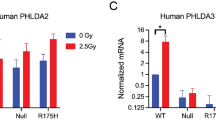
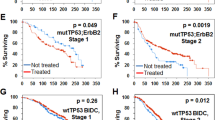
Abstract
Individuals affected with the Gorlin syndrome inherit a germ-line mutation of the patched (Ptc1) developmental gene and, analogously to Ptc1 heterozygous mice, show an increased susceptibility to spontaneous tumor development. Human and mouse Ptc1 heterozygotes (Ptc1+/−) are also hypersensitive to ionizing radiation (IR)-induced tumorigenesis in terms of basal cell carcinoma (BCC) induction. We have analysed the involvement of Ptc1 in the tumorigenic response to a single dose of 3 Gy X-rays in neonatal and adult Ptc1 heterozygous and wild type mice. We report that irradiation dramatically increased the incidence of medulloblastoma development (51%) over the spontaneous rate (7%) in neonatal but not adult Ptc1 heterozygotes, indicating that medulloblastoma induction by IR is subjected to temporal restriction. Analysis of Ptc1 allele status in the tumors revealed loss of the wild type allele in 17 of 18 medulloblastomas from irradiated mice and in two of three spontaneous medulloblastomas. To our knowledge, irradiated newborn Ptc1+/− heterozygous mice constitute the first mouse model of IR-induced medulloblastoma tumorigenesis, providing a useful tool to elucidate the molecular basis of medulloblastoma development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aszterbaum M, Epstein J, Oro A, Douglas V, LeBott PE, Scott MP, Epstein Jr EH . 1999 Nat. Med. 11: 1285–1291
Barnes EA, Kong M, Oleendorff V, Donoghue DJ . 2001 EMBO J. 9: 2214–2223
Chidambaram A, Goldestein AM, Galiani MR, Gerrard B, Bale SJ, Digiovanna JJ, Bale AE, Dean M . 1996 Cancer Res. 56: 4599–4601
Corey R, Jenkins RB, Frederick L, Hebrink D, Alderete B, Fults DW, James CD . 1997 Cancer Res. 57: 842–845
Cowan R, Hoban P, Kelsey A, Birch JM, Gattamaneni R, Evans DG . 1997 Br. J. Cancer 76: 141–145
Goldowitz D, Hamre K . 1998 Trends Neurosci. 21: 375–382
Goodrich LV, Milenkovic L, Higgins KM, Scott MP . 1997 Science 277: 1109–1113
Gorlin RJ . 1987 Medicine (Baltimore) 66: 98–113
Hahn H, Wicking C, Zaphiropoulous PG, Galiani MR, Shanley S, Chidambaram A, Vorechovsky I, Holmberg E, Unden AB, Gillies S, Negus K, Smyth I, Pressman C, Leffell DJ, Gerrard B, Goldestein AM, Dean M, Tofgard R, Chenevix-Trench G, Wainwright B, Bale AE . 1996 Cell 85: 841–851
Hahn H, Wojnowski L, Zimmer AM, Hall J, Miller G, Zimmer A . 1998 Nat. Med. 4: 619–622
Innocente SA, Abrahamson JLA, Cogswell JP, Lee JM . 1999 Proc. Natl. Acad. Sci. USA 96: 2147–2152
Lacombe D, Chateil JF, Fontan D, Battin J . 1990 Genet. Couns. 1: 273–277
Martì E, Bovolenta P . 2002 Trends Neurosci. 25: 89–94
Miller DL, Weinstock MA . 1994 J. Am. Acad. Dermatol. 30: 774–778
Novakovic B . 1994 Mol. Pediatr. Oncol. 23: 480
Pappo AS, Shapiro DN, Crist WM . 1997 Pediatr. Clin. North. Am. 44: 953–972
Rowitch DH, St-Jacques B, Lee SMK, Flax JD, Snyder EY, McMahon AP . 1999 J. Neurosci. 19: 8954–8965
Unden AB, Holmberg E, Lundhrozell B, Stahlebackdahl M, Zaphiropoulos PG, Tofgard R, Vorechovsky I . 1996 Cancer Res. 56: 4562–4565
Wetmore C, Eberhart DE, Curran T . 2000 Cancer Res. 60: 2239–2246
Wetmore C, Eberhart DE, Curran T . 2001 Cancer Res. 61: 513–516
Wicking C, Shanley S, Smyth I, Gillies S, Negus K, Graham S, Suthers G, Haites N, Edwards M, Wainwright B, Chenevix-Trench G . 1997 Am. J. Hum. Genet. 60: 21–26
Xie J, Johnson RL, Zhang X, Bare JW, Waldman FM, Cogen PH, Menon AG, Warren RS, Chen LC, Scott MP, Epstein Jr EH . 1997 Cancer Res. 57: 2369–2372
Zurawel RH, Chiappa SA, Allen C, Raffel C . 1997 Cancer Res. 57: 842–845
Zurawel RH, Allen C, Wechsler-Reya R, Scott MP, Raffel C . 2000 Genes Chromosomes Cancer 28: 77–81
Acknowledgements
This work was partially supported by the Commission of the European Communities under Contract FIGH-CT-1999-00006 and by Progetto Strategico 1998 (L. 449/97-MURST). The authors wish to thank Mrs M Manzotti for assistance in handling and treating the mice. Dr M Tanori was supported by a fellowship from Istituto Nazionale Tumori.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pazzaglia, S., Mancuso, M., Atkinson, M. et al. High incidence of medulloblastoma following X-ray-irradiation of newborn Ptc1 heterozygous mice. Oncogene 21, 7580–7584 (2002). https://doi.org/10.1038/sj.onc.1205973
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205973
Keywords
This article is cited by
-
Out-of-field effects: lessons learned from partial body exposure
Radiation and Environmental Biophysics (2022)
-
Nanog-driven cell-reprogramming and self-renewal maintenance in Ptch1 +/− granule cell precursors after radiation injury
Scientific Reports (2017)
-
A population of Nestin-expressing progenitors in the cerebellum exhibits increased tumorigenicity
Nature Neuroscience (2013)
-
What underlies the diversity of brain tumors?
Cancer and Metastasis Reviews (2013)
-
The Hedgehog's tale: developing strategies for targeting cancer
Nature Reviews Cancer (2011)