Abstract
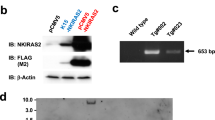
To clarify the role of the H-Ras in vivo, we generated H-ras null mutant mice by gene targeting. In spite of the importance of the Ras in cell proliferation and differentiation, H-ras null mutant mice grew normally and were fertile. The oldest H-ras mutant mice grew to be more than 30 months old. We used the H-ras deficient mice to study the importance of the H-ras and other ras genes in the development of skin tumors induced by initiation with 7,12-dimethylbenz(a)anthracene (DMBA) followed by promotion with 12-O-tetradecanoylphorbol-13-acetate (TPA). We showed that H-ras null mutant mice develop approximately six times less papillomas compared with wild-type littermates after 20 weeks of TPA treatment. While all papillomas examined (17 out of 17) in wild-type mice have mutations of H-ras at codon 61, 13 (62%) out of 21 papillomas in H-ras null mutant mice have mutations of K-ras gene at codon 12, 13, or 61 and another eight (38%) papillomas have no mutations in these codons of K-ras or N-ras genes. This suggests that the activation of H-ras gene is critical in the wild-type mice, but the activation of K-ras gene can replace the H-ras activation in the initiation step of skin tumor development in the H-ras deficient mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Balmain A, Ramsden M, Bowden GT and Smith J . 1984 Nature 307: 658–660
Balmain A and Brown K . 1988 Adv Cancer Res 51: 147–182
Barbacid M . 1987 Annu Rev Biochem 56: 779–827
Bizub D, Wood AW and Skalka AM . 1986 Proc Natl Acad Sci USA 83: 6048–6052
Boutwell RK . 1974 CRC Crit Rev Toxicol 2: 419–443
Bradley A . 1987 Teratocarcinomas and Embryonic Stem Cells A Practical Approach Robertson EJ ed IRL Press London pp 113–151
Brookes P, Cooper CS, Ellis MV, Warren W, Gardner E and Summerhayes IC . 1988 Mol Carcinog 1: 82–88
Brown K, Buchamann A and Balmain A . 1990 Proc Natl Acad Sci USA 87: 538–542
Guerrero I and Pellicer A . 1987 Mutat Res 185: 293–308
Harvey JJ . 1964 Nature 204: 1104–1105
Hecker E, Fusenig NE, Kunz W, Marks F and Thielmann HW eds . 1982 Carcinogenesis A Comprehensive Survey Raven Press New York
Kemp CJ, Donehower LA, Bradley A and Balmain A . 1993 Cell 74: 813–822
Kirsten WH and Mayer LA . 1967 J Natl Cancer Inst 39: 311–335
Koera K, Nakamura K, Nakao K, Miyoshi J, Toyoshima K, Hatta T, Otani H, Aiba A and Katsuki M . 1997 Oncogene 15: 1151–1159
Kuehn MR, Bradley A, Robertson EJ and Evans MJ . 1987 Nature 326: 295–298
Lowy DR and Willumsen BM . 1993 Annu Rev Biochem 62: 851–891
Manam S, Storer RD, Prahalada S, Leander KR, Kraynak AR, Ledwith BJ, van Zwieten MJ, Bradley MO and Nichols WW . 1992 Cancer Res 52: 3347–3352
Moore RJ, Owens DM, Stamp G, Arnott C, Burke F, East N, Holdsworth H, Turner L, Rollins B, Pasparakis M, Kollias G and Balkwill F . 1999 Nat Med 5: 828–831
Quintanilla M, Brown K, Ramsden M and Balmain A . 1986 Nature 322: 78–80
Saez E, Rutberg SE, Mueller E, Oppenheim H, Smoluk J, Yuspa SH and Spiegelman BM . 1995 Cell 82: 721–732
Sekiya T, Fushimi M, Hori H, Hirohashi S, Nishimura S and Sugimura T . 1984 Proc Natl Acad Sci USA 81: 4771–4775
Shimizu K, Goldfarb M, Suard Y, Perucho M, Li Y, Kamata T, Feramisco J, Stanvnezer E, Fogh J and Wigler MH . 1983 Proc Natl Acad Sci USA 80: 2112–2116
Yagi T, Ikawa Y, Yoshida K, Shigetani Y, Takeda N, Mabuchi I, Yamamoto T and Aizawa S . 1990 Proc Natl Acad Sci USA 87: 9918–9922
Yuspa SH and Poirier MC . 1988 Adv Cancer Res 50: 25–70
Yuspa SH . 1994 Cancer Res 54: 1178–1189
Zarbl H, Sukumar S, Arthur AV, Martin-Zanca D and Barbacid M . 1985 Nature 315: 382–385
Acknowledgements
This work was supported in part by Grants-in-Aid for Scientific Research on Priority Areas, for Cancer Research and for Scientific Research from the Ministry of Education, Science, Sports and Culture, Japan, Grants-in-Aid from the Ministry of Health and Welfare, Japan. We also thank K Katsuki and Y Ikeda for their excellent technical assistance and K Tsurui and T Kohyama for their help in maintaining the animals.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ise, K., Nakamura, K., Nakao, K. et al. Targeted deletion of the H-ras gene decreases tumor formation in mouse skin carcinogenesis. Oncogene 19, 2951–2956 (2000). https://doi.org/10.1038/sj.onc.1203600
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1203600
Keywords
This article is cited by
-
Synthetic lethal kinases in Ras/p53 mutant squamous cell carcinoma
Oncogene (2022)
-
Ras isoform-specific expression, chromatin accessibility, and signaling
Biophysical Reviews (2021)
-
Considerations for skin carcinogenesis experiments using inducible transgenic mouse models
BMC Research Notes (2018)
-
The polarity protein Scrib mediates epidermal development and exerts a tumor suppressive function during skin carcinogenesis
Molecular Cancer (2015)
-
Matriptase promotes inflammatory cell accumulation and progression of established epidermal tumors
Oncogene (2015)