Abstract
Interferon therapy is the most effective treatment for achieving clinical cure in chronic hepatitis B (CHB) patients. However, the treatment outcomes of interferon therapy are uncertain, multiple side effects can occur during treatment, and the treatment is expensive. Although these characteristics may affect patients’ quality of life, research examining this topic is limited. We used a cross-sectional design to examine 100 CHB patients receiving interferon, 100 receiving nucleoside/nucleotide analogues, and 87 receiving non-antiviral treatment. Characteristic information, the Hepatitis B Quality of Life Instrument, Connor Davidson Resilience Scale, and Work Productivity and Activity Impairment Questionnaire were used to collect information. We found that quality of life in the interferon treatment group was higher than that in the non-antiviral treatment and nucleoside/nucleotide analogue treatment groups (p < 0.05). The factors influencing quality of life were resilience, presenteeism, hair loss, and antiviral treatment (p < 0.05). Although interferon therapy has some potential side effects, the results suggested that it did not negatively affect quality of life. Overall, interferon therapy did not have a major impact on CHB patients’ daily lives and work.
Similar content being viewed by others
Introduction
Chronic hepatitis B (CHB) is a systemic infectious disease caused by the hepatitis B virus (HBV) with liver damage as the primary cause1. According to World Health Organization data released in 2019, the global prevalence of CHB infection was approximately 3.5%, with 820,000 people dying from liver failure, cirrhosis, and hepatocellular carcinoma caused by HBV infection2. Clinical cure is the ideal treatment target recommended in the latest guidelines for the prevention and treatment of CHB1,2. A previous study reported that patients who are clinically cured can safely discontinue drug treatment for an extended period of time, with a reported incidence of hepatocellular carcinoma of 0%–1% within 5 years3. Immunomodulators such as pegylated interferon alpha (PEG-IFNα), are currently considered to be the most effective antiviral therapy for clinical cure1. However, the uncertainty of the outcomes of interferon therapy, coupled with a substantial incidence of adverse reactions during treatment, the potential for relapse even after achieving clinical cure, the long treatment period, and the high cost, may all have an impact on patients’ physiological, psychological, and social functioning4,5,6. Previous studies of interferon therapy for CHB patients mainly focused on therapeutic efficacy, prognosis, and predictive indicators7, with little attention paid to psychosocial aspects.
Over many years, the traditional medical model has changed to the bio-psycho-social medical model, and medical research has gradually begun to pay attention to the relationships between human mental health and the development and outcomes of diseases. Therefore, the psychosocial aspects of patients undergoing interferon therapy for CHB needs attention. Quality of life is a concept that integrates biomedical, sociological, and psychological aspects to provide a more comprehensive picture of health, and to assess the effectiveness of treatments8,9. Previous studies have shown that quality of life decreases in patients with chronic hepatitis C during interferon therapy10,11. However, it is not clear whether interferon therapy affects quality of life in patients with CHB during interferon therapy. This current study aimed to explore the relationship between interferon therapy and quality of life in patients with CHB.
Methods
Study design and sample
From April 2022 to May 2023, a convenience sampling method was used to recruit 100 CHB patients who received interferon therapy, and 187 non-interferon treated CHB patients in the outpatient clinic of the Infection Department of a Grade III Level A hospital which annually receives nearly 70,000 outpatients with CHB. The sample size was calculated using PASS15.0. In previous study12 assessing the quality of life in patients with CHB and pre-investigation of quality of life in patients with CHB treated with interferon, the quality of life scores of CHB patients were (1952.97 ± 742.19) points and (2274.59 ± 677.26) points. We selected “Two-Sample T-Tests” as the calculation method. When α = 0.05 (bilateral), power = 0.80, and the expected effect size (Cohen’s d) = 0.5, we calculated that 78 cases were needed in each group. The sample size was expanded by 10% to 87 cases in each group.
With the consent of the hospital administration, the researchers conducted an investigation in the outpatient infection department. The purpose of the study was explained to participants before the investigation. After obtaining participants’ consent, data were collected using an online method powered by www.wjx.cn. To control the quality of questionnaire responses, all patients filled out the questionnaire under standardised guidance. If the time taken to answer the questionnaire was less than 6 min, the questionnaire was considered to be of low quality.
