Abstract
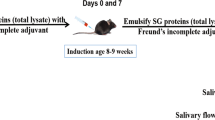
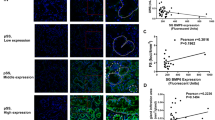
Primary Sjögren’s syndrome (pSS) is a chronic inflammatory autoimmune disease with an unclear pathogenesis, and there is currently no approved drug for the treatment of this disease. Iguratimod, as a novel clinical anti-rheumatic drug in China and Japan, has shown remarkable efficacy in improving the symptoms of patients with pSS in clinical studies. In this study we investigated the mechanisms underlying the therapeutic effect of iguratimod in the treatment of pSS. Experimental Sjögren’s syndrome (ESS) model was established in female mice by immunizing with salivary gland protein. After immunization, ESS mice were orally treated with iguratimod (10, 30, 100 mg·kg−1·d−1) or hydroxychloroquine (50 mg·kg−1·d−1) for 70 days. We showed that iguratimod administration dose-dependently increased saliva secretion, and ameliorated ESS development by predominantly inhibiting B cells activation and plasma cell differentiation. Iguratimod (30 and 100 mg·kg−1·d−1) was more effective than hydroxychloroquine (50 mg·kg−1·d−1). When the potential target of iguratimod was searched, we found that iguratimod bound to TEC kinase and promoted its degradation through the autophagy-lysosome pathway in BAFF-activated B cells, thereby directly inhibiting TEC-regulated B cells function, suggesting that the action mode of iguratimod on TEC was different from that of conventional kinase inhibitors. In addition, we found a crucial role of TEC overexpression in plasma cells of patients with pSS. Together, we demonstrate that iguratimod effectively ameliorates ESS via its unique suppression of TEC function, which will be helpful for its clinical application. Targeting TEC kinase, a new regulatory factor for B cells, may be a promising therapeutic option.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Brito-Zerón P, Baldini C, Bootsma H, Bowman SJ, Jonsson R, Mariette X, et al. Sjögren syndrome. Nat Rev Dis Prim. 2016;2:16047.
Kovács L, Szodoray P, Kiss E. Secondary tumours in Sjögren’s syndrome. Autoimmun Rev. 2010;9:203–6.
Katsiougiannis S, Tenta R, Skopouli FN. Autoimmune epithelitis (Sjögren’s syndrome); the impact of metabolic status of glandular epithelial cells on auto-immunogenicity. J Autoimmun. 2019;104:102335.
Yao YH, Liu Z, Jallal B, Shen N, Rönnblom L. Type I interferons in Sjögren’s syndrome. Autoimmun Rev. 2013;12:558–66.
Talal N, Sylvester RA, Daniels TE, Greenspan JS, Williams RC Jr. T and B lymphocytes in peripheral blood and tissue lesions in Sjögren’s syndrome. J Clin Invest. 1974;53:180–9.
Cornec D, Devauchelle-Pensec V, Tobón GJ, Pers JO, Jousse-Joulin S, Saraux A. B cells in Sjögren’s syndrome: from pathophysiology to diagnosis and treatment. J Autoimmun. 2012;39:161–7.
Nocturne G, Seror R, Fogel O, Belkhir R, Boudaoud S, Saraux A, et al. CXCL13 and CCL11 serum levels and lymphoma and disease activity in Primary Sjögren’s syndrome. Arthritis Rheumatol. 2015;67:3226–33.
Zuckerman NS, Hazanov H, Barak M, Edelman H, Hess S, Shcolnik H, et al. Somatic hypermutation and antigen-driven selection of B cells are altered in autoimmune diseases. J Autoimmun. 2010;35:325–35.
Jackson SW, Kolhatkar NS, Rawlings DJ. B cells take the front seat: dysregulated B cell signals orchestrate loss of tolerance and autoantibody production. Curr Opin Immunol. 2015;33:70–77.
Groom J, Kalled SL, Cutler AH, Olson C, Woodcock SA, Schneider P, et al. Association of BAFF/BLyS overexpression and altered B cell differentiation with Sjögren’s syndrome. J Clin Invest. 2002;109:59–68.
Mariette X, Roux S, Zhang J, Bengoufa D, Lavie F, Zhou T, et al. The level of BLyS (BAFF) correlates with the titre of autoantibodies in human Sjögren’s syndrome. Ann Rheum Dis. 2003;62:168–71.
Carrillo-Ballesteros FJ, Palafox-Sanchez CA, Franco-Topete RA, Munoz-Valle JF, Orozco-Barocio G, Martinez-Bonilla GE, et al. Expression of BAFF and BAFF receptors in primary Sjögren’s syndrome patients with ectopic germinal center-like structures. Clin Exp Med. 2020;20:615–26.
Quartuccio L, Salvin S, Fabris M, Maset M, Pontarini E, Isola M, et al. BLyS upregulation in Sjögren’s syndrome associated with lymphoproliferative disorders, higher ESSDAI score and B-cell clonal expansion in the salivary glands. Rheumatology (Oxf). 2013;52:276–81.
