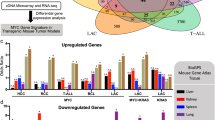
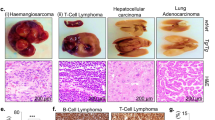
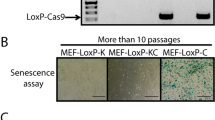
Abstract
The mammalian Runx gene family (Runx1–3) are transcription factors that play essential, lineage-specific roles in development. A growing body of evidence implicates these genes as mutational targets in cancer where, in different contexts, individual family members have been reported to act as tumour suppressors, dominant oncogenes or mediators of metastasis. We are exploring these paradoxical observations by ectopic expression of RUNX genes in primary murine embryonic fibroblasts where, in common with a number of other dominant oncogenes, RUNX1 induces senescence-like growth arrest in the presence of an intact p19ARF-p53 pathway. We now report that, in MEFs lacking functional p53, RUNX1 has apparently prooncogenic effects on cell growth that include cytoskeletal reorganization, reduced contact inhibition at confluence and accelerated tumour expansion in vivo. On the other hand, RUNX1 conferred no obvious growth advantage at low cell density and actually delayed entry of primary MEFs into S phase. We also found that ectopic RUNX1 interferes with the morphological and growth responses of p53-null MEFs to TGFβ indicating that these effects are mediated by overlapping pathways. These observations help to elucidate the context-dependent consequences of loss and gain of Runx activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bae SC, Lee KS, Zhang YW and Ito Y . (2001). Intimate relationship between TGF-beta/BMP signaling and runt domain transcription factor, PEBP2/CBF. J. Bone Joint Surg. Am., 83-A (Suppl. 1), S48–S55.
Baxter EW, Blyth K, Cameron ER and Neil JC . (2001). Selection for loss of p53 function in T-cell lymphomagenesis is alleviated by Moloney murine leukemia virus infection in myc transgenic mice. J. Virol., 75, 9790–9798.
Bernardin F and Friedman AD . (2002). AML1 stimulates G1 to S progression via its transactivation domain. Oncogene, 21, 3247–3252.
Blyth K, Stewart M, Bell M, James C, Evan G, Neil JC and Cameron ER . (2000). Sensitivity to myc-induced apoptosis is retained in spontaneous and transplanted lymphomas of CD2-mycER mice. Oncogene, 19, 773–782.
Blyth K, Terry A, Mackay N, Vaillant F, Bell M, Cameron ER, Neil JC and Stewart M . (2001). Runx2: a novel oncogenic effector revealed by in vivo complementation and retroviral tagging. Oncogene, 20, 295–302.
Brubaker KD, Vessella RL, Brown LG and Corey E . (2003). Prostate cancer expression of runt-domain transcription factor Runx2, a key regulator of osteoblast differentiation and function. Prostate, 56, 13–22.
Cameron ER, Blyth K, Hanlon L, Kilbey A, Mackay N, Stewart M, Terry A, Vaillant F, Wotton S and Neil JC . (2003). The Runx genes as dominant oncogenes. Blood Cells Mol. Dis., 30, 194–200.
Chow M and Rubin H . (2000a). Clonal selection versus genetic instability as the driving force in neoplastic transformation. Cancer Res., 60, 6510–6518.
Chow M and Rubin H . (2000b). Coculturing diverse clonal populations prevents the early-stage neoplastic progression that occurs in the separate clones. Proc. Natl. Acad. Sci. USA, 97, 174–178.
Cordenonsi M, Dupont S, Maretto S, Insinga A, Imbriano C and Piccolo S . (2003). Links between tumor suppressors: p53 is required for TGF-beta gene responses by cooperating with Smads. Cell, 113, 301–314.
Dal Cin P, Atkins L, Ford C, Ariyanayagam S, Armstrong SA, George R, Cleary A and Morton CC . (2001). Amplification of AML1 in childhood acute lymphoblastic leukemias. Genes Chromosomes Cancer, 30, 407–409.
Derynck R, Akhurst RJ and Balmain A . (2001). TGF-beta signaling in tumor suppression and cancer progression. Nat. Genet., 29, 117–129.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery Jr CA, Butel JS and Bradley A . (1992). Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature, 356, 215–221.
