Abstract
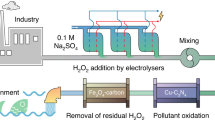
Per- and polyfluoroalkyl substances (PFAS) are highly recalcitrant pollutants in the water environment worldwide. Aqueous film-forming foam (AFFF) used for firefighting is a major source of PFAS pollution. However, complete defluorination (that is, cleaving all C–F bonds into F− ions) of PFAS by non-thermal technology is rare. The destruction of the PFAS mixture in the complex organic matrix of AFFF is even more challenging. Here we designed and demonstrated an ultraviolet/sulfite–electrochemical oxidation (UV/S–EO) process. The tandem UV/S–EO leverages the complementary advantages of UV/S and EO modules in the PFAS transformation mechanism and the engineering process design (for example, foaming control, chemical dosage and energy consumption). At ambient temperature and pressure, the UV/S–EO realized near-complete defluorination and mineralization of most PFAS and organics in AFFF (50–5,000 times diluted, containing up to 200 mg l−1 organic fluorine and 3,764 mg l−1 organic carbon). This work highlights the integration of molecular-level insight and engineering design towards solving the major challenges of AFFF water pollution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information. Source data for all graphs are provided in this paper.
References
Evich, M. G. et al. Per- and polyfluoroalkyl substances in the environment. Science 375, eabg9065 (2022).
Moody, C. A. & Field, J. A. Perfluorinated surfactants and the environmental implications of their use in fire-fighting foams. Environ. Sci. Technol. 34, 3864–3870 (2000).
Awad, E. et al. Long-term environmental fate of perfluorinated compounds after accidental release at Toronto airport. Environ. Sci. Technol. 45, 8081–8089 (2011).
Hu, X. C. et al. Detection of poly- and perfluoroalkyl substances (PFASs) in US drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environ. Sci. Technol. Lett. 3, 344–350 (2016).
Mejia-Avendaño, S. et al. Novel fluoroalkylated surfactants in soils following firefighting foam deployment during the Lac-Megantic railway accident. Environ. Sci. Technol. 51, 8313–8323 (2017).
Liu, M., Munoz, G., Vo Duy, S., Sauvé, S. & Liu, J. Per- and polyfluoroalkyl substances in contaminated soil and groundwater at airports: a Canadian case study. Environ. Sci. Technol. 56, 885–895 (2021).
Cornelsen, M., Weber, R. & Panglisch, S. Minimizing the environmental impact of PFAS by using specialized coagulants for the treatment of PFAS polluted waters and for the decontamination of firefighting equipment. Emerg. Contam. 7, 63–76 (2021).
Lang, J. R. et al. Characterization of per- and polyfluoroalkyl substances on fire suppression system piping and optimization of removal methods. Chemosphere 308, 136254 (2022).
Krause, M. J. et al. Supercritical water oxidation as an innovative technology for PFAS destruction. J. Environ. Eng. 148, 05021006 (2022).
Hao, S. et al. Hydrothermal alkaline treatment for destruction of per- and polyfluoroalkyl substances in aqueous film-forming foam. Environ. Sci. Technol. 55, 3283–3295 (2021).
Pinkard, B. R. Aqueous film-forming foam treatment under alkaline hydrothermal conditions. J. Environ. Eng. 148, 05021007 (2022).
Place, B. J. & Field, J. A. Identification of novel fluorochemicals in aqueous film-forming foams used by the US military. Environ. Sci. Technol. 46, 7120–7127 (2012).
D’Agostino, L. A. & Mabury, S. A. Identification of novel fluorinated surfactants in aqueous film forming foams and commercial surfactant concentrates. Environ. Sci. Technol. 48, 121–129 (2014).
Barzen-Hanson, K. A. et al. Discovery of 40 classes of per- and polyfluoroalkyl substances in historical aqueous film-forming foams (AFFFs) and AFFF-impacted groundwater. Environ. Sci. Technol. 51, 2047–2057 (2017).
Park, H. et al. Reductive defluorination of aqueous perfluorinated alkyl surfactants: effects of ionic headgroup and chain length. J. Phys. Chem. A 113, 690–696 (2009).
Campbell, T. Y., Vecitis, C. D., Mader, B. T. & Hoffmann, M. R. Perfluorinated surfactant chain-length effects on sonochemical kinetics. J. Phys. Chem. A 113, 9834–9842 (2009).
Bentel, M. J. et al. Defluorination of per- and polyfluoroalkyl substances (PFASs) with hydrated electrons: structural dependence and implications to PFAS remediation and management. Environ. Sci. Technol. 53, 3718–3728 (2019).
Liu, Z. et al. Accelerated degradation of perfluorosulfonates and perfluorocarboxylates by UV/sulfite + iodide: reaction mechanisms and system efficiencies. Environ. Sci. Technol. 56, 3699–3709 (2022).
