Abstract
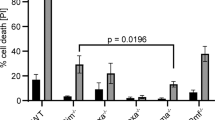
The Bcl6 gene is located at chromosomal band 3q27, a breakpoint for translocation that frequently occurs in B cell lymphomas. Bcl6 has been found to be preferentially expressed in germinal center B cells, and expression of this gene has been shown to be essential for germinal center formation in vivo. The physiological function of Bcl6 and its role in lymphomagenesis, however, are not yet known. Since significant expression of Bcl6 has been demonstrated in skeletal muscle, we have utilized a differentiation-inducible mouse myogenic cell line, C2C12, to elucidate the function of the Bcl6 gene product. Expression of Bcl6 mRNA was very low in growing myocytes, but was increased in differentiating myocytes cultured in serum-starved medium. Incubation of these cells with cytokines or chemicals that are known to block differentiation suppressed this increased Bcl6 message abundance, indicating that Bcl6 induction is related to the process of terminal differentiation in muscle cells. While a fraction of myocytes is known to undergo apoptosis after serum-starvation to induce differentiation, adenovirus-mediated overexpression of Bcl6 enhanced the viability of the differentiating cells by preventing the apoptosis. High levels of Bcl6 antisense mRNA expression induced substantial apoptosis during the differentiation of C2C12 cells, but this was effectively prevented by infection with adenovirus that expressed Bcl6 sense mRNA. These results indicate that Bcl6 acts to prevent apoptotic cell death in differentiating myocytes. The deregulation of expression of this anti-apoptotic gene may also contribute to the development of B cell lymphomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Allman D, Jain A, Dent A, Maile RR, Selvaggi T, Kehry MR and Staudt LM. . 1996 Blood 87: 5257–5268.
Baron BW, Nucifora G, McCabe N, Espinosa R 3rd, Le Beau MM and McKeithan TW. . 1993 Proc. Natl. Acad. Sci. USA 90: 5262–5266.
Bastard C, Tilly H, Lenormand B, Bigorgne C, Boulet D, Kunlin A, Monconduit M and Piguet H. . 1992 Blood 79: 2527–2531.
Benezra R, Davis RL, Lockshon D, Turner DL and Weintraub H. . 1990 Cell 61: 49–59.
Bernardin F, Collyn DM, Quief S, Bastard C, Leprince D and Kerckaert JP. . 1997 Oncogene 14: 849–855.
Braun T, Buschhausen DG, Bober E, Tannich E and Arnold HH. . 1989 EMBO J. 8: 701–709.
Braun T, Rudnicki MA, Arnold HH and Jaenisch R. . 1992 Cell 71: 369–382.
Brennan TJ, Edmondson DG, Li L and Olson EN. . 1991 Proc. Natl. Acad. Sci. USA 88: 3822–3826.
Buskin JN, Jaynes JB, Chamberlain JS and Hauschka SD. . 1985 J. Mol. Evolut. 22: 334–341.
Cattoretti G, Chang CC, Cechova K, Zhang J, Ye BH, Falini B, Louie DC, Offit K, Chaganti RS and Dalla-Favera R. . 1995 Blood 86: 45–53.
Datta R, Banach D, Kojima H, Talanian RV, Alnemri ES, Wong WW and Kufe DW. . 1996 Blood 88: 1936–1943.
Deng C, Zhang P, Harper JW, Elledge SJ and Leder P. . 1995 Cell 82: 675–684.
Dent AL, Shaffer AL, Yu X, Allman D and Staudt LM. . 1997 Science 276: 589–592.
DiBello PR, Withers DA, Bayer CA, Fristrom JW and Guild GM. . 1991 Genetics 129: 385–397.
Fujisawa-Sehara A, Nabeshima Y, Hosoda Y and Obinata T. . 1990 J. Biol. Chem. 265: 15219–15223.
Fukuda T, Miki T, Yoshida T, Hatano M, Ohashi K, Hirosawa S and Tokuhisa T. . 1995 Oncogene 11: 1657–1663.
Fukuda T, Yoshida T, Okada S, Hatano M, Miki T, Ishibashi K, Okabe S, Koseki H, Hirosawa S, Taniguchi M, Miyasaka N and Tokuhisa T. . 1997 J. Exp. Med. 186: 439–448.
Guo K, Wang J, Andres V, Smith RC and Walsh K. . 1995 Mol. Cell. Biol. 15: 3823–1329.
Harrison SD and Travers AA. . 1990 EMBO J. 9: 207–216.
Kanegae Y, Lee G, Sato Y, Tanaka M, Nakai M, Sakaki T, Sugano S and Saito I. . 1995 Nucleic Acids Res. 23: 3816–3821.
Kawamata N, Miki T, Ohashi K, Suzuki K, Fukuda T, Hirosawa S and Aoki N. . 1994 Biochem. Biophys. Res. Commun. 204: 366–374.
Kerckaert JP, Deweindt C, Tilly H, Quief S, Lecocq G and Bastard C. . 1993 Nat. Genet. 5: 66–70.
Kim SJ, Kim KY, Tapscott SJ, Winokur TS, Park K, Fujiki H, Weintraub H and Roberts AB. . 1992 J. Biol. Chem. 267: 15140–15145.
Li L, Zhou J, James G, Heller-Harrison R, Czech MP and Olson EN. . 1992 Cell 71: 1181–1194.
