Abstract
Aim:
To investigate a new strategy to enhance the efficacy of a recombinant pertussis DNA vaccine. The strategy is co-injection with cytokine plasmids as prime, and boosted with purified homologous proteins.
Method:
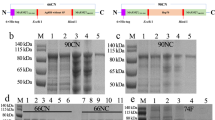
A recombinant pertussis DNA vaccine containing the pertussis toxin subunit 1 (PTS1), fragments of the filamentous hemagglutinin (FHA) gene and pertactin (PRN) gene encoding filamentous hemagglutinin and pertactin were constructed. Balb/c mice were immunized with several DNA vaccines and antigen-specific antibodies anti-PTS1, anti-PRN, anti-FHA, cytokines interleukin (IL)-10, IL-4, IFN-γ, TNF-α, and spleno-cyte-proliferation assay were used to describe immune responses.
Results:
The recombinant DNA vaccine could elicit similar immune responses in mice as that of separate plasmids encoding the 3 fragments, respectively. Mice immunized with DNA and boosted with the corresponding protein elicited more antibodies than those that received DNA as boost. In particular, when the mice were co-immunized with murine granulocyte-macrophage colony-stimulating factor plasmids and boosted with proteins, all 4 cytokines and the 3 antigen-specific antibodies were significantly increased compared to the pVAX1 group. Anti-PTS1, anti-FHA, IL-4 and TNF-α elicited in the colony stimulating factor (CSF) prime-protein boost group showed significant increase compared to all the other groups.
Conclusion:
This prime and boost strategy has proven to be very useful in improving the immunogenicity of DNA vaccines against pertussis.
Similar content being viewed by others
Article PDF
References
Franco E, Giambi C, Ialacci R, Maurici M . Pertussis vaccination for adolescents and adults. Expert Opin Biol Ther 2004; 4: 1669–76.
Pichichero ME, Casey JR . Acellular pertussis vaccines for adolescents. Pediatr Infect Dis J 2005; 24: S117–26.
Kamachi K, Arakawa Y . Expression of a C terminally truncated form of pertussis toxin S1 subunit effectively induces protection against pertussis toxin following DNA-based immunization. Infect Immun 2004; 72: 4293–6.
Halperin SA . Pertussis-a disease and vaccine for all ages. N Engl J Med 2005; 353: 1615–7.
Kamachi K, Konda T, Arakawa Y . DNA vaccine encoding pertussis toxin S1 subunit induces protection against Bordetella pertussis in mice. Vaccine 2003; 21: 4609–15.
Bassinet L, Fitting C, Housset B, Cavaillon JM, Guiso N . Bordetella pertussis adenylate cyclase-hemolysin induces interleukin-6 secretion by human tracheal epithelial cells. Infect Immun 2004; 72: 5530–3.
Burnette WN, Cieplak W, Mar VL, Kaljot KT, Sato H, Keith JM . Pertussis toxin S1 mutant with reduced enzyme activity and a conserved protective epitope. Science 1988; 242: 72–4.
Loosmore SM, Zealey GR, Boux HA, Cockle SA, Radika K, Fahim RE, et al. Engineering of genetically detoxified pertussis toxin analogs for development of a recombinant whooping cough vaccine. Infect Immun 1990; 58: 3653–62.
Charles IG, Dougan G, Pickard D, Chatfield S, Smith M, Novotny P, et al. Molecular cloning and characterization of protective outer membrane protein P.69 from Bordetella pertussis. Proc Natl Acad Sci USA 1989; 86: 3554–8.
Belcher CE, Drenkow J, Kehoe B, Gingeras TR, McNamara N, Lemjabbar H, et al. The transcriptional responses of respiratory epithelial cells to Bordetella pertussis reveal host defensive and pathogen counter-defensive strategies. Proc Natl Acad Sci USA 2000; 97: 13847–52.
Smith AM, Guzman CA, Walker MJ . The virulence factors of Bordetella pertussis: a matter of control. FEMS Microbiol Rev 2001; 25: 309–33.
Wolff JA, Malone RW, Williams P, Chong WG, Acsadi A, Jani A et al. Direct gene transfer into mouse muscle in vivo. Science 1990; 247: 1465–8.
Heininger U . Recent progress in clinical and basic pertussis research. Eur J Pediatr 2001; 160: 203–13.
Doria-Rose NA . Haigwood NL. DNA vaccine strategies: candidates for immune modulation and immunization regimens. Methods 2003; 31: 207–16.
Du DW, Jia ZS, Li GY, Zhou YY . HBV DNA vaccine with adjuvant cytokines induced specific immune responses against HBV infection. World J Gastroenterol 2003; 9: 108–11.