Singh, R. K. et al. Removal of poly- and per-fluorinated compounds from ion exchange regenerant still bottom samples in a plasma reactor. Environ. Sci. Technol. 54, 13973–13980 (2020).
Li, R., Isowamwen, O. F., Ross, K. C., Holsen, T. M. & Thagard, S. M. PFAS–CTAB complexation and its role on the removal of PFAS from a lab-prepared water and a reverse osmosis reject water using a plasma reactor. Environ. Sci. Technol. 57, 12901–12910 (2023).
Zhuo, Q. et al. Degradation of perfluorinated compounds on a boron-doped diamond electrode. Electrochim. Acta 77, 17–22 (2012).
Schaefer, C. E. et al. Electrochemical transformations of perfluoroalkyl acid (PFAA) precursors and PFAAs in groundwater impacted with aqueous film forming foams. Environ. Sci. Technol. 52, 10689–10697 (2018).
Liang, S. et al. Field demonstration of coupling ion-exchange resin with electrochemical oxidation for enhanced treatment of per- and polyfluoroalkyl substances (PFAS) in groundwater. Chem. Eng. J. Adv. 9, 100216 (2022).
Tenorio, R. et al. Destruction of per- and polyfluoroalkyl substances (PFASs) in aqueous film-forming foam (AFFF) with UV–sulfite photoreductive treatment. Environ. Sci. Technol. 54, 6957–6967 (2020).
Gomez-Ruiz, B. et al. Efficient electrochemical degradation of poly- and perfluoroalkyl substances (PFASs) from the effluents of an industrial wastewater treatment plant. Chem. Eng. J. 322, 196–204 (2017).
Schaefer, C. E. et al. Electrochemical treatment of perfluorooctanoic acid and perfluorooctane sulfonate: insights into mechanisms and application to groundwater treatment. Chem. Eng. J. 317, 424–432 (2017).
Macpherson, J. V. A practical guide to using boron doped diamond in electrochemical research. Phys. Chem. Chem. Phys. 17, 2935–2949 (2015).
Radjenovic, J., Duinslaeger, N., Avval, S. S. & Chaplin, B. P. Facing the challenge of poly- and perfluoroalkyl substances in water: is electrochemical oxidation the answer? Environ. Sci. Technol. 54, 14815–14829 (2020).
Yang, Y. Recent advances in the electrochemical oxidation water treatment: spotlight on byproduct control. Front. Environ. Sci. Eng. 14, 85 (2020).
Shin, Y.-U. et al. Electrochemical oxidation of organics in sulfate solutions on boron-doped diamond electrode: multiple pathways for sulfate radical generation. Appl. Catal. B 254, 156–165 (2019).
Yang, S., Fernando, S., Holsen, T. M. & Yang, Y. Inhibition of perchlorate formation during the electrochemical oxidation of perfluoroalkyl acid in groundwater. Environ. Sci. Technol. Lett. 6, 775–780 (2019).
Liu, Y. et al. Enhanced perfluorooctanoic acid degradation by electrochemical activation of sulfate solution on B/N codoped diamond. Environ. Sci. Technol. 53, 5195–5201 (2019).
Farhat, A., Keller, J., Tait, S. & Radjenovic, J. Removal of persistent organic contaminants by electrochemically activated sulfate. Environ. Sci. Technol. 49, 14326–14333 (2015).
Houtz, E. F. & Sedlak, D. L. Oxidative conversion as a means of detecting precursors to perfluoroalkyl acids in urban runoff. Environ. Sci. Technol. 46, 9342–9349 (2012).
Lutze, H. V., Brekenfeld, J., Naumov, S., von Sonntag, C. & Schmidt, T. C. Degradation of perfluorinated compounds by sulfate radicals—new mechanistic aspects and economical considerations. Water Res. 129, 509–519 (2018).
Liu, Z. et al. Near-quantitative defluorination of perfluorinated and fluorotelomer carboxylates and sulfonates with integrated oxidation and reduction. Environ. Sci. Technol. 55, 7052–7062 (2021).
Niu, J., Li, Y., Shang, E., Xu, Z. & Liu, J. Electrochemical oxidation of perfluorinated compounds in water. Chemosphere 146, 526–538 (2016).
Shende, T., Andaluri, G. & Suri, R. Chain-length dependent ultrasonic degradation of perfluoroalkyl substances. Chem. Eng. J. Adv. 15, 100509 (2023).
Isowamwen, O., Li, R., Holsen, T. & Thagard, S. M. Plasma-assisted degradation of a short-chain perfluoroalkyl substance (PFAS): perfluorobutane sulfonate (PFBS). J. Hazard. Mater. 456, 131691 (2023).
Gao, J. et al. Photochemical degradation pathways and near-complete defluorination of chlorinated polyfluoroalkyl substances. Nat. Water 1, 381–390 (2023).
