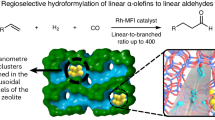
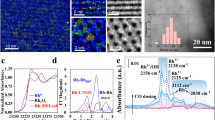
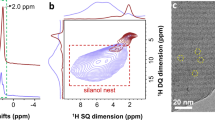
Abstract
Hydroformylation is an industrial process for the production of aldehydes from alkenes1,2. Regioselective hydroformylation of propene to high-value n-butanal is particularly important, owing to a wide range of bulk applications of n-butanal in the manufacture of various necessities in human daily life3. Supported rhodium (Rh) hydroformylation catalysts, which often excel in catalyst recyclability, ease of separation and adaptability for continuous-flow processes, have been greatly exploited4. Nonetheless, they usually consist of rotationally flexible and sterically unconstrained Rh hydride dicarbonyl centres, only affording limited regioselectivity to n-butanal5,6,7,8. Here we show that proper encapsulation of Rh species comprising Rh(I)-gem-dicarbonyl centres within a MEL zeolite framework allows the breaking of the above model. The optimized catalyst exhibits more than 99% regioselectivity to n-butanal and more than 99% selectivity to aldehydes at a product formation turnover frequency (TOF) of 6,500 h−1, surpassing the performance of all heterogeneous and most homogeneous catalysts developed so far. Our comprehensive studies show that the zeolite framework can act as a scaffold to steer the reaction pathway of the intermediates confined in the space between the zeolite framework and Rh centres towards the exclusive formation of n-butanal.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that all the data supporting the findings of this study are available within the paper and its Supplementary Information files and are also available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Pospech, J., Fleischer, I., Franke, R., Buchholz, S. & Beller, M. Alternative metals for homogeneous catalyzed hydroformylation reactions. Angew. Chem. Int. Edn 52, 2852–2872 (2013).
Arnoldy, P. in Rhodium Catalyzed Hydroformylation (Van Leeuwen, P. W. N. M. & Claver, C.) 203–231 (Springer, 2000).
Franke, R., Selent, D. & Borner, A. Applied hydroformylation. Chem. Rev. 112, 5675–5732 (2012).
Liu, B. et al. Heterogeneous hydroformylation of alkenes by Rh-based catalysts. Chem 8, 2630–2658 (2022).
Shylesh, S. et al. In situ formation of Wilkinson-type hydroformylation catalysts: insights into the structure, stability, and kinetics of triphenylphosphine- and xantphos-modified Rh/SiO2. ACS Catal. 3, 348–357 (2013).
Li, C. et al. Single atom dispersed Rh-biphephos&PPh3@porous organic copolymers: highly efficient catalysts for continuous fixed-bed hydroformylation of propene. Green Chem. 18, 2995–3005 (2016).
Riisager, A. et al. Very stable and highly regioselective supported ionic‐liquid‐phase (SILP) catalysis: continuous‐flow fixed‐bed hydroformylation of propene. Angew. Chem. Int. Edn 44, 815–819 (2005).
Srinivas, G. & Chuang, S. S. C. An in-situ infrared study of the formation of n- and iso-butyraldehyde from propylene hydroformylation on Rh/SiO2 and sulfided Rh/SiO2. J. Catal. 144, 131–147 (1993).
Smit, B. & Maesen, T. L. Towards a molecular understanding of shape selectivity. Nature 451, 671–678 (2008).
Ren, L. et al. Designed copper-amine complex as an efficient template for one-pot synthesis of Cu-SSZ-13 zeolite with excellent activity for selective catalytic reduction of NOx by NH3. Chem. Commun. 47, 9789–9791 (2011).
McClure, S. M., Lundwall, M. J. & Goodman, D. W. Planar oxide supported rhodium nanoparticles as model catalysts. Proc. Natl Acad. Sci. USA 108, 931–936 (2011).
Ro, I. et al. Bifunctional hydroformylation on heterogeneous Rh-WOx pair site catalysts. Nature 609, 287–292 (2022).