Frank R, Zhang J, Uchida H, Meyers S, Hiebert SW and Nimer SD . (1995). The AML1/ETO fusion protein blocks transactivation of the GM-CSF promoter by AML1B. Oncogene, 11, 2667–2674.
Frank RC, Sun X, Berguido FJ, Jakubowiak A and Nimer SD . (1999). The t(8;21) fusion protein, AML1/ETO, transforms NIH3T3 cells and activates AP-1. Oncogene, 18, 1701–1710.
Hanai J, Chen LF, Kanno T, Ohtani-Fujita N, Kim WY, Guo WH, Imamura T, Ishidou Y, Fukuchi M, Shi MJ, Stavnezer J, Kawabata M, Miyazono K and Ito Y . (1999). Interaction and functional cooperation of PEBP2/CBF with Smads. Synergistic induction of the immunoglobulin germline Calpha promoter. J. Biol. Chem., 274, 31577–31582.
Harewood L, Robinson H, Harris R, Al Obaidi MJ, Jalali GR, Martineau M, Moorman AV, Sumption N, Richards S, Mitchell C and Harrison CJ . (2003). Amplification of AML1 on a duplicated chromosome 21 in acute lymphoblastic leukemia: a study of 20 cases. Leukemia, 17, 547–553.
Ito Y and Miyazono K . (2003). RUNX transcription factors as key targets of TGF-beta superfamily signaling. Curr. Opin. Genet. Dev., 13, 43–47.
Jakubowiak A, Pouponnot C, Berguido F, Frank R, Mao S, Massague J and Nimer SD . (2000). Inhibition of the transforming growth factor beta 1 signaling pathway by the AML1/ETO leukemia-associated fusion protein. J. Biol. Chem., 275, 40282–40287.
Ji C, Casinghino S, Chang DJ, Chen Y, Javed A, Ito Y, Hiebert SW, Lian JB, Stein GS, McCarthy TL and Centrella M . (1998). CBFa(AML/PEBP2)-related elements in the TGF-beta type I receptor promoter and expression with osteoblast differentiation. J. Cell. Biochem., 69, 353–363.
Kurokawa M, Tanaka T, Tanaka K, Ogawa S, Mitani K, Yazaki Y and Hirai H . (1996). Overexpression of the AML1 proto-oncoprotein in NIH3T3 cells leads to neoplastic transformation depending on the DNA-binding and transactivational potencies. Oncogene, 12, 883–892.
Lee KS, Kim HJ, Li QL, Chi XZ, Ueta C, Komori T, Wozney JM, Kim EG, Choi JY, Ryoo HM and Bae SC . (2000). Runx2 is a common target of transforming growth factor beta1 and bone morphogenetic protein 2, and cooperation between Runx2 and Smad5 induces osteoblast-specific gene expression in the pluripotent mesenchymal precursor cell line C2C12. Mol. Cell. Biol., 20, 8783–8792.
Li J, Shen H, Himmel KL, Dupuy AJ, Largaespada DA, Nakamura T, Shaughnessy Jr JD, Jenkins NA and Copeland NG . (1999). Leukaemia disease genes: large-scale cloning and pathway predictions. Nat. Genet., 23, 348–353.
Li QL, Ito K, Sakakura C, Fukamachi H, Inoue K, Chi XZ, Lee KY, Nomura S, Lee CW, Han SB, Kim HM, Kim WJ, Yamamoto H, Yamashita N, Yano T, Ikeda T, Itohara S, Inazawa J, Abe T, Hagiwara A, Yamagishi H, Ooe A, Kaneda A, Sugimura T, Ushijima T, Bae SC and Ito Y . (2002). Causal relationship between the loss of RUNX3 expression and gastric cancer. Cell, 109, 113–124.
Linggi B, Muller-Tidow C, van de LL, Hu M, Nip J, Serve H, Berdel WE, van der RB, Quelle DE, Rowley JD, Cleveland J, Jansen JH, Pandolfi PP and Hiebert SW . (2002). The t(8;21) fusion protein, AML1 ETO, specifically represses the transcription of the p14(ARF) tumor suppressor in acute myeloid leukemia. Nat. Med., 8, 743–750.
