Abstract
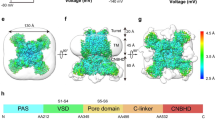
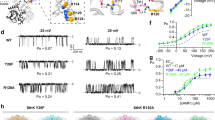
Proteins containing voltage-sensing domains (VSDs) translate changes in membrane potential into changes in ion permeability or enzymatic activity1,2,3. In channels, voltage change triggers a switch in conformation of the VSD, which drives gating in a separate pore domain, or, in channels lacking a pore domain, directly gates an ion pathway within the VSD4,5. Neither mechanism is well understood6. In the Shaker potassium channel, mutation of the first arginine residue of the S4 helix to a smaller uncharged residue makes the VSD permeable to ions (‘omega current’) in the resting conformation (‘S4 down’)7. Here we perform a structure-guided perturbation analysis of the omega conductance to map its VSD permeation pathway. We find that there are four omega pores per channel, which is consistent with one conduction path per VSD. Permeating ions from the extracellular medium enter the VSD at its peripheral junction with the pore domain, and then plunge into the core of the VSD in a curved conduction pathway. Our results provide a model of the resting conformation of the VSD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yu, F. H. & Catterall, W. A. The VGL-chanome: a protein superfamily specialized for electrical signaling and ionic homeostasis. Sci. STKE 2004, re15 (2004)
Bezanilla, F. The voltage sensor in voltage-dependent ion channels. Physiol. Rev. 80, 555–592 (2000)
Murata, Y., Iwasaki, H., Sasaki, M., Inaba, K. & Okamura, Y. Phosphoinositide phosphatase activity coupled to an intrinsic voltage sensor. Nature 435, 1239–1243 (2005)
Ramsey, I. S., Moran, M. M., Chong, J. A. & Clapham, D. E. A voltage-gated proton-selective channel lacking the pore domain. Nature 440, 1213–1216 (2006)
Sasaki, M., Takagi, M. & Okamura, Y. A voltage sensor-domain protein is a voltage-gated proton channel. Science 312, 589–592 (2006)
Tombola, F., Pathak, M. M. & Isacoff, E. Y. How does voltage open an ion channel? Annu. Rev. Cell Dev. Biol. 22, 23–52 (2006)
Tombola, F., Pathak, M. M. & Isacoff, E. Y. Voltage-sensing arginines in a potassium channel permeate and occlude cation-selective pores. Neuron 45, 379–388 (2005)
Long, S. B., Campbell, E. B. & Mackinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309, 897–903 (2005)
Sigworth, F. J. The variance of sodium current fluctuations at the node of Ranvier. J. Physiol. (Lond.) 307, 97–129 (1980)
Gandhi, C. S., Clark, E., Loots, E., Pralle, A. & Isacoff, E. Y. The orientation and molecular movement of a K+ channel voltage-sensing domain. Neuron 40, 515–525 (2003)
Larsson, H. P., Baker, O. S., Dhillon, D. S. & Isacoff, E. Y. Transmembrane movement of the shaker K+ channel S4. Neuron 16, 387–397 (1996)
Yang, N., George, A. L. & Horn, R. Molecular basis of charge movement in voltage-gated sodium channels. Neuron 16, 113–122 (1996)
Yusaf, S. P., Wray, D. & Sivaprasadarao, A. Measurement of the movement of the S4 segment during the activation of a voltage-gated potassium channel. Pflugers Arch. 433, 91–97 (1996)
Ruta, V., Chen, J. & MacKinnon, R. Calibrated measurement of gating-charge arginine displacement in the KvAP voltage-dependent K+ channel. Cell 123, 463–475 (2005)
Cha, A., Snyder, G. E., Selvin, P. R. & Bezanilla, F. Atomic scale movement of the voltage-sensing region in a potassium channel measured via spectroscopy. Nature 402, 809–813 (1999)
Glauner, K. S., Mannuzzu, L. M., Gandhi, C. S. & Isacoff, E. Y. Spectroscopic mapping of voltage sensor movement in the Shaker potassium channel. Nature 402, 813–817 (1999)
Durell, S. R., Shrivastava, I. H. & Guy, H. R. Models of the structure and voltage-gating mechanism of the shaker K+ channel. Biophys. J. 87, 2116–2130 (2004)
Yarov-Yarovoy, V., Baker, D. & Catterall, W. A. Voltage sensor conformations in the open and closed states in ROSETTA structural models of K+ channels. Proc. Natl Acad. Sci. USA 103, 7292–7297 (2006)
Lee, S. Y. & MacKinnon, R. A membrane-access mechanism of ion channel inhibition by voltage sensor toxins from spider venom. Nature 430, 232–235 (2004)
Phillips, L. R. et al. Voltage-sensor activation with a tarantula toxin as cargo. Nature 436, 857–860 (2005)
Chanda, B., Asamoah, O. K., Blunck, R., Roux, B. & Bezanilla, F. Gating charge displacement in voltage-gated ion channels involves limited transmembrane movement. Nature 436, 852–856 (2005)
Starace, D. M. & Bezanilla, F. A proton pore in a potassium channel voltage sensor reveals a focused electric field. Nature 427, 548–553 (2004)
Islas, L. D. & Sigworth, F. J. Electrostatics and the gating pore of Shaker potassium channels. J. Gen. Physiol. 117, 69–89 (2001)
Ahern, C. A. & Horn, R. Focused electric field across the voltage sensor of potassium channels. Neuron 48, 25–29 (2005)
Minor, D. L. A sensitive channel family replete with sense and motion. Nature Struct. Mol. Biol. 13, 388–390 (2006)
Acknowledgements
We thank S. Wiese for valuable technical assistance, S. Chakravarthy for help with PyMOL, H. P. Larsson for advice about fluctuation analysis, V. Yarov-Yarovoy for the PDB coordinates of the ROSETTA model of KV1.2 and for helpful discussion, and S. Kohout and S. Bell for critical comments on the manuscripts. This work was supported by the National Institutes of Health and by a postdoctoral fellowship from the American Heart Association Western States Affiliate (F.T.). P.G. was supported by postdoctoral fellowships from the Generalitat de Catalunya and the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Notes and Data, Supplementary Figures 1-6 with legends, Supplementary Methods and additional references (PDF 1050 kb)
Rights and permissions
About this article
Cite this article
Tombola, F., Pathak, M., Gorostiza, P. et al. The twisted ion-permeation pathway of a resting voltage-sensing domain. Nature 445, 546–549 (2007). https://doi.org/10.1038/nature05396
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05396
This article is cited by
-
Ion currents through the voltage sensor domain of distinct families of proteins
Journal of Biological Physics (2023)
-
Voltage-dependent gating in K channels: experimental results and quantitative models
Pflügers Archiv - European Journal of Physiology (2020)
-
Sensing the heat with TRPM3
Pflügers Archiv - European Journal of Physiology (2018)
-
MLKL forms cation channels
Cell Research (2016)
-
Reciprocal voltage sensor-to-pore coupling leads to potassium channel C-type inactivation
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.