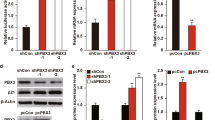
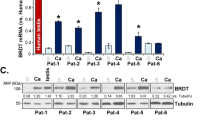
Abstract
The Brn-3b POU domain containing transcription factor is expressed in the developing sensory nervous system as well as in epithelial cells of the breast, cervix, and testes. Brn-3b functionally interacts with the estrogen receptor (ER) and in association with the ER, regulates transcription from estrogen responsive genes. In addition, Brn-3b expression is elevated in breast tumours compared to levels in normal mammary cells. To explore the role of Brn-3b in breast cancer, we established stable cell lines derived from the MCF7 human breast cancer cell line which had been transfected with Brn-3b sense or anti-sense constructs. The Brn-3b over-expressing cell lines exhibited increased growth rate, reached confluence at a higher saturation density, had higher proliferative activity, and an enhanced ability to form colonies in soft agar when compared to the control empty vector transfected cells. Likewise, the Brn-3b anti-sense cell lines showed reduced cellular growth and proliferation, reached confluence at a lower density, and exhibited a decreased ability to form colonies in soft agar when compared to the vector controls. Five to ten per cent of the Brn-3b over-expressing cells exhibited a severely altered morphology characterized by reduced adherence to tissue culture plastic, increased cell size, and a vacuolar cell shape. These results thus further indicate a role for the Brn-3b transcription factor in regulating mammary cell growth and suggest that its elevation in breast cancer is of functional significance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Awgulewitsch A, Jacobs D . 1992 Nature 358: 341–344
Bergson C, McGinnis W . 1990 EMBO J. 9: 4287–4297
Budhram-Mahadeo V, Morris PJ, Lakin ND, Theil T, Ching GY, Lillycrop KA, Moroy T, Liem RK, Latchman DS . 1995 J. Biol. Chem. 270: 2853–2858
Budhram-Mahadeo V, Ndisang D, Ward T, Weber BL, Latchman DS . 1999 Oncogene 18: 6684–6691
Budhram-Mahadeo V, Parker M, Latchman DS . 1998 Mol. Cell. Biol. 18: 1029–1041
Dickson RB, Lippman ME . 1987 Endocr. Rev. 8: 29–43
Dickson RB, Kasid A, Huff KK, Bates SE, Knabbe C, Bronzert D, Gelmann EP, Lippman ME . 1987 Proc. Natl. Acad. Sci. USA 84: 837–841
Elledge RM, Osborne CK . 1997 BMJ 314: 1843–1844
Endoh H, Maruyama K, Masuhiro Y, Kobayashi Y, Goto M, Tai H, Yanagisawa J, Metzger D, Hashimoto S, Kato S . 1999 Mol. Cell. Biol. 19: 5363–5372
Fuqua SA . 1992 J. Natl. Cancer Inst. 84: 554–555
Fuqua SA, Fitzgerald SD, Allred DC, Elledge RM, Nawaz Z, McDonnell DP, O'Malley BW, Greene GL, McGuire WL . 1992 Cancer Res. 52: 483–486
Gerrero MR, McEvilly RJ, Turner E, Lin CR, O'Connell S, Jenne KJ, Hobbs MV, Rosenfeld MG . 1993 Proc. Natl. Acad. Sci. USA 90: 10841–10845
He X, Treacy MN, Simmons DM, Ingraham HA, Swanson LW, Rosenfeld MG . 1989 Nature 340: 35–41
Kato S, Endoh H, Masuhiro Y, Kitamoto T, Uchiyama S, Sasaki H, Masushige S, Gotoh Y, Nishida E, Kawashima H et al . 1995 Science 270: 1491–1494
Latchman DS . 1999 J. Cell. Physiol. 179: 126–133
Lillycrop KA, Budrahan VS, Lakin ND, Terrenghi G, Wood JN, Polak JM, Latchman DS . 1992 Nucleic Acids Res. 20: 5093–5096
Lippman ME, Bates S, Huff KK, Davidson N, Dickson RB . 1987 J. Lab. Clin. Med. 109: 230–235
Liu YZ, Dawson SJ, Latchman DS . 1996 J. Mol. Neurosci. 7: 77–85
Lou L, Bergson C, McGinnis W . 1995 Nucleic. Acids. Res. 23: 3481–3487
Malicki J, Cianetti LC, Peschle C, McGinnis W . 1992 Nature 358: 345–347
Morris PJ, Theil T, Ring CJ, Lillycrop KA, Moroy T, Latchman DS . 1994 Mol. Cell. Biol. 14: 6907–6914
Murphy LC, Sutherland RL . 1983 J. Clin. Endocrinol. Metab. 57: 373–379
Ndisang D, Budhram-Mahadeo V, Latchman DS . 1999 J. Biol. Chem. 274: 28521–28527
Packer AI, Crotty DA, Elwell VA, Wolgemuth DJ . 1998 Development 125: 1991–1998
Popperl H, Featherstone MS . 1992 EMBO J. 11: 3673–3680
Reddel RR, Sutherland RL . 1983 Eur. J. Cancer Clin. Oncol. 19: 1179–1181
Reddel RR, Murphy LC, Sutherland RL . 1983 Cancer Res. 43: 4618–4624
Smith MD, Latchman DS . 1996 Int. J. Cancer 67: 653–660
Smith MD, Dawson SJ, Latchman DS . 1997 J. Biol. Chem. 272: 1382–1388
Sutherland RL, Hall RE, Taylor IW . 1983a Cancer Res. 43: 3998–4006
Sutherland RL, Green MD, Hall RE, Reddel RR, Taylor IW . 1983b Eur. J. Cancer Clin. Oncol. 19: 615–621
Taylor IW, Hodson PJ, Green MD, Sutherland RL . 1983 Cancer Res. 43: 4007–4010
Theil T, McLean Hunter S, Zornig M, Moroy T . 1993 Nucleic Acids Res. 21: 5921–5929
Turner EE, Jenne KJ, Rosenfeld MG . 1994 Neuron 12: 205–218
Acknowledgements
We thank Tarik Moroy for the gift of pLTR and Brn-3b recombinant clones, Martin Smith for the gift of pJ4 Brn-3b anti-sense constructs, and Benjamin Chain and Walter Low for assistance with the proliferation studies. This work was supported by the USA Department of Defence, Breast Cancer Research Program.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dennis, J., Budhram-Mahadeo, V. & Latchman, D. The Brn-3b POU family transcription factor regulates the cellular growth, proliferation, and anchorage dependence of MCF7 human breast cancer cells. Oncogene 20, 4961–4971 (2001). https://doi.org/10.1038/sj.onc.1204491
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204491
Keywords
This article is cited by
-
Vascular dysfunction caused by loss of Brn-3b/POU4F2 transcription factor in aortic vascular smooth muscle cells is linked to deregulation of calcium signalling pathways
Cell Death & Disease (2023)
-
Linking metabolic dysfunction with cardiovascular diseases: Brn-3b/POU4F2 transcription factor in cardiometabolic tissues in health and disease
Cell Death & Disease (2021)
-
The POU4F2/Brn-3b transcription factor is required for the hypertrophic response to angiotensin II in the heart
Cell Death & Disease (2019)
-
Co-expression of POU4F2/Brn-3b with p53 may be important for controlling expression of pro-apoptotic genes in cardiomyocytes following ischaemic/hypoxic insults
Cell Death & Disease (2014)
-
Proliferation-associated POU4F2/Brn-3b transcription factor expression is regulated by oestrogen through ERα and growth factors via MAPK pathway
Breast Cancer Research (2011)