Littlewood TD, Hancock DC, Danielian PS, Parker MG and Evan GI . (1995). A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res., 23, 1686–1690.
Lou J, Cao W, Bernardin F, Ayyanathan K, Rauscher III FJ and Friedman AD . (2000). Exogenous cdk4 overcomes reduced cdk4 RNA and inhibition of G1 progression in hematopoietic cells expressing a dominant-negative CBF – a model for overcoming inhibition of proliferation by CBF oncoproteins. Oncogene, 19, 2695–2703.
Margolin JF, Friedman JR, Meyer WK, Vissing H, Thiesen HJ, Rauscher FJ and III . (1994). Kruppel-associated boxes are potent transcriptional repression domains. Proc. Natl. Acad. Sci. USA, 91, 4509–4513.
Meyers S, Lenny N and Hiebert SW . (1995). The t(8;21) fusion protein interferes with AML-1B-dependent transcriptional activation. Mol. Cell. Biol., 15, 1974–1982.
Mikhail FM, Serry KA, Hatem N, Mourad ZI, Farawela HM, El Kaffash DM, Coignet L and Nucifora G . (2002). AML1 gene over-expression in childhood acute lymphoblastic leukemia. Leukemia, 16, 658–668.
Morgenstern JP and Land H . (1990). Advanced mammalian gene transfer: high titre retroviral vectors with multiple drug selection markers and a complementary helper-free packaging cell line. Nucleic Acids Res., 18, 3587–3596.
Moustakas A and Stournaras C . (1999). Regulation of actin organisation by TGF-beta in H-ras-transformed fibroblasts. J. Cell Sci., 112 (Part 8), 1169–1179.
Nelson PJ and Daniel TO . (2002). Emerging targets: Molecular mechanisms of cell contact-mediated growth control. Kidney Int. Suppl., 61 (Suppl 1), 99–105.
Niini T, Kanerva J, Vettenranta K, Saarinen-Pihkala UM and Knuutila S . (2000). AML1 gene amplification: a novel finding in childhood acute lymphoblastic leukemia. Haematologica, 85, 362–366.
Okuda T, Cai Z, Yang S, Lenny N, Lyu CJ, van Deursen JM, Harada H and Downing JR . (1998). Expression of a knocked-in AML1-ETO leukemia gene inhibits the establishment of normal definitive hematopoiesis and directly generates dysplastic hematopoietic progenitors. Blood, 91, 3134–3143.
Preudhomme C, Warot-Loze D, Roumier C, Grardel-Duflos N, Garand R, Lai JL, Dastugue N, Macintyre E, Denis C, Bauters F, Kerckaert JP, Cosson A and Fenaux P . (2000). High incidence of biallelic point mutations in the Runt domain of the AML1/PEBP2 alpha B gene in Mo acute myeloid leukemia and in myeloid malignancies with acquired trisomy 21. Blood, 96, 2862–2869.
Roberts AB and Wakefield LM . (2003). The two faces of transforming growth factor beta in carcinogenesis. Proc. Natl. Acad. Sci. USA, 100, 8621–8623.
Sakamoto Y, Takeda Y, Takagi H, Tsuchiya T, Shoji A, Miyazaki K and Umeda M . (1999). Inhibition of focus formation of transformed cloned cells by contact with non-transformed BALB/c 3T3 A31-1-1 cells. Cancer Lett., 136, 159–165.
Serrano M, Lin AW, McCurrach ME, Beach D and Lowe SW . (1997). Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell, 88, 593–602.
Siegel PM and Massague J . (2003). Cytostatic and apoptotic actions of TGF-beta in homeostasis and cancer. Nat. Rev. Cancer, 3, 807–820.
Song WJ, Sullivan MG, Legare RD, Hutchings S, Tan X, Kufrin D, Ratajczak J, Resende IC, Haworth C, Hock R, Loh M, Felix C, Roy DC, Busque L, Kurnit D, Willman C, Gewirtz AM, Speck NA, Bushweller JH, Li FP, Gardiner K, Poncz M, Maris JM and Gilliland DG . (1999). Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia (see comments). Nat. Genet., 23, 166–175.
