Abstract
The intestinal immune response is crucial in maintaining a healthy gut, but the enhanced migration of macrophages in response to pathogens is a major contributor to disease pathogenesis. Integrins are ubiquitously expressed cellular receptors that are highly involved in immune cell adhesion to endothelial cells while in the circulation and help facilitate extravasation into tissues. Here we show that specific deletion of the Tln1 gene encoding the protein talin-1, an integrin-activating scaffold protein, from cells of the myeloid lineage using the Lyz2-cre driver mouse reduces epithelial damage, attenuates colitis, downregulates the expression of macrophage markers, decreases the number of differentiated colonic mucosal macrophages, and diminishes the presence of CD68-positive cells in the colonic mucosa of mice infected with the enteric pathogen Citrobacter rodentium. Bone marrow-derived macrophages lacking expression of Tln1 did not exhibit a cell-autonomous phenotype; there was no impaired proinflammatory gene expression, nitric oxide production, phagocytic ability, or surface expression of CD11b, CD86, or major histocompatibility complex II in response to C. rodentium. Thus, we demonstrate that talin-1 plays a role in the manifestation of infectious colitis by increasing mucosal macrophages, with an effect that is independent of macrophage activation.
Similar content being viewed by others
Introduction
Macrophages are an essential component of the intestinal immune response. Under normal conditions, the intestinal lamina propria is home to the largest population of mononuclear leukocytes in the body with macrophages being the most abundant subpopulation1. Intestinal macrophages are unique compared to other tissue macrophages in that they are continuously replenished from circulating blood monocytes as they help maintain epithelial renewal and immune homeostasis in the presence of the gut microbiota1. Inflammatory conditions of the intestines are often the result of perturbation of the normal gut flora, pathogenic infection, or an inherent dysregulation of the immune response. During infection with pathogenic bacteria, such as enteropathogenic Escherichia coli (EPEC) and Shiga toxin-producing E. coli, the enhanced immune cell recruitment and activation can be both protective and deleterious, and is a major cause of the resulting pathology2.
Recruited circulating monocytes enter tissues through integrin binding to adhesion molecules on endothelial cells3,4,5. Integrin ligand affinity and activation is facilitated by talin-1, a cytoskeletal scaffold protein that interacts with the cytoplasmic domain of the β-subunit of integrins and induces a confirmational change to the extracellular domain6,7. In this study, we sought to elucidate the role of talin-1 in macrophages during colonic infection by Citrobacter rodentium, the rodent equivalent of EPEC, a well-established model of infectious colitis8,9,10. Myeloid cell-specific knockdown of Tln1 using the Lyz2-cre driver mouse resulted in decreased epithelial damage and inflammation that was associated with decreased transcript levels of macrophage markers, less macrophages, and less differentiated macrophages in the infected tissues. This outcome was independent of a cell-autonomous effect of talin-1 in macrophage activation and function ex vivo. Overall, our data indicate that talin-1 regulates the recruitment of macrophages in response to the enteric murine pathogen C. rodentium and thus facilitates the inflammatory response.
Results
Mice lacking talin-1 in the myeloid cell lineage exhibit less epithelial damage and inflammation during pathogenic colitis
Tln1fl/fl mice were crossed with C57BL/6 Lyz2cre/cre mice and the resulting offspring were crossed to generate Tln1fl/fl;Lyz2+/+ (Tln1fl/fl) and Tln1fl/fl;Lyz2cre/cre (Tln1Δmye) littermates. The knockdown of Tln1 mRNA (Fig. 1A) and talin-1 protein (Fig. 1B,C; Supplementary Fig. S1) was evidenced in bone marrow-derived macrophages (BMmacs) isolated from Tln1Δmye mice and infected or not ex vivo with C. rodentium. Note that C. rodentium infection had no effect on the expression of Tln1 mRNA (Fig. 1A) or talin-1 protein (Fig. 1B and C; Supplementary Fig. S1) in Tln1fl/fl BMmacs. Male mice were then infected via oral gavage with 5 × 108 colony forming units (CFUs) of C. rodentium and monitored for 14 days, as previously described8,9,10. We did not observe differences in body weight loss (Fig. 2A) or bacterial burden (Fig. 2B) between Tln1fl/fl and Tln1Δmye mice. Consistent with previous studies11,12, infection with C. rodentium resulted in mucosal hyperplasia, erosion and ulceration of the epithelium, and immune cell invasion of the mucosa and submucosa (Fig. 2C). Notably, histologic injury scores were attenuated in Tln1Δmye mice compared to Tln1fl/fl littermates (Fig. 2D), due to a reduction in both components of the composite score, epithelial damage, and total inflammation (Fig. 2E).
Myeloid cell-specific deletion of Tln1. BMmacs were generated form Tln1fl/fl and Tln1Δmye mice and infected with C. rodentium. (A) Expression of Tln1 mRNA determined by RT-real-time PCR 6 h post-infection; n = 3–4 mice per genotype. (B) Western blot of talin-1 protein (270 kDa) expression and (C) densitometry analysis at 24 h post-infection; n = 3 mice per group. All values are reported as mean ± SEM. Statistical analyses, where shown; *P < 0.05 determined by 1-way ANOVA and Tukey post hoc test.
