Abstract
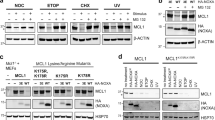
MDMX is a p53 binding protein, which shares a high degree of homology with MDM2, a negative regulator of the tumor suppressor p53. MDMX has been shown to counteract MDM2-dependent p53 degradation and to stabilize p53 in its inactive form. In this study: we identify two MDMX proteolytic pathways that control its intracellular levels, and show that MDMX post-translational processing may be regulated by p53. Mouse MDMX is cleaved in vitro and in vivo by caspase activity, between aminoacids 358 and 361, producing a p54 minor form. In addition, MDMX is subjected to proteasome-mediated degradation, which concurs to MDMX proteolysis mainly through degradation of p54. A D361A-MDMX mutant, resistant to caspase cleavage, exhibits prolonged intracellular lifetime in comparison to wild-type protein, indicating that caspase cleavage affects stability of MDMX protein probably by modulating its further degradation. Overexpression of exogenous p53 increases the intracellular levels of p54 product. Similarly, activation of endogenous p53 by adriamycin enhances MDMX cleavage and produces a marked decrease of its intracellular levels, while not affecting the D361A-MDMX mutant. In addition, the D361A-MDMX mutant lacks the ability to inhibit p53 transactivation in respect to wild-type MDMX, suggesting that MDMX caspase cleavage play an important functional role. In conclusion, our results demonstrate that, in analogy to MDM2, MDMX may be subjected to proteolytic modifications that regulate its intracellular levels. Moreover, decrease of MDMX protein levels following p53 activation suggests a p53-dependent regulatory feedback of MDMX function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Baek SH, Woo SK, Lee J, Yoo YJ, Cho CM, Kang MS, Tanaka K, Chung CH . 1997 Biochem. J. 325: 325–330
Barak Y, Juven T, Haffner R, Oren M . 1993 EMBO J. 2: 461–468
Bottger V, Bottger A, Garcia-Echeverria C, Ramos YFM, van der Eb AJ, Jochemsen AG, Lane DP . 1999 Oncogene 18: 189–199
Buschmann T, Fuchs SY, Lee CH, Pan ZQ, Ronai Z . 2000 Cell 101: 753–762
Chang YC, Lee YS, Tejima T, Tanaka K, Omura S, Heintz NH, Magae J . 1998 Cell Growth Differ. 9: 79–84
Chen J, Marechal V, Levine AJ . 1993 Mol. Cell Biol. 13: 4107–4114
Chen L, Marechal V, Moreau J, Levine AJ, Chen J . 1997 J. Biol. Chem. 272: 22966–22973
Fenteany G, Standaert RF, Lane WS, Choi S, Corey EJ, Schreiber SL . 1995 Science 268: 726–731
Freedman DA, Levine AJ . 1999 Cancer Res. 59: 1–7
Gottlieb E, Oren M . 1998 EMBO J. 17: 3587–3596
Graham F, Prevec L . 1991 Manipulation of adenovirus In Methods of Molecular Biology Murray EJ (ed) Humana Press: Clifton, NJ pp 3587–3596
Haupt Y, Maya A, Kazaz A, Oren M . 1997 Nature 387: 296–299
Honda R, Tanaka H, Yasuda H . 1997 FEBS Lett. 420: 25–27
Jackson MW, Berberich SJ . 1999 DNA and Cell Biology 18: 693–700
Jackson MW, Berberich SJ . 2000 Mol. Cell Biol. 20: 1001–1007
Jackson MW, Lindstrom MS, Berberich SJ . 2001 J. Biol. Chem. 276: 25336–25341
Kamijo T, Weber JD, Zambetti G, Zindy F, Roussel MF, Sherr CJ . 1998 Proc. Natl. Acad. Sci. USA 95: 8292–8297
Kastan MB, Onyekwere O, Sindransky D, Vogelstein B, Craig RW . 1991 Cancer Res. 51: 6304–6311
Khosravi R, Maya R, Gottlieb T, Oren M, Shiloh Y, Shkedy D . 1999 Proc. Natl. Acad. Sci. USA 96: 14973–14977
Kozak M . 1996 Mammalian Genome 7: 563–574
Kubbutat MHG, Jones KH, Vousden KH . 1997 Nature 387: 299–303
Mayo DL, Turchi JJ, Berberich SJ . 1997 Cancer Res. 57: 5013–5016
Mendrysa SM, Perry ME . 2000 Mol. Cell Biol. 20: 2023–2030
Momand J, Zambetti GP, Olson DC, George D, Levine AJ . 1992 Cell 69: 1237–1245
Moretti F, Farsetti A, Soddu S, Misiti S, Crescenzi M, Filetti S, Andreoli M, Sacchi A, Pontecorvi A . 1997 Oncogene 14: 729–740
Oliner JD, Pietenpol JA, Thiagalingam S, Gyuris J, Kinzler KW, Vogelstein B . 1993 Nature 362: 857–860