Houtz, E. F., Higgins, C. P., Field, J. A. & Sedlak, D. L. Persistence of perfluoroalkyl acid precursors in AFFF-impacted groundwater and soil. Environ. Sci. Technol. 47, 8187–8195 (2013).
Singh, R. K. et al. Rapid removal of poly- and perfluorinated compounds from investigation-derived waste (IDW) in a pilot-scale plasma reactor. Environ. Sci. Technol. 53, 11375–11382 (2019).
Dalvi, V. H. & Rossky, P. J. Molecular origins of fluorocarbon hydrophobicity. Proc. Natl Acad. Sci. USA 107, 13603–13607 (2010).
Crittenden, J. C., Trussell R. R., Hand, D. W., Howe, K. J. & Tchobanoglous, G. MWH’s Water Treatment: Principles and Design (John Wiley & Sons, 2012).
Shields, E. P. et al. Pilot-scale thermal destruction of per- and polyfluoroalkyl substances in a legacy aqueous film forming foam. ACS ES&T Eng. 3, 1308–1317 (2023).
Maga, D., Aryan, V. & Bruzzano, S. Environmental assessment of various end-of-life pathways for treating per- and polyfluoroalkyl substances in spent fire-extinguishing waters. Environ. Toxicol. Chem. 40, 947–957 (2021).
Gao, J. et al. Defluorination of omega-hydroperfluorocarboxylates (ω-HPFCAs): distinct reactivities from perfluoro and fluorotelomeric carboxylates. Environ. Sci. Technol. 55, 14146–14155 (2021).
Bentel, M. J. et al. Degradation of perfluoroalkyl ether carboxylic acids with hydrated electrons: structure–reactivity relationships and environmental implications. Environ. Sci. Technol. 54, 2489–2499 (2020).
Gao, J., Xie, S., Liu, F. & Liu, J. Preparation and synergy of supported Ru0 and Pd0 for rapid chlorate reduction at pH 7. Environ. Sci. Technol. 57, 3962–3970 (2023).
Ren, C., Bi, E. Y., Gao, J. & Liu, J. Molybdenum-catalyzed perchlorate reduction: robustness, challenges, and solutions. ACS ES&T Eng. 2, 181–188 (2022).
Liu, J. & Gao, J. Catalytic reduction of water pollutants: knowledge gaps, lessons learned, and new opportunities. Front. Environ. Sci. Eng. 17, 26 (2022).
Yang, N. et al. Solvent-free nonthermal destruction of PFAS chemicals and PFAS in sediment by piezoelectric ball milling. Environ. Sci. Technol. Lett. 10, 198–203 (2023).
Li, X. et al. Efficient reductive dechlorination of monochloroacetic acid by sulfite/UV process. Environ. Sci. Technol. 46, 7342–7349 (2012).
Bentel, M. J. et al. Enhanced degradation of perfluorocarboxylic acids (PFCAs) by UV/sulfite treatment: reaction mechanisms and system efficiencies at pH 12. Environ. Sci. Technol. Lett. 7, 351–357 (2020).
Acknowledgements
Financial support was provided by the Strategic Environmental Research and Development Program (ER22-3184 for Y.G., Z.L., N.Y., J.L. and Y.Y.) and the National Science Foundation (CBET-2120452 for S.Y., L.E.Q.-C. and Y.Y.). We thank S. Fernando and T. Holsen at Clarkson University for providing the Ansulite AFFF sample and the assistance in non-targeted PFAS analyses.
Author information
Authors and Affiliations
Contributions
Y.G. conducted PFAS degradation experiments, analysed the data and drafted the paper. Z.L. conducted PFAS degradation and NMR analysis. N.Y., S.Y. and L.E.Q.-C. assisted in the liquid chromatography high-resolution tandem mass spectrometry analysis. J.L. and Y.Y. designed and supervised the research and revised the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Shilai Hao and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary Texts 1–3, Tables 1–5 and Figs. 1–21.
Source data
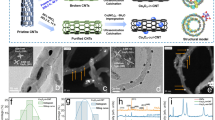
Source Data Fig. 1
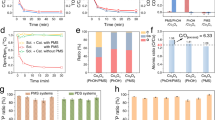
EO treatment of individual PFAS.
Source Data Fig. 2
UV/S–EO degradation and defluorination of individual PFAS.
Source Data Fig. 3
Transformation of PFOA during UV/S–EO treatment.
Source Data Fig. 4
UV/S–EO treatment of AFFF.
Source Data Fig. 5
Variation of TOC and foaming potentials during UV/S–EO treatment; energy consumption evaluation.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guan, Y., Liu, Z., Yang, N. et al. Near-complete destruction of PFAS in aqueous film-forming foam by integrated photo-electrochemical processes. Nat Water (2024). https://doi.org/10.1038/s44221-024-00232-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s44221-024-00232-7
This article is cited by
-
Cutting all carbonâfluorine bonds from PFAS
Nature Water (2024)