Loss of talin-1 in myeloid cells protects mice from C. rodentium-induced injury. Tln1fl/fl and Tln1Δmye male littermates were orally inoculated with 5 × 108 CFU of C. rodentium by gavage and observed for 14 days. n = 7 uninfected mice and n = 13–16 infected mice per genotype. Data pooled from 2 independent experiments. (A) Daily body weights depicted as a percent of initial body weight. (B) C. rodentium colonization of the colon determined by counting CFUs from serial dilutions of homogenized tissues and normalized to tissue weight on day 14 post-infection. (C) Representative H&Es of Swiss-rolled fixed tissues. Note that the low and high magnification images are from the same slide for each genotype. Scale bars represent 200 μm. (D) Histologic injury score assessed by a pathologist using the H&E-stained tissues and is the sum of (E) epithelial damage and total inflammation. All values are reported as mean ± SEM. Statistical analyses, where shown; *P < 0.05, ***P < 0.001, and ****P < 0.0001 determined by (D) 1-way ANOVA and Tukey post hoc test and (E) Student’s t test.
To determine if these findings are specific to bacterially-induced colitis, we treated Tln1fl/fl and Tln1Δmye littermates with 4% dextran sulfate sodium (DSS), a chemically-induced model of epithelial injury9,13. When compared to Tln1fl/fl mice, Tln1Δmye mice did not show differences in body weight loss (Supplementary Fig. S2A), colon length (Supplementary Fig. S2B), or histologic injury score (Supplementary Fig. S2C). These results suggest that talin-1 in myeloid cells contributes to C. rodentium pathogenesis and is specific to pathogenic infection.
Mice with myeloid cell-specific deletion of Tln1 express lower levels of macrophage markers and exhibit less macrophages in the colon with C. rodentium infection
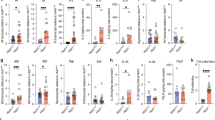
We next assessed the expression of immune effectors typically associated with C. rodentium infection in the colon. The genes encoding for markers of inflammation, namely Tnf, Il1b, Il6, Il23a, and Cxcl10 were induced with infection in Tln1fl/fl mice and significantly decreased in the colon tissues from infected Tln1Δmye mice compared to infected Tln1fl/fl mice (Fig. 3A). Additionally, the levels of the transcripts encoding arginase 1 (Arg1) and tumor necrosis factor super family 14 (Tnfsf14; Light), which are anti-inflammatory during colitis (22,23), were also significantly decreased in infected Tln1Δmye colon tissues (Fig. 3B). The expression levels of Il10 and Tgfb1 were induced in infected mice and not different between genotypes (Fig. 3B). We observed a significant reduction in the expression of the Th1 T cell marker Ifng, but no change in the expression of the Th17 marker Il17a, or the Th22 marker Il22, in infected Tln1Δmye colon tissues (Fig. 3C).
Knockdown of talin-1 in myeloid cells reduces expression of macrophage markers in vivo. Whole colon tissue mRNA expression of (A) proinflammatory, (B) anti-inflammatory, and (C) T cell markers analyzed by RT-real time PCR. n = 3–4 uninfected mice and n = 6–7 infected mice per genotype. Each symbol is a different mouse. All values are reported as mean ± SEM. Statistical analyses, where shown; *P < 0.05 and **P < 0.01 determined by 1-way ANOVA with Tukey post hoc test or Kruskal–Wallis test, followed by Mann–Whitney U tests.
Thus, we next assessed the population of immune cells present in the colon via immunohistochemistry and quantification of positively-stained cells. Consistent with the gene expression we observed, the CD68+ macrophages in the colon were increased with infection in Tln1fl/fl tissues, but were significantly less abundant in infected Tln1Δmye mice (Fig. 4A). As expected during infection, there were increases in the number of Ly6G+ neutrophils and CD11c+ dendritic cells, but there were no differences between Tln1fl/fl and Tln1Δmye mice (Fig. 4B,C). The Lyz2-flox system is used to target specific gene deletions in cells of the myeloid cell lineage and affects macrophages, neutrophils, and dendritic cells to varying degrees14 suggesting that talin-1 is important for the recruitment of macrophages, but not dendritic cells or neutrophils during C. rodentium infection. In addition, the number of T cells, marked by CD3+, were similar in Tln1Δmye mice compared to Tln1fl/fl mice with infection (Fig. 4D), matching the comparable expression of Il17a and Il22.
Myeloid cell talin-1 contributes to macrophage recruitment in response to infection in the mouse colon. Representative immunohistochemistry images and the quantification of positive cells per mm2 of Swiss-rolled colon tissues immunoperoxidase-stained for (A) CD68, (B) Ly6G, (C) CD11c, and (D) CD3. n = 4 uninfected mice and n = 5 infected mice per genotype. All values are reported as mean ± SEM. Statistical analyses, where shown; *P < 0.05, **P < 0.01, and ****P < 0.0001 determined by 1-way ANOVA and Tukey post hoc test. Scale bars represent 100 μm.