Matthews CC and Feldman EL. . 1996 J. Cell Physiol. 166: 323–331.
Miki T, Kawamata N, Arai A, Ohashi K, Nakamura Y, Kato A, Hirosawa S and Aoki N. . 1994 Blood 83: 217–222.
Miner JH and Wold B. . 1990 Proc. Natl. Acad. Sci. USA 87: 1089–1093.
Miyake S, Makimura M, Kanegae Y, Harada S, Sato Y, Takamori K, Tokuda C and Saito I. . 1996 Proc. Natl. Acad. Sci. USA 93: 1320–1324.
Niwa H, Yamamura K and Miyazaki J. . 1991 Gene 108: 193–199.
Offit K, Jhanwar S, Ebrahim SA, Filippa D, Clarkson BD and Chaganti RS. . 1989 Blood 74: 1876–1879.
Ohashi K, Miki T, Hirosawa S and Aoki N. . 1995 Biochem. Biophys. Res. Commun. 214: 461–467.
Okada S, Zhang H, Hatano M and Tokuhisa T. . 1998 J. Immunol. 160: 2590–2596.
Onizuka T, Moriyama M, Yamochi T, Kuroda T, Kazama A, Kanazawa N, Sato K, Kato T, Ota H and Mori S. . 1995 Blood 86: 28–37.
Rhodes SJ and Konieczny SF. . 1989 Genes Dev. 3: 2050–2061.
Rudnicki MA, Braun T, Hinuma S and Jaenisch R. . 1992 Cell 71: 383–390.
Stewart CE and Rotwein P. . 1996 J. Biol. Chem. 271: 11330–11338.
Schlegel J, Peters I, Orrenius S, Miller DK, Thornberry NA, Yamin TT and Nicholson DW. . 1996 J. Biol. Chem. 271: 1841–1844.
Vaidya TB, Rhodes SJ, Taparowsky EJ and Konieczny SF. . 1989 Mol. Cell. Biol. 9: 3576–3579.
Wang J and Walsh K. . 1996 Science 273: 359–361.
Wang J, Guo K, Wills KN and Walsh K. . 1997 Cancer Res. 57: 351–354.
Wissing D, Mouritzen H, Egeblad M, Poirier GG and Jaattela M. . 1997 Proc. Natl. Acad. Sci. USA 94: 5073–5077.
Wright WE, Sassoon DA and Lin VK. . 1989 Cell 56: 607–617.
Ye BH, Lista F, Lo Coco F, Knowles DM, Offit K, Chaganti RS and Dalla-Favera R. . 1993 Science 262: 747–750.
Ye BH, Chaganti S, Chang CC, Niu H, Corradini P, Chaganti RS and Dalla-Favera R. . 1995 EMBO J. 14: 6209–6217.
Ye BH, Cattoretti G, Shen Q, Zhang J, Hawe N, de Waard R, Leung C, Nouri-shirazi M, Orazi A, Chaganti RS, Rothman P, Stall AH, Pandolfi P and Dalla-Favera R. . 1997 Nat. Genet. 16: 161–170.
Acknowledgements
We thank Drs Izumu Saitoh and Yumi Kanegae (Laboratory of Molecular Genetics, The Institute of Medical Science, University of Tokyo) for the adenovirus vector and the recombinant adenovirus AxCALacZ, Dr Shousei Yoshida (Department of Molecular Genetics, National Institute of Neuroscience) for C2C12 cells and myogenin cDNA, Dr Kenneth Walsh (Division of Cardiovascular Research, St Elizabeth's Medical Center and Tufts University School of Medicine) for 10T1/2 and 10T1/2-MyoD cells, Dr Andrew B Lassar (Department of Biological Chemistry and Molecular Pharmacology, Harvard Medical School) for MyoD cDNA, Dr Jun-ichi Miyazaki (Department of Molecular Embryology Institute of Development, Aging and Cancer, Tohoku University) for pCAGGS, Dr Seiji Okada (Division of Developmantal Genetics, Center for Biological Science, Chiba University School of Medicine) for bcl2 cDNA and Dr Shigeo Ohta (Nippon Medical School, Kawasaki, Japan) for rat bclxL cDNA. We also thank Dr Junko Nishio and Ms Kaori Okada for technical assistance. This work was supported by a Grant-in-Aid for Scientific Research to Tohru Miki and Shinsaku Hirosawa, from the Ministry of Education, Science, Sports and Culture of Japan.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumagai, T., Miki, T., Kikuchi, M. et al. The proto-oncogene Bcl6 inhibits apoptotic cell death in differentiation-induced mouse myogenic cells. Oncogene 18, 467–475 (1999). https://doi.org/10.1038/sj.onc.1202306
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202306
Keywords
This article is cited by
-
IL-33 priming and antigenic stimulation synergistically promote the transcription of proinflammatory cytokine and chemokine genes in human skin mast cells
BMC Genomics (2023)
-
The landscape of chromatin accessibility in skeletal muscle during embryonic development in pigs
Journal of Animal Science and Biotechnology (2021)
-
miR-127 enhances myogenic cell differentiation by targeting S1PR3
Cell Death & Disease (2017)
-
Hemimegalencephaly: a fetal case with neuropathological confirmation and review of the literature
Acta Neuropathologica (2010)
-
Transcription factor FBI-1 acts as a dual regulator in adipogenesis by coordinated regulation of cyclin-A and E2F-4
Journal of Molecular Medicine (2008)