Loureiro-Amigo J, Franco-Jarava C, Perurena-Prieto J, Palacio C, Martinez-Valle F, Solans-Laque R. Serum CXCL13, BAFF, IL-21 and IL-22 levels are related to disease activity and lymphocyte profile in primary Sjögren’s syndrome. Clin Exp Rheumatol. 2021;39:131–9.
Nair JJ, Singh TP. Sjögren’s syndrome: Review of the aetiology, pathophysiology & potential therapeutic interventions. J Clin Exp Dent. 2017;9:e584–e9.
Saraux A, Pers JO, Devauchelle-Pensec V. Treatment of primary Sjögren syndrome. Nat Rev Rheumatol. 2016;12:456–71.
Brito-Zeron P, Retamozo S, Kostov B, Baldini C, Bootsma H, De Vita S, et al. Efficacy and safety of topical and systemic medications: a systematic literature review informing the EULAR recommendations for the management of Sjögren’s syndrome. RMD Open. 2019;5:e001064.
Alvarez-Rivas N, Sang-Park H, Diaz Del Campo P, Fernandez-Castro M, Corominas H, Andreu JL, et al. Efficacy of belimumab in Primary Sjögren’s syndrome: a systematic review. Reumatol Clin (Engl Ed). 2021;17:170–4.
Mariette X, Barone F, Baldini C, Bootsma H, Clark KL, De Vita S, et al. A randomized, phase II study of sequential belimumab and rituximab in primary Sjögren’s syndrome. JCI Insight. 2022;7:e163030.
Wang B, Chen SJ, Li Y, Xuan JX, Liu Y, Shi GX. Targeted therapy for Primary Sjögren’s syndrome: where are we now? BioDrugs. 2021;35:593–610.
Bowman SJ, Fox R, Dörner T, Mariette X, Papas A, Grader-Beck T, et al. Safety and efficacy of subcutaneous ianalumab (VAY736) in patients with primary Sjögren’s syndrome: a randomised, double-blind, placebo-controlled, phase 2b dose-finding trial. Lancet. 2022;399:161–71.
Kaklamanos A, Goules AV, Tzioufas AG. Experimental models of Sjögren’s syndrome: differences and similarities with human disease. Clin Exp Rheumatol. 2022;40:2398–412.
Tanaka K, Yamaguchi T, Hara M. Iguratimod for the treatment of rheumatoid arthritis in Japan. Expert Rev Clin Immunol. 2015;11:565–73.
Luo Q, Sun Y, Liu W, Qian C, Jin B, Tao FF, et al. A novel disease-modifying antirheumatic drug, iguratimod, ameliorates murine arthritis by blocking IL-17 signaling, distinct from methotrexate and leflunomide. J Immunol. 2013;191:4969–78.
Luo Q, Liu YJ, Shi K, Shen XC, Yang YQ, Liang XJ, et al. An autonomous activation of interleukin-17 receptor signaling sustains inflammation and promotes disease progression. Immunity. 2023;56:2006–20.
Liu WP, Han X, Li Q, Sun LQ, Wang JB. Iguratimod ameliorates bleomycin-induced pulmonary fibrosis by inhibiting the EMT process and NLRP3 inflammasome activation. Biomed Pharmacother. 2022;153:113460.
Zhang M, Lei YS, Meng XW, Liu HY, Li LG, Zhang J, et al. Iguratimod alleviates myocardial ischemia/reperfusion injury through inhibiting inflammatory response induced by cardiac fibroblast pyroptosis via COX2/NLRP3 signaling pathway. Front Cell Dev Biol. 2021;9:746317.
Li GR, Yamasaki R, Fang M, Masaki K, Ochi H, Matsushita T, et al. Novel disease-modifying anti-rheumatic drug iguratimod suppresses chronic experimental autoimmune encephalomyelitis by down-regulating activation of macrophages/microglia through an NF-kappaB pathway. Sci Rep. 2018;8:1933.
Xia Y, Fang X, Dai XJ, Li MY, Jin L, Tao JH, et al. Iguratimod ameliorates nephritis by modulating the Th17/Treg paradigm in pristane-induced lupus. Int Immunopharmacol. 2021;96:107563.
Jiang XP, Huang XL, Yang ZP, Wang SC, Xie W, Miao L, et al. Iguratimod ameliorates inflammatory responses by modulating the Th17/Treg paradigm in dextran sulphate sodium-induced murine colitis. Mol Immunol. 2018;93:9–19.
Zeng LT, He Q, Yang KL, Hao WS, Yu GP, Chen H. A systematic review and meta-analysis of 19 randomized controlled trials of iguratimod combined with other therapies for Sjögren’s syndrome. Front Immunol. 2022;13:924730.
Pu JC, Wang X, Riaz F, Zhang TYZ, Gao RL, Pan SN, et al. Effectiveness and safety of iguratimod in treating primary Sjögren’s syndrome: a systematic review and meta-analysis. Front Pharmacol. 2021;12:621208.