The number of differentiated macrophages in the gut is decreased in Tln1 Δmye mice
The reduced expression of macrophage markers in the colon of infected of Tln1Δmye mice (see Figs. 3 and 4) suggests that less macrophages were present in the mucosa. To test this postulate, we isolated immune cells from the whole colon of uninfected and infected Tln1fl/fl and Tln1Δmye mice and assessed monocyte-macrophages by flow cytometry using previously defined parameters15. We identified three subsets of CD45+CD11b+ macrophages based on their expression of Ly6C and MHCII15. The waterfall phenotype, which is a feature of macrophage differentiation in the mucosa, was observed in the uninfected and infected Tln1fl/fl and Tln1Δmye mice (Fig. 5A). While monocyte Population 1 (P1; Ly6C+MHCII– cells) and Population 2 (P2; Ly6C+MHCII+ cells) were not significantly different between the infected groups (Fig. 5A; Supplementary Fig. S3), there was a significant decrease in Population 3/4 (P3/4; Ly6C–MHCII+ cells), which correspond to differentiated macrophages, in the infected Tln1Δmye mice compared to the infected Tln1fl/fl mice (Fig. 5A,B).
Tln1fl/fl mice exhibit increased macrophage differentiation in the infected colon compared to Tln1Δmye mice. Cells isolated from the colonic lamina propria of Tln1fl/fl and Tln1Δmye mice infected with C. rodentium or mock, were assessed by flow cytometry for P1, P2, and P3/4 differentiation makers (A). Data shown are the concatenated results from all mice in the experiment. The frequency of Ly6C–MHCII+ macrophages (P3/4) is depicted in (B). n = 2–3 uninfected mice and n = 5–7 infected mice per genotype. All values are reported as mean ± SEM. Statistical analyses, *P < 0.05 determined by 1-way ANOVA with a Kruskal–Wallis test, followed by Mann–Whitney U tests.
Loss of talin-1 does not have a cell-intrinsic effect on macrophage activation and function ex vivo
We also tested whether talin-1 has a direct role in macrophage activation and function, independent of the colonic mucosal niche. To study talin-1 in macrophages ex vivo, we generated bone marrow-derived macrophages (BMmacs) and confirmed CD11b expression, a β2 integrin that is a marker of macrophages. Tln1-deficient BMmacs expressed a modest 6% increase of CD11b compared to Tln1fl/fl BMmacs with and without infection (Supplementary Fig. S4). This indicates that loss of talin-1 did not impede macrophage differentiation ex vivo. BMmacs derived from Tln1fl/fl and Tln1Δmye mice displayed similar levels of induced Tnf, Il1b, Il6, and Il23a transcripts after infection with C. rodentium (Fig. 6A). In addition, Tln1-deficient BMmacs retained the ability to produce nitric oxide (Fig. 6B), a commonly used marker to evaluate the activation and polarization of inflammatory macrophages16,17. We also assessed the effect of Tln1 deletion in bone marrow-derived dendritic cells (BMDCs; Supplementary Fig. S5A). We found that stimulation with C. rodentium similarly did not affect gene expression of Tnf, Il1b, Il6, or IL23a (Supplementary Fig. S5B), or levels of nitric oxide (Supplementary Fig. S5C). These data suggest that the decreased gene expression and inflammation that we observed in the tissues of Tln1Δmye mice during C. rodentium infection is not due to impaired capacity for macrophage or dendritic cell activation.
Talin-1 does not contribute to macrophage activation or phagocytic ability. BMmacs derived from Tln1fl/fl and Tln1Δmye mice were infected or not with C. rodentium, n = 3–4 mice per genotype. (A) Expression of pro-inflammatory macrophage markers at 6 h post-infection. (B) The concentration of NO2– in cell supernatants 24 h post-infection measured by the Griess reaction. (C) The amount of C. rodentium phagocytosed by Tln1fl/fl and Tln1Δmye BMmacs 1 h post-infection determined by gentamicin assay. (D-E) Representative flow plots and graphs depicting the surface expression and mean fluorescence intensity (MFI) of (D) CD86 and (E) MHCII on BMmacs 24 h post-infection. All values are reported as mean ± SEM. Statistical analyses, where shown; **P < 0.01, ***P < 0.001, and ****P < 0.0001 determined by 1-way ANOVA and Tukey post hoc test.
To further assess the role of talin-1 in the function of macrophages, we accessed phagocytosis and antigen presentation. We found that Tln1-deficient BMmacs engulfed the same number of C. rodentium as Tln1fl/fl BMmacs (Fig. 6C), signifying that talin-1 is not required for the phagocytosis of C. rodentium by macrophages. We next determined if talin-1 might have a role in the ability of macrophages to present antigens. We therefore measured the surface expression of MHCII and the co-stimulatory molecule, CD86. While the percentage of CD11b+ cells were not different between genotypes, we found that CD11b+ Tln1-deficient BMmacs expressed higher levels of CD86 (Fig. 6D; Supplementary Fig. S6), but not MHCII (Fig. 6E; Supplementary Fig. S6), compared to Tln1fl/fl BMmacs with infection. These data suggest that talin-1 is not required for the expression of markers of antigen-presenting cells.
