Featured
-
-
Article
| Open Access Structural insights into the function of type VI secretion system TssA subunits
Structural insights into the function of type VI secretion system TssA subunits
TssA is an important component of the bacterial type VI secretion system (T6SS). Here, Dix et al. integrate structural, phylogenetic and functional analysis of the TssA subunits, providing new insights into their role in T6SS assembly and function.
- Samuel R. Dix
- , Hayley J. Owen
- & Mark S. Thomas
-
Article
| Open Access Proteomics reveals signal peptide features determining the client specificity in human TRAP-dependent ER protein import
Proteomics reveals signal peptide features determining the client specificity in human TRAP-dependent ER protein import
While Sec61 enables ER import of all polypeptides with N-terminal signal peptides, only selected clients are accepted for TRAP-assisted ER import. Here, the authors use a proteomics approach to characterize TRAP-dependent clients, identifying signal peptide features that govern recognition by TRAP.
- Duy Nguyen
- , Regine Stutz
- & Richard Zimmermann
-
Article
| Open Access T cell microvilli constitute immunological synaptosomes that carry messages to antigen-presenting cells
T cell microvilli constitute immunological synaptosomes that carry messages to antigen-presenting cells
Microvilli can participate in adhesion or migration of T cells, but whether they are involved in function regulation is unclear. Here the authors show that T cell microvilli form budding vesicles containing T cell signalling components for deposition onto antigen presenting cells (APC) and modulation of APC functions.
- Hye-Ran Kim
- , YeVin Mun
- & Chang-Duk Jun
-
Article
| Open Access Revealing the mechanisms of membrane protein export by virulence-associated bacterial secretion systems
Revealing the mechanisms of membrane protein export by virulence-associated bacterial secretion systems
Many bacteria export effector proteins even when two incompatible signal sequences are present, one which would lead to export and the other to inner membrane targeting. Here the authors show that such proteins feature decreased hydrophobicity or cognate chaperone binding to prevent erroneous targeting.
- Lea Krampen
- , Silke Malmsheimer
- & Samuel Wagner
-
Article
| Open Access SUMOylation of VEGFR2 regulates its intracellular trafficking and pathological angiogenesis
SUMOylation of VEGFR2 regulates its intracellular trafficking and pathological angiogenesis
VEGFR2 is a central regulator of angiogenesis. Here Zhou et al. report that SUMOylation of VEGFR2 regulates its subcellular localisation and activity, and that endothelial-specific knockout of the SUMO endopeptidase SENP1 protects against VEGFR2-mediated pathological angiogenesis.
- Huanjiao Jenny Zhou
- , Zhe Xu
- & Wang Min
-
Article
| Open Access SKP2- and OTUD1-regulated non-proteolytic ubiquitination of YAP promotes YAP nuclear localization and activity
SKP2- and OTUD1-regulated non-proteolytic ubiquitination of YAP promotes YAP nuclear localization and activity
Regulation of Yes-associated protein (YAP) through the Hippo pathway is well established, but its Hippo-independent regulation remains to be elucidated. Here, the authors show that non-proteolytic ubiquitination presents another means of YAP regulation, promoting its nuclear localization and activity.
- Fan Yao
- , Zhicheng Zhou
- & Li Ma
-
Article
| Open Access Cyclophilin A enables specific HIV-1 Tat palmitoylation and accumulation in uninfected cells
Cyclophilin A enables specific HIV-1 Tat palmitoylation and accumulation in uninfected cells
It is not clear whether and how incoming HIV-1 Tat accumulates in uninfected cells. Here, Chopard et al. show that, in uninfected cells, incoming Tat is palmitoylated on Cys31 by DHHC-20, which increases its affinity for PI(4,5)P2 and results in its accumulation at the plasma membrane.
- Christophe Chopard
- , Phuoc Bao Viet Tong
- & Bruno Beaumelle
-
Article
| Open Access A novel small molecule chaperone of rod opsin and its potential therapy for retinal degeneration
A novel small molecule chaperone of rod opsin and its potential therapy for retinal degeneration
Mutations that lead to misfolding of rhodopsin can cause retinitis pigmentosa. Here, the authors carry out a high throughput screen to identify a small molecule chaperone of rod opsin, and show that it protects mouse models of retinitis pigmentosa from retinal degeneration.