Yates, D. J. C., Murrell, L. L. & Prestridge, E. B. Ultradispersed rhodium rafts: their existence and topology. J. Catal. 57, 41–63 (1979).
Qi, L. et al. Ethene hydroformylation catalyzed by rhodium dispersed with zinc or cobalt in silanol nests of dealuminated zeolite beta. J. Am. Chem. Soc. 145, 2911–2929 (2023).
Matsubu, J. C., Yang, V. N. & Christopher, P. Isolated metal active site concentration and stability control catalytic CO2 reduction selectivity. J. Am. Chem. Soc. 137, 3076–3084 (2015).
Goellner, J. F., Gates, B. C., Vayssilov, G. N. & Rösch, N. Structure and bonding of a site-isolated transition metal complex: rhodium dicarbonyl in highly dealuminated zeolite Y. J. Am. Chem. Soc. 122, 8056–8066 (2000).
Choi, M., Wu, Z. & Iglesia, E. Mercaptosilane-assisted synthesis of metal clusters within zeolites and catalytic consequences of encapsulation. J. Am. Chem. Soc. 132, 9129–9137 (2010).
Goel, S., Wu, Z., Zones, S. I. & Iglesia, E. Synthesis and catalytic properties of metal clusters encapsulated within small-pore (SOD, GIS, ANA) zeolites. J. Am. Chem. Soc. 134, 17688–17695 (2012).
Li, C. et al. Xantphos doped Rh/POPs-PPh3 catalyst for highly selective long-chain olefins hydroformylation: chemical and DFT insights into Rh location and the roles of Xantphos and PPh3. J. Catal. 353, 123–132 (2017).
Pruett, R. L. & Smith, J. A. Low-pressure system for producing normal aldehydes by hydroformylation of α-olefins. J. Org. Chem. 34, 327–330 (1969).
Zheng, Y. et al. Boosting the hydroformylation activity of a Rh/CeO2 single-atom catalyst by tuning surface deficiencies. ACS Catal. 13, 7243–7255 (2023).
Farpón, M. G. et al. Rhodium single-atom catalyst design through oxide support modulation for selective gas-phase ethylene hydroformylation. Angew. Chem. Int. Edn 135, e202214048 (2022).
Aireddy, D. R. & Ding, K. Heterolytic dissociation of H2 in heterogeneous catalysis. ACS Catal. 12, 4707–4723 (2022).
Lu, J., Aydin, C., Browning, N. D. & Gates, B. C. Hydrogen activation and metal hydride formation trigger cluster formation from supported iridium complexes. J. Am. Chem. Soc. 134, 5022–5025 (2012).
Torrent, M., Sola, M. & Frenking, G. Theoretical studies of some transition-metal-mediated reactions of industrial and synthetic importance. Chem. Rev. 100, 439–494 (2000).
Liu, L. et al. Regioselective generation and reactivity control of subnanometric platinum clusters in zeolites for high-temperature catalysis. Nat. Mater. 18, 866–873 (2019).
Sun, Q. et al. Zeolite‐encaged single‐atom rhodium catalysts: highly‐efficient hydrogen generation and shape‐selective tandem hydrogenation of nitroarenes. Angew. Chem. Int. Edn 58, 18570–18576 (2019).
Kaftan, A. et al. Supported homogeneous catalyst makes its own liquid phase. J. Catal. 321, 32–38 (2015).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Rice, C. A., Worley, S. D., Curtis, C. W., Guin, J. A. & Tarrer, A. R. The oxidation state of dispersed Rh on Al2O3. J. Chem. Phys. 74, 6487–6497 (1981).
Marinkovic, N. S., Sasaki, K. & Adzic, R. R. Determination of single- and multi-component nanoparticle sizes by X-ray absorption spectroscopy. J. Electrochem. Soc. 165, J3222–J3230 (2018).
Jentys, A. Estimation of mean size and shape of small metal particles by EXAFS. Phys. Chem. Chem. Phys. 1, 4059–4063 (1999).