Discussion
Talin-1 is a large cytoplasmic protein that creates a mechanical link between the actin cytoskeleton and integrins. Previous studies have demonstrated that talin-1 is required for the migration and recruitment of leukocytes, including neutrophils, dendritic cells, and T cells to sites of inflammation18,19,20. In general, migration of monocytes into tissues has been linked to the involvement of integrins. Specific to our findings, talin-1 has been directly linked to monocyte adhesive events 21,22. Using mice with myeloid-specific knockdown of Tln1, we demonstrate that talin-1 is essential in regulating the number of macrophages in the colonic mucosa during C. rodentium infection. Talin-1 did not have a cell-intrinsic role in macrophages as the loss of Tln1 did not affect gene expression, NO production, or phagocytosis of C. rodentium ex vivo. Therefore, considering the prior literature3,4,5,21,23 and our results, we conclude that the most likely explanation for the reduced inflammation that we observe is due to impaired tissue migration of monocytes, which develop into macrophages in the colonic mucosa. Collectively, our data demonstrate that loss of macrophage talin-1 is protective in the C. rodentium model of infectious colitis through a non-cell-autonomous manner and suggest that talin-1 is a promising target to limit the recruitment of macrophages during infectious colitis.
When infected with the mouse pathogen C. rodentium, mice that lacked expression of Tln1 in myeloid cells exhibited lower histologic injury scores and a decrease in both proinflammatory and anti-inflammatory gene expression in the colonic tissue. The decrease in histologic injury was a result of decreases in both epithelial injury and inflammation, but there was no change in the level of C. rodentium burden in the colon. It has been previously shown that mice with a weakened response of neutrophils and/or macrophages, but have an intact lymphoid cell compartment, exhibit decreased colitis and epithelial damage, yet can still control bacterial growth24,25. In contrast, immunocompromised mice or mice that lack an effective type 3 T cell or innate lymphoid cell response are unable to control the bacterial burden and exhibit higher rates of mortality26,27,28.
We observed that Tln1Δmye mice displayed decreased numbers of CD68+ cells in the mucosa while maintaining similar numbers of neutrophils, dendritic cells, and T cells as marked by Ly6G, CD11c, and CD3, respectively, to their genetically normal littermates. Furthermore, the monocyte-macrophage waterfall assay showed less differentiated macrophages in the infected Tln1Δmye mice compared to Tln1fl/fl animals, suggesting that fewer monocytes were infiltrating into the lamina propria at the beginning of the infection. This reduction in the number of macrophages in the colon of Tln1Δmye mice could be due to different events, including lower recruitment to the site of infection or migration through the endothelium to the lamina propria, or increased cell death in the mucosa. In addition, the decrease in transcript expression that we observed in Tln1Δmye mice are from genes that are normally associated with proinflammatory and anti-inflammatory cells of the innate immune response. A decrease in proinflammatory cytokines creates a feedback loop that then results in less macrophage activation, and less inflammation and tissue damage. These data suggest that the decrease of expression of these genes may be due to the decreased number of macrophages and not due to polarization state. Supporting this concept, we found that BMmacs and BMDCs from Tln1fl/fl and Tln1Δmye mice are similarly activated ex vivo by C. rodentium. It should also be noted that some heterogeneity in cell populations can be observed after ex vivo stimulation, such as the presence of macrophages in GM-CSF-treated bone marrow cells29. Our data suggest that talin-1 is not involved in the process of macrophage stimulation. However, further experiments, including transfer of macrophages from Tln1fl/fl and Tln1Δmye mice into Ccr2-deficient animals, which are characterized by an empty intestinal macrophage niche30, or by mixed bone marrow chimera experiments with cells from both genotypes, could be utilized to support the conclusion that talin-1 plays a role in the recruitment of macrophages in the intestine. We demonstrate no differences in the mRNA levels of Il22 and Il17 which are often expressed by Th22 and Th17 cells in the colon, respectively, and are important for controlling bacterial growth, but not disease development during enteropathogenic infection27,31,32.
