Abstract
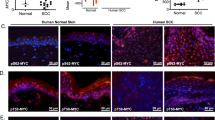
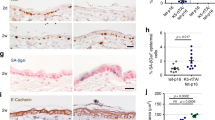
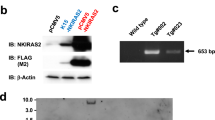
The homeostatic integrity of skin epidermis is maintained by a balance between keratinocyte proliferation, on one hand, and terminal differentiation combined with outward migration and shedding, on the other. Perturbation of this balance in favor of proliferation can result in hyperplasia and, potentially, tumorigenesis. We have previously described a reversible transgenic mouse model of epidermal neoplasia in which expression of an acutely regulatable form of Myc, MycERTAM, is targeted to epidermis via the involucrin promoter. In this model, sustained activation of MycERTAM induces a complex neoplastic lesion involving marked hyperplasia of less-differentiated suprabasal cells, angiogenesis and overt papillomatosis. Subsequent deactivation of MycERTAM triggers complete papilloma regression. Here, we provide evidence that Myc-induced papillomas are self-limiting because of the eventual differentiation of MycERTAM-expressing keratinocytes. Thus, keratinocyte differentiation eventually prevails over Myc-induced proliferation. We also show that regression of Myc-induced papillomas following MycERTAM deactivation occurs through a combination of growth arrest and irreversible differentiation. Finally, we demonstrate that transient deactivation of Myc is sufficient to expel keratinocytes irreversibly from the proliferative compartment and render them refractory to the mitogenic influence of subsequent Myc reactivation. Such observations illustrate the potential utility of even short-term inhibition of oncogenic lesions in the treatment of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Arnold I and Watt FM . (2001). Curr. Biol., 11, 558–568.
Chin L, Tam A, Pomerantz J, Wong M, Holash J, Bardeesy N, Shen Q, O'Hagan R, Pantginis J, Zhou H, Horner II JW, Cordon-Cardo C, Yancopoulos GD and DePinho RA . (1999). Nature, 400, 468–472.
Dotto GP . (1999). Crit. Rev. Oral. Biol. Med., 10, 442–457.
Evan G and Littlewood T . (1998). Science, 281, 1317–1322.
Evan GI and Vousden KH . (2001). Nature, 411, 342–348.
Gandarillas A, Goldsmith LA, Gschmeissner S, Leigh IM and Watt FM . (1999). Exp. Dermatol., 8, 71–79.
Gandarillas A and Watt FM . (1997). Genes Dev., 11, 2869–2882.
Hertle MD, Kubler MD, Leigh IM and Watt FM . (1992). J. Clin. Invest., 89, 1892–1901.
Huitfeldt HS, Heyden A, Clausen OP, Thrane EV, Roop D and Yuspa SH . (1991). Carcinogenesis, 12, 2063–2067.
Jain M, Arvanitis C, Chu K, Dewey W, Leonhardt E, Trinh M, Sundberg CD, Bishop JM and Felsher DW . (2002). Science, 297, 102–104.
Koster MI, Kim S, Mills AA, DeMayo FJ and Roop DR . (2004). Genes Dev., 18, 126–131.
Li ER, Owens DM, Djian P and Watt FM . (2000). Exp. Dermatol., 9, 431–438.
Liefer KM, Koster MI, Wang XJ, Yang A, McKeon F and Roop DR . (2000). Cancer Res., 60, 4016–4020.
Moore JP, Hancock DC, Littlewood TD and Evan GI . (1987). Oncogene Res., 2, 65–80.
Parsa R, Yang A, McKeon F and Green H . (1999). J. Invest. Dermatol., 113, 1099–1105.
Pelengaris S, Khan M and Evan GI . (2002). Cell, 109, 321–334.
Pelengaris S, Littlewood T, Khan M, Elia G and Evan G . (1999). Mol. Cell, 3, 565–577.
Pellegrini G, Dellambra E, Golisano O, Martinelli E, Fantozzi I, Bondanza S, Ponzin D, McKeon F and De Luca M . (2001). Proc. Natl. Acad. Sci. USA, 98, 3156–3161.
Polakowska RR, Piacentini M, Bartlett R, Goldsmith LA and Haake AR . (1994). Dev. Dyn., 199, 176–188.
Regnier M, Vaigot P, Darmon M and Prunieras M . (1986). J. Invest. Dermatol., 87, 472–476.
Roop DR, Krieg TM, Mehrel T, Cheng CK and Yuspa SH . (1988). Cancer Res., 48, 3245–3252.
Schweizer J, Kinjo M, Furstenberger G and Winter H . (1984). Cell, 37, 159–170.
Stoler A, Kopan R, Duvic M and Fuchs E . (1988). J. Cell. Biol., 107, 427–446.
Waikel RL, Wang XJ and Roop DR . (1999). Oncogene, 18, 4870–4878.
Watt FM . (1984). J. Cell Biol., 98, 16–21.
Yang A, Schweitzer R, Sun D, Kaghad M, Walker N, Bronson RT, Tabin C, Sharpe A, Caput D, Crum C and McKeon F . (1999). Nature, 398, 714–718.
Acknowledgements
We are grateful to all members of the Evan laboratory for critical advice and discussion, and especially to our UCSF colleagues Drs Lisa Coussens and Jeffrey Arbeit (the latter is now at Washington University, St Louis).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Flores, I., Murphy, D., Swigart, L. et al. Defining the temporal requirements for Myc in the progression and maintenance of skin neoplasia. Oncogene 23, 5923–5930 (2004). https://doi.org/10.1038/sj.onc.1207796
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207796
Keywords
This article is cited by
-
MYC, a downstream target of BRD-NUT, is necessary and sufficient for the blockade of differentiation in NUT midline carcinoma
Oncogene (2014)
-
Mitotic and mitogenic Wnt signalling
The EMBO Journal (2012)
-
Deciphering c-MYC-regulated genes in two distinct tissues
BMC Genomics (2011)
-
MYC in mammalian epidermis: how can an oncogene stimulate differentiation?
Nature Reviews Cancer (2008)
-
Modelling Myc inhibition as a cancer therapy
Nature (2008)