Cao, L. et al. Atomically dispersed iron hydroxide anchored on Pt for preferential oxidation of CO in H2. Nature 565, 631–635 (2019).
Cho, H. J., Kim, D., Li, J., Su, D. & Xu, B. Zeolite-encapsulated Pt nanoparticles for tandem catalysis. J. Am. Chem. Soc. 140, 13514–13520 (2018).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104–154123 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comp. Chem. 32, 1456–1465 (2011).
Blochl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Teter, M. P., Payne, M. C. & Allan, D. C. Solution of Schrödinger’s equation for large systems. Phys. Rev. B 40, 12255–12263 (1989).
Bylander, D. M., Kleinman, L. & Lee, S. Self-consistent calculations of the energy bands and bonding properties of B12C3. Phys. Rev. B 42, 1394–1403 (1990).
Methfessel, M. & Paxton, A. T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40, 3616–3621 (1989).
Henkelman, G. & Jónsson, H. A dimer method for finding saddle points on high dimensional potential surfaces using only first derivatives. J. Chem. Phys. 111, 7010–7022 (1999).
Heyden, A., Bell, A. T. & Keil, F. J. Efficient methods for finding transition states in chemical reactions: comparison of improved dimer method and partitioned rational function optimization method. J. Chem. Phys. 123, 224101–224115 (2005).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Szécsényi, Á. et al. Breaking linear scaling relationships with secondary interactions in confined space: A case study of methane oxidation by Fe/ZSM-5 zeolite. ACS Catal. 9, 9276–9284 (2019).
Jacobs, I., de Bruin, B. & Reek, J. N. Comparison of the full catalytic cycle of hydroformylation mediated by mono‐ and bis‐ligated triphenylphosphine–rhodium complexes by using DFT calculations. ChemCatChem 7, 1708–1718 (2015).
Kozuch, S. & Shaik, S. How to conceptualize catalytic cycles? The energetic span model. Acc. Chem. Res. 44, 101–110 (2011).
Farkas, A., Hess, F. & Over, H. Experiment-based kinetic Monte Carlo simulations: CO oxidation over RuO2(110). J. Phys. Chem. C 116, 581–591 (2012).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Frisch, M. J. et al. Gaussian 16, Revision A.03 (Gaussian, Inc., 2016).
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. 38, 3098–3010 (1988).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Mehio, N., Dai, S. & Jiang, D. Quantum mechanical basis for kinetic diameters of small gaseous molecules. J. Phys. Chem. A 118, 1150–1154 (2014).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (nos. 21972161 and 22172186), Major Science and Technology Project of Ordos City (no. 2022EEDSKJZDZX001), Autonomous Research Project of State Key Laboratory of Coal Conversion (no. 2022BWZ009), Inner Mongolia Key Research and Development Program (no. 2023YFHH0009), Institute of Coal Chemistry, Chinese Academy of Sciences and Synfuels China Technology Co., Ltd. We also acknowledge the Shanghai Synchrotron Radiation Facility (SSRF) for providing the facility for XAS measurements and the Centre for High-resolution Electron Microscopy (CħEM) for the TEM tests.
Author information
Authors and Affiliations
Contributions
Z.C., Y.Y. and H.W. conceived the project and designed the study. X.Zha. and T.Y. conducted synthesis work, catalytic tests and other characterizations. J.Y. performed the theoretical calculations. H.H., F.S., Y.W. and Q.Z. conducted the structural characterizations. X.S., M.D., W.F., J.W., Y.S., X.Zho., K.W. and Y.L. helped with the data analysis. Z.C. and H.W. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–37 and Supplementary Tables 1–8.
Supplementary Data
The structural files and corresponding total energy of theoretical models.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Yan, T., Hou, H. et al. Regioselective hydroformylation of propene catalysed by rhodium-zeolite. Nature 629, 597–602 (2024). https://doi.org/10.1038/s41586-024-07342-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-024-07342-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.