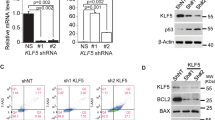
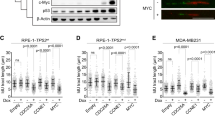
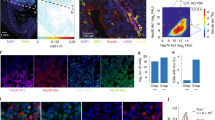
Abstract
p53 tumor suppressor gene controls cell response to a variety of stresses inducing growth arrest or apoptosis in damaged cells. It largely determines the sensitivity of tumor and normal cells to radiation and chemotherapy, and, therefore, defines both the efficacy and limitations of anti-cancer treatment. To determine molecular mechanisms of p53-dependent stress response in normal tissues we identified and compared the spectra of radiation-responsive genes in cells of different origin and p53 status using a cDNA array hybridization technique. The majority of genes identified were p53-dependent and cell type specific. Several of the new p53 responders encode known secreted growth inhibitory factors. This suggests that p53, in addition to its intrinsic antiproliferation activity, can cause `bystander effect' by inducing export of growth suppressive stimuli from damaged cells to neighboring cells. Consistently, a p53-dependent accumulation of factors, which causes growth inhibitory effects in a variety of cell lines, was found after gamma irradiation in the media from established and primary cell cultures and in the urine of irradiated mice. Moreover, p53-dependent factors released by normal human fibroblasts potentiated the cytotoxic effect of a chemotherapeutic drug on co-cultivated tumor cells. This suggests a previously unknown role for normal cells in chemo- and radiation therapy of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Komarova, E., Diatchenko, L., Rokhlin, O. et al. Stress-induced secretion of growth inhibitors: a novel tumor suppressor function of p53. Oncogene 17, 1089–1096 (1998). https://doi.org/10.1038/sj.onc.1202303
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202303
Keywords
This article is cited by
-
Effects of radiation on metastasis and tumor cell migration
Cellular and Molecular Life Sciences (2016)
-
Radiation dose-rate effects on gene expression for human biodosimetry
BMC Medical Genomics (2015)
-
Role of JNK and Contribution of p53 into Growth Potential of Mesenchymal Progenitor Cells In Vitro
Bulletin of Experimental Biology and Medicine (2015)
-
Role of JNK and Contribution of p53 to the Realization of the Growth Potential of Mesenchymal Precursor Cells under the Effect of Fibroblast Growth Factor
Bulletin of Experimental Biology and Medicine (2015)
-
Structural and Mechanical Properties of Gastropod Connective and Smooth Muscle Tissue
Experimental Mechanics (2014)