- Yuanyuan Chen
- , Yu Chen
- & Krzysztof Palczewski
-
Correspondence
| Open Access Correspondence: Reply to ‘Challenging a proposed role for TRPC5 in aortic baroreceptor pressure-sensing’
Correspondence: Reply to ‘Challenging a proposed role for TRPC5 in aortic baroreceptor pressure-sensing’
- On-Chai Lau
- , Bing Shen
- & Xiaoqiang Yao
-
Correspondence
| Open Access Correspondence: Challenging a proposed role for TRPC5 in aortic baroreceptor pressure-sensing
Correspondence: Challenging a proposed role for TRPC5 in aortic baroreceptor pressure-sensing
- Pratish Thakore
- , Susan D. Brain
- & David J. Beech
-
Article
| Open Access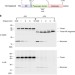 Folding of a bacterial integral outer membrane protein is initiated in the periplasm
Folding of a bacterial integral outer membrane protein is initiated in the periplasm
The Bam complex promotes the insertion of β-barrel proteins (such as UpaG, a trimeric autotransporter adhesin) into the bacterial outer membrane. Here, Sikdar et al. show that UpaG β-barrel segments fold into a trimeric structure in the periplasm before they interact with the Bam complex.
- Rakesh Sikdar
- , Janine H. Peterson
- & Harris D. Bernstein
-
Article
| Open Access Efficient protein production by yeast requires global tuning of metabolism
Efficient protein production by yeast requires global tuning of metabolism
The contribution of metabolic pathways to protein secretion is largely unknown. Here, the authors find conserved metabolic patterns in yeast by examining genome-wide transcriptional responses in high protein secretion mutants and reveal critical factors that can be tuned for efficient protein secretion.
- Mingtao Huang
- , Jichen Bao
- & Jens Nielsen
-
Article
| Open Access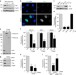 Exploiting the kinesin-1 molecular motor to generate a virus membrane penetration site
Exploiting the kinesin-1 molecular motor to generate a virus membrane penetration site
How non-enveloped viruses cross host membranes is incompletely understood. Here, Ravindranet al. show that polyomavirus SV40 recruits kinesin-1 to construct a penetration site on the ER membrane.
- Madhu Sudhan Ravindran
- , Martin F. Engelke
- & Billy Tsai
-
Article
| Open Access Allosteric modulation of peroxisomal membrane protein recognition by farnesylation of the peroxisomal import receptor PEX19
Allosteric modulation of peroxisomal membrane protein recognition by farnesylation of the peroxisomal import receptor PEX19
PEX19 is a chaperone and import receptor for peroxisomal membrane proteins (PMPs). Here the authors present the structure of the farnesylated C-terminal domain of PEX19, and its interaction with PMPs reveals how the farnesyl moiety allosterically reshapes the PMP binding surface and modulates PEX19 function.
- Leonidas Emmanouilidis
- , Ulrike Schütz
- & Michael Sattler
-
Article
| Open Access Dissecting the molecular organization of the translocon-associated protein complex
Dissecting the molecular organization of the translocon-associated protein complex
The translocon-associated protein complex (TRAP) is a crucial component of the endoplasmic reticulum protein translocon. Here the authors study native translocon structures from human disease patients and algae cells to determine the molecular organization of the TRAP complex.
- Stefan Pfeffer
- , Johanna Dudek
- & Friedrich Förster
-
Article
| Open Access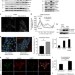 Starved epithelial cells uptake extracellular matrix for survival
Starved epithelial cells uptake extracellular matrix for survival
Inhibition of PI3K/mTOR, which mimics nutrient starvation, causes death of detached but not matrix-attached cancer cells. Here the authors show that nutrient restriction of epithelial cells causes uptake of the matrix protein laminin, which results in increased intracellular amino acids and enhanced mTORC1 signalling.