Olson DC, Marechal V, Momand J, Chen J, Romocki C, Levine AJ . 1993 Oncogene 8: 2353–2360
Passalaris TM, Benanti JA, Gewin L, Kiyono T, Galloway DA . 1999 Mol. Cell Biol. 19: 5872–5881
Perry ME, Mendrysa SM, Saucedo LJ, Tannous P, Holubar M . 2000 J. Biol. Chem. 275: 5733–5738
Pochampally R, Fodera B, Chen L, Lu W, Chen J . 1999 J. Biol. Chem. 274: 15271–15277
Pochampally R, Fodera B, Chen L, Shao W, Levine E, Chen J . 1998 Oncogene 17: 2629–2636
Pomerantz J, Schreiber-Agus N, Liegeois NJ, Silverman A, Alland L, Chin L, Potes J, Chen K, Orlow I, Lee H-W . 1998 Cell 92: 713–723
Rallapalli R, Strachan G, Cho B, Mercer E, Hall DJ . 1999 J. Biol. Chem. 274: 8299–8308
Ramos YFM, Stad R, Attema J, Peltenburg LTC, van der Eb A, Jochemsen AG . 2001 Cancer Res. 61: 1839–1842
Ratovitski EA, Patturajan M, Hibi K, Trink B, Yamaguchi K, Sidransky D . 2001 Proc. Natl. Acad. Sci. U.S.A. 98: 1817–1822
Rock KL, Gramm C, Rothestein L, Clark K, Stein R, Dick L, Hwang D, Goldberg AL . 1994 Cell 78: 761–771
Saucedo LJ, Myers CD, Perry ME . 1999 J. Biol. Chem. 274: 8161–8168
Sharp DA, Kratowicz SA, Sank MJ, George DL . 1999 J. Biol. Chem. 274: 38189–38196
Shieh SY, Ikeda M, Taya Y, Prives C . 1997 Cell 91: 325–334
Sigalas I, Calvert AH, Anderson JJ, Neal DE, Lunec J . 1996 Nature Medicine 2: 912–917
Siu WY, Arooz T, Poon RY . 1999 Exp. Cell Res. 250: 131–141
Siliciano JD, Canman CE, Taya Y, Sakaguchi K, Appella E, Kastan MB . 1997 Genes Dev. 11: 3471–3481
Shvarts A, Steegenga WT, Riteco N, van Laar T, Dekker P, Bazuine M, van Ham RCA, van der Houven van Oordt W, Hateboer G, van der Eb AJ, Jochemsen AG . 1996 EMBO J. 15: 5349–5357
Shvarts A, Bazuine M, Dekker P, Ramos YFM, Steegenga WT, Merckx G, van Ham RCA, van der Houven van Oordt W, Hateboer G, van der Eb AJ, Jochemsen AG . 1997 Genomics 43: 34–42
Stad R, Ramos YFM, Little N, Grivell S, Attema J, van der Eb AJ, Jochemsen AG . 2000 J. Biol. Chem. 275: 28039–28044
Stott FJ, Bates S, James MC, McConnell BB, Starborg M, Brookes S, Palmero I, Ryan K, Hra E, Vousden KH, Peters G . 1998 EMBO J. 17: 5001–5014
Szak ST, Mays D, Pietenpol JA . 2001 Mol. Cel. Biol. 21: 3375–3386
Tanimura S, Ohtsuka S, Mitsui K, Shirouzu K, Yoshimura A, Ohtsubo M . 1999 FEBS Lett 447: 5–9
Utz PJ, Anderson P . 2000 Cell Death Differ. 7: 589–602
Wolf BB, Green DR . 1999 J. Biol. Chem. 274: 20049–20052
Zhang Y, Xiong Y, Yarbrough WG . 1998 Cell 92: 725–734
Acknowledgements
We thank M Oren for the gift of MDM2 plasmid and AJ Levine for anti-MDM2 antibodies. We are grateful to Silvia Soddu (Regina Elena Cancer Institute, Rome, Italy) for helpful discussion. This work was supported by research grants from Ministero dell'Università e della Ricerca Scientifica e Tecnologica (MURST), Ministero della Sanità, Progetto Italia-USA and Associazione Italiana per la Ricerca sul Cancro (AIRC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gentiletti, F., Mancini, F., D'Angelo, M. et al. MDMX stability is regulated by p53-induced caspase cleavage in NIH3T3 mouse fibroblasts. Oncogene 21, 867–877 (2002). https://doi.org/10.1038/sj.onc.1205137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205137
Keywords
This article is cited by
-
Interplay between MDM2, MDMX, Pirh2 and COP1: the negative regulators of p53
Molecular Biology Reports (2011)
-
Regulation of MDM4 (MDMX) function by p76MDM2: a new facet in the control of p53 activity
Oncogene (2010)
-
Full-length hdmX transcripts decrease following genotoxic stress
Oncogene (2008)
-
Multiple neurotoxic stresses converge on MDMX proteolysis to cause neuronal apoptosis
Cell Death & Differentiation (2007)
-
Cross-talk between calpain and caspase-3/-7 in cisplatin-induced apoptosis of melanoma cells: a major role of calpain inhibition in cell death protection and p53 status
Oncogene (2007)