Leachman SA, Tigelaar RE, Shlyankevich M, Slade MD, Irwin M, Chang E, et al. Granulocyte-macrophage colony-stimulating factor priming plus papillomavirus E6 DNA vaccination: effects on papilloma formation and regression in the cottontail rabbit papillomavirus-rabbit model. J Virol 2000; 74: 8700–8.
Nimal S, McCormick AL, Thomas MS, Heath AW . An interferon gamma-gp120 fusion delivered as a DNA vaccine induces enhanced priming. Vaccine 2005; 23: 3984–90.
Liu DF, Phillips E, Wizemann TM, Siegel MM, Tabei K, Cowell JL, et al. Characterization of a recombinant fragment that contains a carbohydrate recognition domain of the filamentous hemagglutinin. Infect Immun 1997; 65: 3465–8.
He Q, Makinen J, Berbers G, Mooi FR, Viljanen MK, Arvilommi H, et al. Bordetella pertussis protein pertactin induces type-specific antibodies: one possible explanation for the emergence of antigenic variants? J Infect Dis 2003; 187: 1200–5.
Carter CR, Dagg BM, Whitmore KM, Keeble JR, Asokanathan C . Xing D, et al. The effect of pertussis whole cell and acellular vaccines on pulmonary immunology in an aerosol challenge model. Cell Immunol 2004; 227: 51–8.
Lasaro MO, Luiz WB, Sbrogio-Almeida ME, Ferreira LC . Prime- boost vaccine regimen confers protective immunity to human-derived enterotoxigenic Escherichia coli. Vaccine 2005; 23: 2430–8.
Mielcarek N, Cornette J, Schacht AM, Pierce RJ, Locht C, Capron A, et al. Intranasal priming with recombinant Bordetella pertussis for the induction of a systemic immune response against a heterologous antigen. Infect Immun 1997; 65: 544–50.
Kim JJ, Yang JS, Dentchev T, Dang K, Weiner DB . Chemokine gene adjuvants can modulate immune responses induced by DNA vaccines. J Interferon Cytokine Res 2000; 20: 487–98.
Sun X, Hodge LM, Jones HP, Tabor L, Simecka JW . Co-expression of granulocyte-macrophage colony-stimulating factor with antigen enhances humoral and tumor immunity after DNA vaccination. Vaccine 2002; 20: 1466–74.
Kamath AT, Hanke T, Briscoe H, Britton WJ . Co-immunization with DNA vaccines expressing granulocyte-macrophage colony-stimulating factor and mycobacterial secreted proteins enhances T-cell immunity, but not protective efficacy against Mycobacte-rium tuberculosis. Immunology 1999; 96: 511–6.
Sin JI, Sung JH, Suh YS, Lee AH, Chung JH, Sung YC . Protective immunity against heterologous challenge with encephalo-myo-carditis virus by VP1 DNA vaccination: effect of coinjection with a granulocyte-macrophage colony stimulating factor gene. Vaccine 1997; 15: 1827–33.
Tanghe A, D'Souza S, Rosseels V, Denis O, Ottenhoff TH, Dalemans W, et al. Improved immunogenicity and protective efficacy of a tuberculosis DNA vaccine encoding Ag85 by protein boosting. Infect Immun 2001; 69: 3041–7.
Wang QM, Sun SH, Hu ZL, Yin M, Xiao CJ, Zhang JC . Improved immunogenicity of a tuberculosis DNA vaccine encoding ESAT6 by DNA priming and protein boosting. Vaccine 2004; 22: 3622–7.
Bharati K, Appaiahgari MB, Vrati S . Effect of cytokine-encoding plasmid delivery on immune response to Japanese encephalitis virus DNA vaccine in mice. Microbiol Immunol 2005; 49: 349–53.
He X, Tsang TC, Zhang T, Luo P, Harris DT . Antigen epitope-expressing cytokines for DNA immunization. Vaccine 2005; 23: 1966–72.
Chattergoon MA, Saulino V, Shames JP, Stein J, Montaner LJ, Weiner DB . Co-immunization with plasmid IL-12 generates a strong T-cell memory response in mice. Vaccine 2004; 22: 1744–50.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported in part by Shanghai Leading Academic Discipline Project (No T0206).
Rights and permissions
About this article
Cite this article
Li, Qt., Zhu, Yz., Chu, Jy. et al. Granulocyte-macrophage colony-stimulating factor DNA prime-protein boost strategy to enhance efficacy of a recombinant pertussis DNA vaccine. Acta Pharmacol Sin 27, 1487–1494 (2006). https://doi.org/10.1111/j.1745-7254.2006.00456.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00456.x