Inclusion criteria: (1) patients met the relevant diagnostic criteria of the Chronic Hepatitis B Prevention and Treatment Guidelines1; (2) patients in the interferon group received PEG-IFNα treatment for ≥ 1 month; (3) patients volunteered to participate in this study and provided written informed consent. Exclusion criteria: (1) patients had other types of hepatitis virus infectious; (2) patients had cirrhosis, hepatocellular carcinoma, major diseases of the heart, brain, lungs, or other organs; (3) the quality of patients’ questionnaire responses was poor, or they experienced difficulties in communication, reading, writing, or comprehension.
Measurements
Socio-demographic and health status. A self-designed questionnaire was used, including gender, age, education level, occupation, marital status, type of medical insurance, hepatitis B history, family history of hepatitis B, side effects, hepatitis B surface antigen (HBsAg), hepatitis B e antigen (HBeAg) and hepatitis B virus DNA (HBV DNA).
The Hepatitis B Quality of Life Instrument (HBQOL). The Hepatitis B Quality of Life Instrument (version 1.0, HBQOL V.1.0) was produced by Spiegel in 200713,14. The HBQOL has a total of 31 items, and seven dimensions: psychological well-being, anticipation, vitality, stigma, vulnerability, transmission, viral response, each item is scored on a 5-point scale, and the conversion formula of the score is: (5-selected option) multiplied by 25. The higher the score, the higher the quality of life of the CHB patients. Cronbach’s α coefficient was 0.960.
Connor Davidson Resilience Scale (CD-RISC). The Connor Davidson Resilience Scale (CD-RISC) was produced by Connor and Davidson in 200315. In this study, the Chinese version of the CD-RISC was used, which was translated and adapted by Yu and Zhang in 200716, which included 25 items in three dimensions: tenacity, strength, and optimism. A 5-point Likert scale method was adopted, and Cronbach’s α coefficient was 0.937.
Work Productivity and Activity Impairment Questionnaire Specific Health Problem (WPAI-SHP). The WAPI-SHP was compiled by Reilly and colleages17 in 1993. This questionnaire consists of six questions and is used to determine employment status, time of absence caused by illness, time of absence for other reasons, actual working time, degree of impact of illness on work productivity at work, and degree of impact of illness on activities outside work. The WAPI-SHP has four components, providing scores for absenteeism, presenteeism, activity impairment, and overall work impairment. Higher scores indicate greater negative impact.
Statistical analysis
Continuous and categorical variables were reported as mean ± standard deviation (SD) or frequencies (percentages). Demographic data and health data were analyzed using descriptive statistics. The quality of life, general information, mental resilience, and working ability of the interferon treatment group, non-antiviral treatment group, and nucleoside/nucleotide analogues (NAs) treatment group were compared using χ2 tests, analysis of variance, and the Kruskal–Wallis H test. Multiple linear regression analysis was performed to examine the influencing factors of quality of life in CHB patients. All analyses were performed using SPSS 22.0 Statistics (IBM Corporation, Armonk New York, USA). The level of statistical significance was set at p < 0.05.
Ethical approval and consent to participate
The study protocol was approved by the Biomedical Ethics Committee of Xi’an Jiaotong University Health Science Center (Approval No. 2022–1430). Participation was voluntary, and informed consent was given at the top of the questionnaire. Participants anonymously answered questions that were consistent with their agreement to participate and agreed to the publication of the results. The investigation conformed with the principles outlined in the Declaration of Helsinki (Br Med J 1964;ii:177).
Results
A total of 300 questionnaires were collected. 13 participants were excluded from the questionnaire because of low quality responses, and the effective recovery rate was 95.7%.
Demographic analysis of CHB patients
Demographic analysis of CHB patients receiving interferon treatment, NAs treatment, and non-antiviral therapy revealed no significant differences in gender, age, education, occupation, marital status, medical insurance, family history, or hepatitis B history (p > 0.05), as shown in Table 1.
Factors influencing quality of life in CHB patients
Univariate analysis showed that the factors affecting the quality of life of CHB patients were as follows: level of education, profession, health insurance, hair loss, antiviral treatment, HBsAg, absenteeism, presenteeism, overall work impairment, activity impairment, resilience, tenacity, strength, and optimism (p < 0.05), as shown in Tables 2 and 3. Continuous variables, such as quality of life, resilience, presenteeism, and HBsAg were assigned as measured values. Binary variables were assigned, with a value of 1 for hair loss and 0 for no hair loss. Dummy variables were introduced to describe the multi-categorical variable “antiviral treatment,” with a value of “0,0” for interferon treatment, “1,0” for NAs treatment, and “0,1” for non-antiviral treatment. The quality-of-life score of CHB patients was used as the dependent variable, and level of education, profession, and health insurance were used as independent variables. The regression model had statistical significance F = 21.516, (p < 0.001), and adjusted R2 = 0.315. The results indicated that the factors affecting the quality of life of CHB patients were resilience, presenteeism, hair loss, and antiviral treatment (p < 0.05), as shown in Table 4.
