Abstract
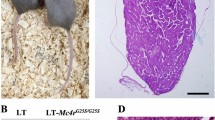
Germ cell tumors (GCTs) are among the most common malignancies in young men. We have previously documented that patients with GCT frequently produce serum antibodies directed against proteins encoded by human endogenous retrovirus (HERV) type K sequences. Transcripts originating from the env gene of HERV-K, including the rec-relative of human immunodeficiency virus rev, are highly expressed in GCTs. We report here that mice that inducibly express HERV-K rec show a disturbed germ cell development and may exhibit, by 19 months of age, changes reminiscent of carcinoma in situ, the predecessor lesion of classic seminoma in humans. This provides the first direct evidence that the expression of a human endogenous retroviral gene previously established as a marker in human germ cell tumors may contribute to organ-specific tumorigenesis in a transgenic mouse model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Barbulescu M, Turner G, Seaman MI, Deinard AS, Kidd KK and Lenz J . (1999). Curr. Biol., 9, 861–868.
Barna M, Hawe N, Niswander L and Pandolfi PP . (2000). Nat. Genet., 25, 166–172.
Boese A, Sauter M, Galli U, Best B, Herbst H, Mayer J, Kremmer E, Roemer K and Mueller-Lantzsch N . (2000). Oncogene, 19, 4328–4336.
Buaas FW, Kirsh AL, Sharma M, McLean DJ, Morris JL, Griswold MD, de Rooij DG and Braun RE . (2004). Nat. Genet., 36, 647–652 (Epub 2004 May 2023).
Costoya JA, Hobbs RM, Barna M, Cattoretti G, Manova K, Sukhwani M, Orwig KE, Wolgemuth DJ and Pandolfi PP . (2004). Nat. Genet., 36, 653–659 (Epub 2004 May 2023).
Herbst H, Sauter M and Mueller-Lantzsch N . (1996). Am. J. Pathol., 149, 1727–1735.
Jurka J . (1998). Curr. Opin. Struct. Biol., 8, 333–337.
Leib-Mosch C and Seifarth W . (1996). Virus Genes, 11, 133–145.
Looijenga LH and Oosterhuis JW . (1999). Rev. Reprod., 4, 90–100.
Lower R, Boller K, Hasenmaier B, Korbmacher C, Muller-Lantzsch N, Lower J and Kurth R . (1993). Proc. Natl. Acad. Sci. USA, 90, 4480–4484.
Lower R, Lower J and Kurth R . (1996). Proc. Natl. Acad. Sci. USA, 93, 5177–5184.
Lower R, Tonjes RR, Korbmacher C, Kurth R and Lower J . (1995). J. Virol., 69, 141–149.
Magin C, Lower R and Lower J . (1999). J. Virol., 73, 9496–9507.
Mangeney M and Heidmann T . (1998). Proc. Natl. Acad. Sci. USA, 95, 14920–14925.
Mayer J, Sauter M, Racz A, Scherer D, Mueller-Lantzsch N and Meese E . (1999). Nat. Genet., 21, 257–258.
McConnell MJ, Chevallier N, Berkofsky-Fessler W, Giltnane JM, Malani RB, Staudt LM and Licht JD . (2003). Mol. Cell. Biol., 23, 9375–9388.
Meng X, de Rooij DG, Westerdahl K, Saarma M and Sariola H . (2001). Cancer Res., 61, 3267–3271.
Meng X, Lindahl M, Hyvonen ME, Parvinen M, de Rooij DG, Hess MW, Raatikainen-Ahokas A, Sainio K, Rauvala H, Lakso M, Pichel JG, Westphal H, Saarma M and Sariola H . (2000). Science, 287, 1489–1493.
Moller H . (1993). Eur. Urol., 23, 8–13 (discussion 14–15).
Mostofi FK . (1980). Cancer, 45, 1735–1754.
Patience C, Wilkinson DA and Weiss RA . (1997). Trends Genet., 13, 116–120.
Sauter M, Roemer K, Best B, Afting M, Schommer S, Seitz G, Hartmann M and Mueller-Lantzsch N . (1996). Cancer Res., 56, 4362–4365.
Skakkebaek NE, Berthelsen JG, Giwercman A and Muller J . (1987). Int. J. Androl., 10, 19–28.
Veeramachaneni DN and Vandewoude S . (1999). Int. J. Androl., 22, 97–101.
Yang J, Bogerd HP, Peng S, Wiegand H, Truant R and Cullen BR . (1999). Proc. Natl. Acad. Sci. USA, 96, 13404–13408.
Acknowledgements
We thank J Mayer, Institute for Human Genetics, University of Saarland Medical School, for discussion. This work was supported by grant Mu 452/5 from the German Research Foundation (DFG) to NM-L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Galli, U., Sauter, M., Lecher, B. et al. Human endogenous retrovirus rec interferes with germ cell development in mice and may cause carcinoma in situ, the predecessor lesion of germ cell tumors. Oncogene 24, 3223–3228 (2005). https://doi.org/10.1038/sj.onc.1208543
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208543
Keywords
This article is cited by
-
Human Endogenous Retroviruses as Biomedicine Markers
Virologica Sinica (2021)
-
A human endogenous retrovirus encoded protease potentially cleaves numerous cellular proteins
Mobile DNA (2019)
-
Transcriptional profiling of HERV-K(HML-2) in amyotrophic lateral sclerosis and potential implications for expression of HML-2 proteins
Molecular Neurodegeneration (2018)
-
Modulation of human endogenous retrovirus (HERV) transcription during persistent and de novo HIV-1 infection
Retrovirology (2015)
-
HERV-K(HML-2) rec and np9 transcripts not restricted to disease but present in many normal human tissues
Mobile DNA (2015)