Integrins are a vital component of innate leukocytes and are involved in bidirectional signaling across the membrane. We therefore were interested in whether the decrease in colitis results from decreased macrophage function. Talin-1 has also been implicated in the phagocytic ability of macrophages; however, this is dependent on the integrin β-subunit and the target of the engulfment. Talin-1 is dispensable for integrin β5-mediated phagocytosis of apoptotic cells33, while talin-1 is required for the integrin β2-mediated phagocytosis of C3bi-opsinized red blood cells34. We observed that talin-1 was not necessary for the phagocytosis of C. rodentium by macrophages. These cells also have the dual responsibility of directly killing pathogens and acting as professional antigen presenting cells to activate and direct T cells35. Talin-1 has been shown to be involved in T cell activation in an antigen presenting cell contact-dependent manner through stabilization of the immune synapse36. Lim et al. demonstrate that Tln1-deficient dendritic cells using the Cd11c-cre expressed lower levels of CD86 and MHCII when stimulated with LPS that was also associated with decreased gene expression of proinflammatory markers20. However, we show using a different Cre driver and stimuli that there were no differences in induced expression of proinflammatory markers in response to C. rodentium by dendritic cells or macrophages. We did not observe any differences in the ability of Tln1-deficient macrophages to express MHCII but show that Tln1-deficient macrophages express modestly higher surface levels of CD86 and the β2 integrin CD11b, which can be used to identify inflammatory macrophages in the lung during acute inflammation37. In addition, myeloid-derived talin-1 does not contribute to DSS colitis. These data indicate that talin-1 is not consistently essential for macrophage or dendritic cell differentiation or function, but our other data show that talin-1 is important for the presence of differentiated macrophages in the colonic mucosa during challenge with pathogenic bacteria.
In summary, the results from this study indicate that myeloid talin-1 affects the number of differentiated macrophages in the colon in the context of pathogenic colitis. Our data indicate that the expression of talin-1 by macrophages contributes to C. rodentium-induced colonic inflammation, with the increased number of colonic macrophages leading to more generation of inflammatory mediators, while failing to help eradicate the pathogen. These findings provide further insight into the development of bacteria-induced colitis and highlights a potential target to control macrophage movement into tissues that leads to the propagation of inflammation.
Materials and methods
Mice
C57BL/6 Tln1fl/fl mice provided by Dr. Roy Zent at Vanderbilt University Medical Center (Nashville, TN). These mice were crossed with C57BL/6 Lyz2cre/cre mice and the resulting offspring were crossed to generate Tln1fl/fl;Lyz2+/+ (Tln1fl/fl) and Tln1fl/fl;Lyz2cre/cre (Tln1Δmye) littermates. Animals were euthanized by exposure to carbon dioxide followed by cervical dislocation. All experiments were approved by the IACUC at Vanderbilt University and Institutional Biosafety Committee and the Research and Development Committee of the Veterans Affairs Tennessee Valley Healthcare System under the protocol V2000018 and performed in accordance with these institutional guidelines. The study was carried out in compliance with the ARRIVE guidelines.
Generation of bone marrow-derived immune cells
BMmacs and BMDCs were generated as described17. Briefly, cells were flushed from the femur and tibia of naïve mice, and RBCs were lysed prior to counting and plating using ACK Lysing Buffer (Thermo Fisher). Cells were differentiated for 7 days in complete medium (DMEM media supplemented with 10% FBS, 100 U/ml penicillin/streptomycin, 25 mM HEPES, and 20 ng/ml recombinant M-CSF (PeproTech). BMDCs were differentiated for 8 days in complete medium (DMEM media supplemented with 10% FBS, 100 U/ml penicillin/streptomycin, 25 mM HEPES, and 10 ng/ml recombinant GM-CSF (PeproTech).
Infection with C. rodentium
Male adult (6–12 wk) littermates were orally inoculated by gavage with a sublethal dose, 5 × 108, C. rodentium strain DBS100 as previously described8,9,10. Control mice received sterile Luria–Bertani broth alone. Animals were monitored and weighed daily for 14 days. Mice were sacrificed, and the colons were removed, measured, cleaned, weighed, and Swiss-rolled for fixation in 10% neutral buffered formalin for histology. Prior to fixation, three proximal and distal pieces were collected; two pieces were flash frozen and the third was weighed, homogenized, serially diluted, and cultured on McConkey agar plates to determine bacterial colonization by counting the CFUs. Hematoxylin and eosin (H&E) stained slides were scored by a gastrointestinal pathologist (M.B.P.), blinded to the mouse genotype and infection status, for histologic injury (0–21), a composite of the total inflammation (0–18) plus epithelial injury (0–3) with 0: no injury; 0.5: mucus depletion 1: erosion (mild to moderate); 2: superficial ulcers or extensive erosion; 3: deep ulcers and/or necrosis of the mucosa8,9,10. Total inflammation is the combination of acute inflammation (0–3) based on polymorphonuclear leukocytes, plus chronic inflammation (0–3) based on mononuclear cell infiltrates and multiplied by the depth of inflammation (0–3) with 0: no inflammation; 1: mucosa; 2: submucosa; 3: muscular propria or beyond.
BMmacs and BMDCs were infected with C. rodentium at a multiplicity of infection of 10 for 3 h in antibiotic-free media. The cells were then washed and provided media containing penicillin and streptomycin for an additional 3 h (mRNA) or 21 h (protein, flow cytometry).