Xiao F, Lin X, Tian J, Wang XH, Chen Q, Rui K, et al. Proteasome inhibition suppresses Th17 cell generation and ameliorates autoimmune development in experimental Sjögren’s syndrome. Cell Mol Immunol. 2017;14:924–34.
Scofield RH, Dickey WD, Jackson KW, James JA, Harley JB. A common autoepitope near the carboxyl terminus of the 60-kD Ro ribonucleoprotein: sequence similarity with a viral protein. J Clin Immunol. 1991;11:378–88.
Rui K, Hong Y, Zhu QG, Shi XF, Xiao F, Fu HL, et al. Olfactory ecto-mesenchymal stem cell-derived exosomes ameliorate murine Sjögren’s syndrome by modulating the function of myeloid-derived suppressor cells. Cell Mol Immunol. 2021;18:440–51.
Yu S, Wu ML, Feng Y, Shen JG, Lu LW, Lin X. Detection of T follicular helper cells and T follicular regulatory cells in experimental Sjögren’s syndrome. Methods Mol Biol. 2022;2380:211–24.
Tian J, Zhang BB, Yuan QF, Sun XQ, Peng N, Zhu B, et al. GITRL impairs the immunosuppressive function of MDSCs via PTEN-mediated signaling pathway in experimental Sjögren syndrome. Inflamm Res. 2022;71:1577–88.
Wei YX, Peng N, Deng C, Zhao F, Tian J, Tang Y, et al. Aryl hydrocarbon receptor activation drives polymorphonuclear myeloid-derived suppressor cell response and efficiently attenuates experimental Sjögren’s syndrome. Cell Mol Immunol. 2022;19:1361–72.
Verstappen GM, Pringle S, Bootsma H, Kroese FGM. Epithelial-immune cell interplay in primary Sjögren syndrome salivary gland pathogenesis. Nat Rev Rheumatol. 2021;17:333–48.
Tomlinson MG, Kane LP, Su J, Kadlecek TA, Mollenauer MN, Weiss A. Expression and function of Tec, Itk, and Btk in lymphocytes: evidence for a unique role for Tec. Mol Cell Biol. 2004;24:2455–66.
Li XL, Liu XC, Song YL, Hong RT, Shi H. Suspected drug-induced liver injury associated with iguratimod: a case report and review of the literature. BMC Gastroenterol. 2018;18:130.
Vidarsson G, Dekkers G, Rispens T. IgG subclasses and allotypes: from structure to effector functions. Front Immunol. 2014;5:520.
Ye Y, Liu M, Tang LH, Du F, Liu YH, Hao P, et al. Iguratimod represses B cell terminal differentiation linked with the inhibition of PKC/EGR1 axis. Arthritis Res Ther. 2019;21:92.
Schwartzberg PL, Finkelstein LD, Readinger JA. TEC-family kinases: regulators of T-helper-cell differentiation. Nat Rev Immunol. 2005;5:284–95.
McDonald C, Xanthopoulos C, Kostareli E. The role of Bruton’s tyrosine kinase in the immune system and disease. Immunology. 2021;164:722–36.
Ellmeier W, Jung S, Sunshine MJ, Hatam F, Xu Y, Baltimore D, et al. Severe B cell deficiency in mice lacking the Tec kinase family members Tec and Btk. J Exp Med. 2000;192:1611–24.
de Bruijn MJW, Rip J, van der Ploeg EK, van Greuningen LW, Ta VTB, Kil LP, et al. Distinct and overlapping functions of TEC kinase and BTK in B cell receptor signaling. J Immunol. 2017;198:3058–68.
Ringheim GE, Wampole M, Oberoi K. Bruton’s tyrosine kinase (BTK) inhibitors and autoimmune diseases: making sense of BTK iInhibitor specificity profiles and recent clinical trial successes and failures. Front Immunol. 2021;12:662223.
Acknowledgements
This study was supported by National Natural Science Foundation of China (82230116, 21937005, 81773743, 81973331), Innovation Team and Talents Cultivation Program of National Administration of Traditional Chinese Medicine (ZYYCXTD-C-202208), and the Fundamental Research Funds for the Central Universities of China (0208/14380185).
Author information
Authors and Affiliations
Contributions
QX and QL conceived, designed, and directed the study. YQY, YJL and WJ performed the in vivo experiments and analyzed data. YQY and WXQ performed the cell experiments in vitro and analyzed data. SWZ and YXY provided experimental materials and scientific suggestions. YQY and QL wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest. All the research data in this manuscript were completed by Xu’s group and supported by the listed funding. Authors SWZ and YXY were employed by Jiangsu Simcere Pharmaceutical Co., Ltd. and were involved in the discussion of the content of the paper. However, they declare no conflict of interest on the publication of the research paper.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Yq., Liu, Yj., Qiao, Wx. et al. Iguratimod suppresses plasma cell differentiation and ameliorates experimental Sjögren’s syndrome in mice by promoting TEC kinase degradation. Acta Pharmacol Sin (2024). https://doi.org/10.1038/s41401-024-01288-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41401-024-01288-7