba S, Hirai H, Ogawa S and Kurokawa M . (2004). Nat. Med., 10, 299–304.
Levanon D, Glusman G, Bettoun D, Ben-Asher E, Negreanu V, Bernstein Y, Harris-Cerruti C, Brenner O, Eilam R, Lotem J, Fainaru O, Goldenberg D, Pozner A, Woolf E, Xiao C, Yarmus M and Groner Y . (2003). Blood Cells Mol. Dis., 30, 161–163.
Li QL, Ito K, Sakakura C, Fukamachi H, Inoue K, Chi XZ, Lee KY, Nomura S, Lee CW, Han SB, Kim HM, Kim WJ, Yamamoto H, Yamashita N, Yano T, Ikeda T, Itohara S, Inazawa J, Abe T, Hagiwara A, Yamagishi H, Ooe A, Kaneda A, Sugimura T, Ushijima T, Bae SC and Ito Y . (2002). Cell, 109, 113–124.
Linggi B, Muller-Tidow C, van de Locht L, Hu M, Nip J, Serve H, Berdel WE, van der Reijden B, Quelle DE, Rowley JD, Cleveland J, Jansen JH, Pandolfi PP and Hiebert SW . (2002). Nat. Med., 8, 743–750.
Lutterbach B and Hiebert SW . (2000). Gene, 245, 223–235.
Maruyama F, Yang P, Stass SA, Cork A, Freireich EJ, Lee MS and Chang KS . (1993). Cancer Res., 53, 4449–4451.
Meyers S, Lenny N and Hiebert SW . (1995). Mol. Cell. Biol., 15, 1974–1982.
Miyoshi H, Kozu T, Shimizu K, Enomoto K, Maseki N, Kaneko Y, Kamada N and Ohki M . (1993). Embo. J., 12, 2715–2721.
Mulloy JC, Cammenga J, MacKenzie KL, Berguido FJ, Moore MA and Nimer SD . (2002). Blood, 99, 15–23.
Okuda T, Cai Z, Yang S, Lenny N, Lyu CJ, van Deursen JM, Harada H and Downing JR . (1998). Blood, 91, 3134–3143.
Okuda T, van Deursen J, Hiebert SW, Grosveld G and Downing JR . (1996). Cell, 84, 321–330.
Petrovick MS, Hiebert SW, Friedman AD, Hetherington CJ, Tenen DG and Zhang DE . (1998). Mol. Cell. Biol., 18, 3915–3925.
Sasaki K, Yagi H, Bronson RT, Tominaga K, Matsunashi T, Deguchi K, Tani Y, Kishimoto T and Komori T . (1996). Proc. Natl. Acad. Sci. USA, 93, 12359–12363.
Sasmono RT, Oceandy D, Pollard JW, Tong W, Pavli P, Wainwright BJ, Ostrowski MC, Himes SR and Hume DA . (2003). Blood, 101, 1155–1163.
Satake M, Inuzuka M, Shigesada K, Oikawa T and Ito Y . (1992). Jpn J. Cancer Res., 83, 714–722.
Sawyers CL . (1999). N. Engl. J. Med., 340, 1330–1340.
Sudo T, Nishikawa S, Ogawa M, Kataoka H, Ohno N, Izawa A and Hayashi S . (1995). Oncogene, 11, 2469–2476.
Tagoh H, Himes R, Clarke D, Leenen PJ, Riggs AD, Hume D and Bonifer C . (2002). Genes Dev., 16, 1721–1737.
Thompson PW, Lucas GS, Davies MF and Whittaker JA . (1991). Cancer Genet. Cytogenet., 55, 269–271.
Tracey WD and Speck NA . (2000). Semin. Cell. Dev. Biol., 11, 337–342.
Wang Q, Stacy T, Binder M, Marin-Padilla M, Sharpe AH and Speck NA . (1996). Proc. Natl. Acad. Sci. USA, 93, 3444–3449.
Wang SW and Speck NA . (1992). Mol. Cell. Biol., 12, 89–102.
Yang Y, Wang W, Cleaves R, Zahurak M, Cheng L, Civin CI and Friedman AD . (2002). Cancer Res., 62, 2232–2235.
Zhang DE, Hetherington CJ, Meyers S, Rhoades KL, Larson CJ, Chen HM, Hiebert SW and Tenen DG . (1996a). Mol. Cell. Biol., 16, 1231–1240.
Zhang DE, Hohaus S, Voso MT, Chen HM, Smith LT, Hetherington CJ and Tenen DG . (1996b). Curr. Top Microbiol. Immunol., 211, 137–147.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Himes, S., Cronau, S., Mulford, C. et al. The Runx1 transcription factor controls CSF-1-dependent and -independent growth and survival of macrophages. Oncogene 24, 5278–5286 (2005). https://doi.org/10.1038/sj.onc.1208657
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208657
Keywords
This article is cited by
-
Expression and localization of MrgprD in mouse intestinal tract
Cell and Tissue Research (2019)
-
Deletion of a Csf1r enhancer selectively impacts CSF1R expression and development of tissue macrophage populations
Nature Communications (2019)
-
The evolution of the macrophage-specific enhancer (Fms intronic regulatory element) within the CSF1R locus of vertebrates
Scientific Reports (2017)
-
CARD9 facilitates microbe-elicited production of reactive oxygen species by regulating the LyGDI-Rac1 complex
Nature Immunology (2009)