Effect of different antiviral therapy on quality of life in patients with CHB
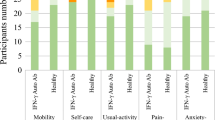
CHB patients treated with interferon scored higher for quality of life, psychological well-being, anticipation, stigma, vulnerability, transmission, and viral response compared with non-antiviral and NAs treatment patients (p < 0.05), as shown in Table 5 and Fig. 1.
Comparison of quality of life of CHB patients in interferon treated group, untreated group and NAs treated group. The total score for quality of life (Total) Psychological Well-Being (PW), Anticipation (AA), Vitality (Vitality), Stigma (SG), Vulnerability (VL), Transmission (TS), Viral Response (VR). *P < 0.05, **P < 0.001.
Discussion
Our study found that CHB patients treated with interferon had higher quality of life compared with those treated with antiviral therapy and NAs. Interferon therapy may be associated with an improvement in quality of life for CHB patients.
There are several possible explanations for the higher quality of life of CHB patients treated with interferon. First, interferon therapy has been reported to induce negative HBV DNA conversion, HBeAg serological conversion, and negative HBsAg conversion to achieve clinical cure; CHB patients who achieve clinical cure are reported to have a lower incidence of liver cirrhosis and hepatocellular carcinoma18, which is a key factor affecting the psychosocial health of CHB patients19. A previous study reported that CHB patients who exhibited better responses during antiviral therapy had a higher quality of life20. Second, decreased quality of life caused by HBV infection is reported to be primarily related to impacts on mental health and social health, with little impact on physical health21. Although people with CHB have fewer physical symptoms and are more stable, the risk of disease progression and severe psychosocial harms19,22,23 (e.g., fear of transmitting to others, disclosure of HBV infection, discrimination, and anxiety, depression, etc.) can compromise their quality of life. A multicenter study conducted in Turkey24 reported that CHB patients receiving antiviral therapy exhibited more severe disease symptoms but had better mental health and social functioning and less anxiety compared with CHB patients who did not receive antiviral therapy. In the current study, although interferon therapy caused some adverse reactions, quality of life was still higher for CHB patients receiving interferon therapy compared with those that received non-interferon treatment, indicating that interferon treatment had a beneficial effect on psychosocial health in CHB patients.
In the current study, the factors affecting the quality of life of CHB patients were resilience, presenteeism, hair loss, and antiviral treatment. The resilience score of CHB patients treated with interferon in the current study was 64.6 ± 16.26 points, which was slightly higher than that that reported in a previous study (61.64 ± 15.36)25. Previous studies have reported that patients with a high level of resilience tend to have a positive attitude and exhibit coping behaviors, and can manage the stress and impact of their disease in a positive way, leading to improved quality of life in a wide range of chronic diseases16,26,27,28.
As an important aspect of people’s social lives, work is a major factor in social health, which is one of the components of quality of life29,30. Some of the items in the quality- of-life scale used in the current study were directly or indirectly related to the impact of current health conditions on work, such as “fear that having hepatitis B will be discovered by someone, such as a supervisor.” Decreased work productivity may reflect a decrease in social health, which can affect mental health, thus further affecting the quality of life.
Loss of hair occurring in any disease can negatively impact quality of life, and is often associated with loss of self-esteem and psychosocial problems31. Hair loss is one of the common side effects of interferon therapy32,33, and the incidence of hair loss in the interferon treatment group in our study was 27%. The impact on life and work, and potential side effects, are significant reasons for CHB patients’ dissatisfaction with interferon therapy34. However, the side effects of interferon treatment, including interferon-induced hair loss, are temporary, and most people regain growth within 3 to 6 months after stopping interferon. Therefore, the impact of potential hair loss on daily life should not be a major concern for CHB patients.
The results of the current study indicated a positive association between interferon therapy and quality of life in CHB patients. Improvement in quality of life was found to be influenced by resilience, presenteeism, and hair loss. Despite potential temporary side effects, the results indicated that interferon therapy alleviated psychological and social distress, contributing to enhanced quality of life. Thus, the impact of interferon therapy on the daily lives and work of CHB patients does not appear to be a major cause for concern.
Limitations
Because of time constraints, the present study was a cross-sectional investigation of CHB patients treated with interferon. In future, longitudinal studies should be carried out to further determine the impact of interferon treatment on CHB patients’ quality of life by examining changes in quality of life over time during treatment.