DSS-induced model of colitis
Male adult littermates were treated with 4% DSS in the drinking water for 5 days after which the DSS was removed, and the mice remained on regular water for an additional 5 days prior to euthanasia9,13. Mice were sacrificed, and the colons were removed, measured, cleaned, weighed, and Swiss-rolled for fixation in 10% neutral buffered formalin for histology. Hematoxylin and eosin (H&E) stained slides were scored by a gastrointestinal pathologist (M.K.W.), blinded to the mouse genotype and treatment status, for histologic injury (0–40), a composite of the total inflammation (0–24) plus crypt damage (0–16). Total inflammation is a combination of inflammation (0–3) plus depth of inflammation (0–3) with 0: no inflammation; 1: mucosa; 2: submucosa; 3: transmural multiplied but the percent of involved area (1–4) with 1: ≤ 25%; 2: 26–50%; 3: 51–75%; 4: > 75%. Crypt damage is a combination of the amount of damage (1–4) with 1: basal 1/3 damaged; 2: basal 2/3 damaged; 3: only surface intact; 4: entire crypt and surface lost plus the percent of the tissue involved by crypt damage with 1: 10–25%; 2: 26–50%; 3:56–75%; 4: > 75%.
mRNA analysis
Total RNA was isolated from cells and the flash frozen colonic tissues using the RNeasy Mini Kit (QIAGEN). Total RNA samples were reverse transcribed into cDNA using the SuperScript III Reverse Transcriptase (Thermo Fisher), Oligo (dT) primers (Thermo Fisher), and dNTP Mix (Applied Biosystems). Real-time PCR was performed using the PowerUp SYBR Green Master Mix (Applied Biosystems). The primers are listed in Supplementary Table S1.
Western blot and densitometric analysis
Protein isolation, SDS-PAGE separation, nitrocellulose membrane transfer, and band visualization were performed as described17. The membranes were incubated with a rabbit anti-talin-1 (Cell Signaling), or a mouse anti-β-actin (Sigma) followed by HRP-labeled goat anti-rabbit IgG (Jackson ImmunoResearch) or HRP-labeled goat anti-mouse IgG (Promega), respectively. Densitometric analysis was performed with Fiji (ImageJ)38.
Immunohistochemistry
Paraffin-embedded Swiss-rolled murine colon tissues were processed as previously described17 overnight at 4 °C using the following antibodies: anti-CD68 (pre-diluted, Biocare Medical), anti-Ly6G (1:1000; Abcam), anti-CD11c (Cell Signaling), or anti-CD3 (1:150; Abcam). All slides were imaged and analyzed using a Cytation C10 Confocal Imaging Reader and Gen 5+ software (Agilent BioTek). The average number of CD68-, Ly6G-, Cd11c-, and CD3-positive cells was quantified by the Cell Analysis function of the Gen 5+ software and was limited to the mucosa to reduce inclusion of non-specific staining and normalized to tissue surface area.
Measurement of NO2 –
NO2− concentrations were assessed using the standard Griess reaction (Promega) at 24 h post-infection with C. rodentium.
Gentamicin assay
BMmacs were infected at a multiplicity of infection of 10 for 1 h prior to washing, counting, and treating with 200 g/ml gentamicin for 1 h39. The cells were then lysed with 0.1% saponin for 30 min at 37 °C and the lysate was serially diluted and cultured on McConkey agar plates. CFUs were counted and normalized to cell count.
Colonic lamina propria isolation
Isolation of the colonic lamina propria was performed as previously described40. Briefly, colons were removed from the mice, cut longitudinally, washed with cold PBS, cut into 5 mm pieces, and incubated in 25 ml of pre-warmed RPMI 1640 media with 5% heat-inactivated FBS, 5 mM EDTA, 1 mM dithiothreitol (Thermo Fisher Scientific), and 25 mM HEPES at 37 °C for 40 min in a non-CO2 MaxQ4450 horizontal shaker (Thermo Fisher Scientific). The media was strained through a sieve (Everyday Living) and the intestinal pieces were placed into 25 ml cold RMPI media with 2 mM EDTA and 25 mM HEPES. The tubes containing the tissues were shaken 20 times and strained. The intestinal pieces were minced and placed into 25 ml pre-warmed RPMI 1640 media containing 0.1 mg/ml Liberase TL (Roche), 0.05% DNAse I (Sigma-Aldrich), and 25 mM HEPES and shaken at 37 °C for 30 min. The cell/media mixture was pulled through a 10 ml syringe 20 times and filtered through a 70 μm cell strainer into an equal volume of cold RPMI 1640 media containing 5% heat-inactivated FBS, 0.05% DNAse I, 25 mM HEPES on ice. Cells were spun for 10 min at 4 °C and 475 × g and resuspended in 40% Percoll (Sigma-Aldrich) solution and underlaid using 90% Percoll. The 40/90 gradient was spun for 25 min at 20 °C at 475 × g with no brake or acceleration applied, resulting in an interphase layer. The interphase layer was recovered using a 1 ml pipet and then washed in fluorescence activated cell sorting (FACS) buffer (PBS with 2% FBS and 2 mm EDTA) and spun again for 10 min at 20 °C and 475 × g before staining. The yield of lamina propria cells per colon was 738,612 ± 189,455 (n = 5) in uninfected mice and 267,929 ± 75,895 (n = 12) in C. rodentium-infected mice.