Speck NA and Gilliland DG . (2002). Core-binding factors in haematopoiesis and leukaemia. Nat. Rev. Cancer, 2, 502–513.
Stewart M, Mackay N, Cameron ER and Neil JC . (2002). The common retroviral insertion locus Dsi1 maps 30 kilobases upstream of the P1 promoter of the murine Runx3/Cbfa3/Aml2 gene. J. Virol., 76, 4364–4369.
Stewart M, Terry A, Hu M, O'Hara M, Blyth K, Baxter E, Cameron E, Onions DE and Neil JC . (1997). Proviral insertions induce the expression of bone-specific isoforms of PEBP2alphaA (CBFA1): evidence for a new myc collaborating oncogene. Proc. Natl. Acad. Sci. USA, 94, 8646–8651.
Strom DK, Nip J, Westendorf JJ, Linggi B, Lutterbach B, Downing JR, Lenny N and Hiebert SW . (2000). Expression of the AML-1 oncogene shortens the G(1) phase of the cell cycle. J. Biol. Chem., 275, 3438–3445.
Sun L, Vitolo M and Passaniti A . (2001). Runt-related gene 2 in endothelial cells: inducible expression and specific regulation of cell migration and invasion. Cancer Res., 61, 4994–5001.
Vaillant F, Blyth K, Andrew L, Neil JC and Cameron ER . (2002). Enforced expression of Runx2 perturbs T cell development at a stage coincident with beta-selection. J. Immunol., 169, 2866–2874.
Vaillant F, Blyth K, Terry A, Bell M, Cameron ER, Neil J and Stewart M . (1999). A full-length Cbfa1 gene product perturbs T-cell development and promotes lymphomagenesis in synergy with myc. Oncogene, 18, 7124–7134.
Wotton S, Stewart M, Blyth K, Vaillant F, Kilbey A, Neil JC and Cameron ER . (2002). Proviral insertion indicates a dominant oncogenic role for Runx1/AML-1 in T-cell lymphoma. Cancer Res., 62, 7181–7185.
Yergeau DA, Hetherington CJ, Wang Q, Zhang P, Sharpe AH, Binder M, Marin-Padilla M, Tenen DG, Speck NA and Zhang DE . (1997). Embryonic lethality and impairment of haematopoiesis in mice heterozygous for an AML1-ETO fusion gene. Nat. Genet., 15, 303–306.
Yeung F, Law WK, Yeh CH, Westendorf JJ, Zhang Y, Wang R, Kao C and Chung LW . (2002). Regulation of human osteocalcin promoter in hormone-independent human prostate cancer cells. J. Biol. Chem., 277, 2468–2476.
Zhang YW, Yasui N, Ito K, Huang G, Fujii M, Hanai J, Nogami H, Ochi T, Miyazono K and Ito Y . (2000). A RUNX2/PEBP2alpha A/CBFA1 mutation displaying impaired transactivation and Smad interaction in cleidocranial dysplasia. Proc. Natl. Acad. Sci. USA, 97, 10549–10554.
Acknowledgements
We thank Dr B Linggi and Dr S Hiebert for providing pBabe-AML plasmid and helpful discussions. This work was supported by Cancer Research UK and the Leukaemia Research Fund of Great Britain. ADF is a Leukemia and Lymphoma Society Scholar.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wotton, S., Blyth, K., Kilbey, A. et al. RUNX1 transformation of primary embryonic fibroblasts is revealed in the absence of p53. Oncogene 23, 5476–5486 (2004). https://doi.org/10.1038/sj.onc.1207729
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207729
Keywords
This article is cited by
-
The landscape of human tissue and cell type specific expression and co-regulation of senescence genes
Molecular Neurodegeneration (2022)
-
Multifaceted C-terminus of HSP70-interacting protein regulates tumorigenesis via protein quality control
Archives of Pharmacal Research (2019)
-
The RUNX family: developmental regulators in cancer
Nature Reviews Cancer (2015)
-
RUNX1 and its fusion oncoprotein derivative, RUNX1-ETO, induce senescence-like growth arrest independently of replicative stress
Oncogene (2009)
-
Gene array analysis reveals a common Runx transcriptional programme controlling cell adhesion and survival
Oncogene (2008)