Data availability
All data generated or analysed during this study are included in this published article. The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Terrault, N. A. et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology 67, 1560–1599. https://doi.org/10.1002/hep.29800 (2018).
Jeng, W.-J., Papatheodoridis, G. V. & Lok, A. S. F. Hepatitis B.. Lancet 401, 1039–1052. https://doi.org/10.1016/s0140-6736(22)01468-4 (2023).
Yip, T. C. et al. Impact of age and gender on risk of hepatocellular carcinoma after hepatitis B surface antigen seroclearance. J. Hepatol. 67, 902–908. https://doi.org/10.1016/j.jhep.2017.06.019 (2017).
Liu, Y. et al. Analysis of clinical characteristics of thyroid disorders in patients with chronic hepatitis B treated with pegylated-interferon alpha. BMC Endocr. Disord. https://doi.org/10.1186/s12902-023-01371-w (2023).
Ye, J. & Chen, J. Interferon and hepatitis B: Current and future perspectives. Front. Immunol. https://doi.org/10.3389/fimmu.2021.733364 (2021).
Dai, Z. et al. Cost-effectiveness analysis of first-line treatment for chronic hepatitis B in China. Clin. Microbiol. Infect. https://doi.org/10.1016/j.cmi.2021.06.024 (2022).
Li, Q. et al. Interferon and interferon-stimulated genes in HBV treatment. Front. Immunol. https://doi.org/10.3389/fimmu.2022.1034968 (2022).
Buti, M. et al. Chronic hepatitis D associated with worse patient-reported outcomes than chronic hepatitis B. Jhep. Rep. https://doi.org/10.1016/j.jhepr.2021.100280 (2021).
Younossi, Z. M. et al. Clinical and patient-reported outcome profile of patients with hepatitis B viral infection from the Global Liver Registry (TM). J. Viral Hepat. 30, 335–344. https://doi.org/10.1111/jvh.13800 (2023).
Younossi, Z. M. et al. Minimal impact of sofosbuvir and ribavirin on health related quality of life in Chronic Hepatitis C (CH-C). J. Hepatol. 60, 741–747. https://doi.org/10.1016/j.jhep.2013.12.006 (2014).
Ahn, S. H. et al. Impact of interferon-based treatment on quality of life and work-related productivity of Korean patients with chronic hepatitis C. Gut Liver 14, 368–376. https://doi.org/10.5009/gnl18100 (2020).
Liu, Z., Liu, H. & Meng, Q. Test of reliability, validity and sensitivity of Chinese version of Ouality of Life Scale for patients with chronic HBV infection. Chin. Nurs. Res. 31, 4078–4081. https://doi.org/10.3969/j.issn.1009-6493.2017.32.012 (2017).
Ha, Y. et al. Validation of the liver disease quality of life instrument 1.0 in patients with chronic hepatitis B: A prospective study. J. Clin. Med. https://doi.org/10.3390/jcm8050656 (2019).
Spiegel, B. M. et al. Development and validation of a disease-targeted quality of life instrument in chronic hepatitis B: The hepatitis B quality of life instrument, version 1.0. Hepatology 46, 113–121. https://doi.org/10.1002/hep.21692 (2007).
Campbell-Sills, L. & Stein, M. B. Psychometric analysis and refinement of the Connor-Davidson Resilience Scale (CD-RISC): Validation of a 10-item measure of resilience. J. Traum. Stress 20, 1019–1028. https://doi.org/10.1002/jts.20271 (2007).
Vatanparast, M. et al. Resilience as the predictor of quality of life in the infertile couples as the most neglected and silent minorities. J. Reprod. Infant Psychol. 40, 216–227. https://doi.org/10.1080/02646838.2020.1843613 (2022).
Reilly, M. C., Zbrozek, A. S. & Dukes, E. M. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics 4, 353–365. https://doi.org/10.2165/00019053-199304050-00006 (1993).
Vittal, A. et al. Systematic review with meta-analysis: the impact of functional cure on clinical outcomes in patients with chronic hepatitis B. Aliment. Pharmacol. Therap. 55, 8–25. https://doi.org/10.1111/apt.16659 (2022).
Tu, T., Block, J. M., Wang, S., Cohen, C. & Douglas, M. W. The lived experience of chronic hepatitis B: A broader view of its impacts and why we need a cure. Viruses https://doi.org/10.3390/v12050515 (2020).