Monocyte-macrophage waterfall assay
Cells isolated from the colonic lamina propria were blocked with TruStain FcXtm (Biolegend) for 5 min at 4 °C in the dark and then incubated in the antibody cocktail (Supplementary Table S2) in PBS for 15 min at 4 °C in the dark. Cells were gated on live single CD45+CD11b+Lineageneg. Lineage markers used were CD11c, CD3, CD19, NK1.1, and Ly6G. A live/dead stain (Thermo Fisher Scientific) was also used to restrict analysis to live cells. Data were acquired using a Cytek® Aurora CS and flow cytometric analysis was performed using FlowJo (BD Biosciences).
Immunophenotyping by flow cytometry
BMmacs infected or not with C. rodentium for 24 h were washed with PBS and fixed with CytoFix (BD Biosciences) for 20 min at 4 °C, washed, and then labeled with anti-CD11b (1:200; BD Biosciences), anti-CD86 (1:200; Invitrogen), and anti-MHCII (1:200; Invitrogen).
Statistics
All the data shown represent the mean ± SEM. C. rodentium infection data is pooled from two independent experiments. A minimum of three biological replicates were used for ex vivo studies. GraphPad Prism 9.4 (GraphPad Software) was used to perform statistical analyses and significance was set at P < 0.05. For normally distributed data, a 2-tailed Student’s t test or a 1-way ANOVA with the Tukey post hoc test were performed to compare differences between two or more test groups, respectively. Non-normally distributed data was analyzed by a 1-way ANOVA with the Kruskal–Wallis test followed by a Mann–Whitney U test, or square root transformed and reassessed.
Data availability
The data of this study are available from the corresponding author upon reasonable request.
References
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Lebeis, S. L., Sherman, M. A. & Kalman, D. Protective and destructive innate immune responses to enteropathogenic Escherichia coli and related A/E pathogens. Future Microbiol. 3, 315–328 (2008).
Weerasinghe, D. et al. A role for the alphavbeta3 integrin in the transmigration of monocytes. J. Cell Biol. 142, 595–607 (1998).
Jin, H., Su, J., Garmy-Susini, B., Kleeman, J. & Varner, J. Integrin alpha4beta1 promotes monocyte trafficking and angiogenesis in tumors. Cancer Res. 66, 2146–2152 (2006).
Schittenhelm, L., Hilkens, C. M. & Morrison, V. L. b2 integrins as regulators of dendritic cell, monocyte, and macrophage function. Front. Immunol. 8, 1866 (2017).
Calderwood, D. A. et al. The Talin head domain binds to integrin beta subunit cytoplasmic tails and regulates integrin activation. J. Biol. Chem. 274, 28071–28074 (1999).
Critchley, D. R. Biochemical and structural properties of the integrin-associated cytoskeletal protein talin. Annu. Rev. Biophys. 38, 235–254 (2009).
Singh, K. et al. The L-arginine transporter solute carrier family 7 member 2 mediates the immunopathogenesis of attaching and effacing bacteria. PLoS Pathog. 12, e1005984 (2016).
Gobert, A. P. et al. Distinct immunomodulatory effects of spermine oxidase in colitis induced by epithelial injury or infection. Front. Immunol. 9, 1242 (2018).
Gobert, A. P. et al. Hypusination orchestrates the antimicrobial response of macrophages. Cell Rep. 33, 108510 (2020).
Luperchio, S. A. & Schauer, D. B. Molecular pathogenesis of Citrobacter rodentium and transmissible murine colonic hyperplasia. Microbes Infect. 3, 333–340 (2001).
Collins, J. W. et al. Citrobacter rodentium: infection, inflammation and the microbiota. Nat. Rev. Microbiol. 12, 612–623 (2014).
Gobert, A. P. et al. Protective role of spermidine in colitis and colon carcinogenesis. Gastroenterology 162, 813–827 (2022).
Clausen, B. E., Burkhardt, C., Reith, W., Renkawitz, R. & Forster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 8, 265–277 (1999).
Tamoutounour, S. et al. CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur. J. Immunol. 42, 3150–3166 (2012).
Hardbower, D. M. et al. Arginase 2 deletion leads to enhanced M1 macrophage activation and upregulated polyamine metabolism in response to Helicobacter pylori infection. Amino Acids 48, 2375–2388 (2016).
Latour, Y. L. et al. Cystathionine gamma-lyase exacerbates Helicobacter pylori immunopathogenesis by promoting macrophage metabolic remodeling and activation. JCI Insight 7, e155338 (2022).
Lammermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Lim, T. J. F. & Su, I. H. Talin1 methylation is required for neutrophil infiltration and lipopolysaccharide-induced lethality. J. Immunol. 201, 3651–3661 (2018).
Lim, T. J. F., Bunjamin, M., Ruedl, C. & Su, I. H. Talin1 controls dendritic cell activation by regulating TLR complex assembly and signaling. J. Exp. Med. 217 (2020).
Hyduk, S. J. et al. Talin-1 and kindlin-3 regulate alpha4beta1 integrin-mediated adhesion stabilization, but not G protein-coupled receptor-induced affinity upregulation. J. Immunol. 187, 4360–4368 (2011).