Kim, J. H. et al. Virologic response to therapy increases health-related quality of life for patients with chronic hepatitis B. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 10, 291–296. https://doi.org/10.1016/j.cgh.2011.09.031 (2012).
Zhang, M. et al. Assessing health-related quality of life and health utilities in patients with chronic hepatitis B-related diseases in China: A cross-sectional study. BMJ open 11, e047475. https://doi.org/10.1136/bmjopen-2020-047475 (2021).
Li, G. et al. Effects of depression, anxiety, stigma, and disclosure on health-related quality of life among chronic hepatitis B patients in Dalian, China. Am. J. Trop. Med. Hyg. 102, 988–994. https://doi.org/10.4269/ajtmh.19-0007 (2020).
Han, B. et al. The experience of discrimination of individuals living with chronic hepatitis B in four provinces of China. PloS one 13, e0195455. https://doi.org/10.1371/journal.pone.0195455 (2018).
Karacaer, Z. et al. Quality of life and related factors among chronic hepatitis B-infected patients: A multi-center study, Turkey. Health Qual. Outc. 14, 153. https://doi.org/10.1186/s12955-016-0557-9 (2016).
Wang, X., Zhang, A., Yan, J., Liu, Y. & Long, F. Path analysis of self-efficacy, coping style and social support on resilience in patients with chronic hepatitis B. Chin J Behav Med Brain Sci 28, 988–993 (2019).
Dai, W. et al. The actuality of resilience, social support and quality of life among patients with inflammatory bowel disease in China. Nursing Open 9, 2190–2198. https://doi.org/10.1002/nop2.946 (2022).
Liu, Z., Zhou, X., Zhang, W. & Zhou, L. Factors associated with quality of life early after ischemic stroke: The role of resilience. Top. Stroke Rehabil. 26, 335–341. https://doi.org/10.1080/10749357.2019.1600285 (2019).
Temprado Albalat, M. D., Garcia Martinez, P., Ballester Arnal, R. & Collado-Boira, E. J. The relationship between resilience and quality of life in patients with a drainage enterostomy. J. Health Psychol. 25, 1483–1497. https://doi.org/10.1177/1359105318761555 (2020).
Bernuzzi, C., Sommovigo, V. & Setti, I. The role of resilience in the work-life interface: A systematic review. Work 73, 1147–1165. https://doi.org/10.3233/wor-205023 (2022).
Cooke, F. L., Cooper, B., Bartram, T., Wang, J. & Mei, H. X. Mapping the relationships between high-performance work systems, employee resilience and engagement: A study of the banking industry in China. Int. J. Hum. Res. Manag. 30, 1239–1260. https://doi.org/10.1080/09585192.2015.1137618 (2019).
Liu, L. Y., King, B. A. & Craiglow, B. G. Health-related quality of life (HRQoL) among patients with alopecia areata (AA): A systematic review. J. Am. Acad. Dermatol. 75, 806-U264. https://doi.org/10.1016/j.jaad.2016.04.035 (2016).
Marcellin, P. et al. Combination of tenofovir disoproxil fumarate and peginterferon α-2a increases loss of hepatitis B surface antigen in patients with chronic hepatitis B. Gastroenterology 150, 134. https://doi.org/10.1053/j.gastro.2015.09.043 (2016).
Wang, H. M. et al. Clinical effect and influencing factors of pegylated interferon alfa-2a and entecavir monotherapy among children with HBeAg-positive chronic hepatitis B based on a real-world study. Zhonghua gan zang bing za zhi = Zhonghua ganzangbing zazhi = Chin. J. Hepatol. 30, 1056–1062. https://doi.org/10.3760/cma.j.cn501113-20210225-00094 (2022).
Duan, Z. P. et al. Awareness of HBV functional cure and attitude toward related clinical trials among patients with chronic hepatitis B in china. Pat. Prefer. Adher. 17, 2063–2072. https://doi.org/10.2147/ppa.S422916 (2023).
Acknowledgements
We would like to thank all participants in this study. We thank Benjamin Knight, MSc., from Liwen Bianji (Edanz) (www.liwenbianji.cn) for editing the English text of a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
M.Z. wrote the main manuscript text, and condected the study, and the data analysis; M.W., W.W, supported the data collection and data analysis; S.L., Z.X. designed the study and supervised the data collection and the manuscript; all authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, M., Wan, M., Wang, W. et al. Effect of interferon therapy on quality of life in patients with chronic hepatitis B. Sci Rep 14, 2461 (2024). https://doi.org/10.1038/s41598-024-51292-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-51292-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.