Wen, L., Moser, M. & Ley, K. Molecular mechanisms of leukocyte b2 integrin activation. Blood 139, 3480–3492 (2022).
Lefort, C. T. et al. Distinct roles for talin-1 and kindlin-3 in LFA-1 extension and affinity regulation. Blood 119, 4275–4282 (2012).
Khan, M. A. et al. Toll-like receptor 4 contributes to colitis development but not to host defense during Citrobacter rodentium infection in mice. Infect. Immun. 74, 2522–2536 (2006).
Gibson, D. L. et al. Toll-like receptor 2 plays a critical role in maintaining mucosal integrity during Citrobacter rodentium-induced colitis. Cell Microbiol. 10, 388–403 (2008).
Vallance, B. A., Deng, W., Knodler, L. A. & Finlay, B. B. Mice lacking T and B lymphocytes develop transient colitis and crypt hyperplasia yet suffer impaired bacterial clearance during Citrobacter rodentium infection. Infect. Immun. 70, 2070–2081 (2002).
Zheng, Y. et al. Interleukin-22 mediates early host defense against attaching and effacing bacterial pathogens. Nat. Med. 14, 282–289 (2008).
Chan, J. M. et al. CD4+ T cells drive goblet cell depletion during Citrobacter rodentium infection. Infect. Immun. 81, 4649–4658 (2013).
Helft, J. et al. GM-CSF mouse bone marrow cultures comprise a heterogeneous population of CD11c(+)MHCII(+) macrophages and dendritic cells. Immunity. 42, 1197–1211 (2015).
Desalegn, G. & Pabst, O. Inflammation triggers immediate rather than progressive changes in monocyte differentiation in the small intestine. Nat. Commun. 10, 3229 (2019).
Basu, R. et al. Th22 cells are an important source of IL-22 for host protection against enteropathogenic bacteria. Immunity 37, 1061–1075 (2012).
Zindl, C. L. et al. A nonredundant role for T cell-derived interleukin 22 in antibacterial defense of colonic crypts. Immunity 55, 494–511 (2022).
Singh, S., Dmello, V., van Bergen en Henegouwen, P. & Birge, R. B. A NPxY-independent beta5 integrin activation signal regulates phagocytosis of apoptotic cells. Biochem. Biophys. Res. Commun. 364, 540–548 (2007).
Lim, J. et al. An essential role for talin during alpha(M)beta(2)-mediated phagocytosis. Mol. Biol. Cell 18, 976–985 (2007).
Mosser, D. M. & Edwards, J. P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 8, 958–969 (2008).
Wernimont, S. A. et al. Contact-dependent T cell activation and T cell stopping require talin1. J. Immunol. 187, 6256–6267 (2011).
Duan, M. et al. CD11b immunophenotyping identifies inflammatory profiles in the mouse and human lungs. Mucosal. Immunol. 9, 550–563 (2016).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Gobert, A. P. et al. Bacterial pathogens hijack the innate immune response by activation of the reverse transsulfuration pathway. mBio 10, e02174-e12119 (2019).
Latour, Y. L. et al. Epithelial talin-1 protects mice from Citrobacter rodentium-induced colitis by restricting bacterial crypt intrusion and enhancing T cell immunity. Gut Microbes 15, 2192623 (2023).
Acknowledgements
We kindly thank Dr. Roy Zent for providing the C57BL/6 Tln1fl/fl mice.
Funding
This work was supported by: NIH Grants R01DK128200, P01CA028842, P01CA116087, U.S. Department of Veterans Affairs Merit Review Grants I01CX002171 and I01CX002473, U.S. Department of Defense Grant W81XWH-21-1-0617, Crohn’s & Colitis Foundation Senior Research Award 703003, a gift from Cure for IBD, the James Rowen Fund, and the Thomas F. Frist Sr. Endowment (all to K.T.W.) and core services from the Vanderbilt University Medical Center Digestive Disease Research Center funded by NIH Grant P30DK058404. YLL was supported by NIH Grant T32AI138932 and KMM was supported by NIH Grants T32CA009592 and F31CA278330.
Author information
Authors and Affiliations
Contributions
Y.L.L., A.P.G., and K.T.W. conceived of the study; Y.L.L., K.M.M., M.M.A., D.P.B., T.M.S., M.A., K.J.W., C.V.H., J.J., J.A.G., A.G.D., M.B.P., and M.K.W. conducted the investigation; Y.L.L. and K.M.M. conducted the formal analysis; Y.L.L. contributed to visualization during the studies and wrote the original draft of the manuscript; Y.L.L., K.M.M., A.P.G., and K.T.W. reviewed and edited the manuscript; A.P.G. and K.T.W. supervised conducting of the study; and K.T.W. acquired funding for the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Latour, Y.L., McNamara, K.M., Allaman, M.M. et al. Myeloid deletion of talin-1 reduces mucosal macrophages and protects mice from colonic inflammation. Sci Rep 13, 22368 (2023). https://doi.org/10.1038/s41598-023-49614-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-49614-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.