- Taru Muranen
- , Marcin P. Iwanicki
- & Nada Y. Kalaany
-
Article
| Open Access The non-canonical mitochondrial inner membrane presequence translocase of trypanosomatids contains two essential rhomboid-like proteins
The non-canonical mitochondrial inner membrane presequence translocase of trypanosomatids contains two essential rhomboid-like proteins
The mitochondrial protein import machinery is crucial for eukaryotes but little is known about its evolutionary origin. Here, the authors characterize the translocase of the inner membrane (TIM) in trypanosomes, showing that it contains two rhomboid-like proteins essential for protein import.
- Anke Harsman
- , Silke Oeljeklaus
- & André Schneider
-
Article
| Open Access Wnt5a induces renal AQP2 expression by activating calcineurin signalling pathway
Wnt5a induces renal AQP2 expression by activating calcineurin signalling pathway
The water channel AQP2 mediates the concentration of urine in the kidney. Here Ando et al. show that Wnt5 promotes collecting duct permeability by regulating AQP2 expression and localization through activation of the calmodulin/calcineurin signalling pathway.
- Fumiaki Ando
- , Eisei Sohara
- & Shinichi Uchida
-
Article
| Open Access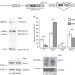 Sec16 alternative splicing dynamically controls COPII transport efficiency
Sec16 alternative splicing dynamically controls COPII transport efficiency
The transport of secretory proteins from the endoplasmic reticulum to the Golgi depends on COPII-coated vesicles. Here, the authors show that activation-induced alternative splicing of Sec16 controls adaptation of COPII transport to increased secretory cargo upon T cell activation.
- Ilka Wilhelmi
- , Regina Kanski
- & Florian Heyd
-
Article
| Open Access MCOLN1 is a ROS sensor in lysosomes that regulates autophagy
MCOLN1 is a ROS sensor in lysosomes that regulates autophagy
Reactive oxygen species (ROS) damage cell components, necessitating their clearance through autophagy. Here, the authors show that ROS can induce autophagy by triggering TRPML1 to release Ca2+from the lysosomal lumen, in turn activating the autophagy and lysosomal biogenesis regulator TFEB.
- Xiaoli Zhang
- , Xiping Cheng
- & Haoxing Xu
-
Article
| Open Access Real-time quantification of protein expression at the single-cell level via dynamic protein synthesis translocation reporters
Real-time quantification of protein expression at the single-cell level via dynamic protein synthesis translocation reporters
Single cells can display large heterogeneity in gene induction. Here, Aymoz et al. present an expression reporter based on protein translocation that can accurately measure both the levels and dynamics of protein synthesis in live single cells with a temporal resolution of less than one minute.
- Delphine Aymoz
- , Victoria Wosika
- & Serge Pelet
-
Article
| Open Access VAMP7 regulates constitutive membrane incorporation of the cold-activated channel TRPM8
VAMP7 regulates constitutive membrane incorporation of the cold-activated channel TRPM8
The temperature-sensitive TRPM8 channel is essential for cold sensing and has been linked to pathological cold hypersensitivity. Here, the authors find TRPM8 insertion in the cell membrane is mediated by VAMP7 following atypical LAMP1-containing vesicle transport, and that loss of VAMP7 leads to reduced cold avoidance in vivo.
- Debapriya Ghosh
- , Silvia Pinto
- & Thomas Voets
-
Article
| Open Access Export of malaria proteins requires co-translational processing of the PEXEL motif independent of phosphatidylinositol-3-phosphate binding
Export of malaria proteins requires co-translational processing of the PEXEL motif independent of phosphatidylinositol-3-phosphate binding
Export of Plasmodium falciparum proteins into infected erythrocytes relies upon the PEXEL motif in target proteins. Here Boddey et al.challenge the hypothesis that the PEXEL motif mediates export by binding PI(3)P and instead suggest it acts via cleavage by plasmepsin V.
- Justin A. Boddey
- , Matthew T. O’Neill
- & Alan F. Cowman
-
Article
| Open Access Conversion of graded phosphorylation into switch-like nuclear translocation via autoregulatory mechanisms in ERK signalling
Conversion of graded phosphorylation into switch-like nuclear translocation via autoregulatory mechanisms in ERK signalling
While ERK signalling can produce switch-like cell behaviour, phosphorylation of ERK increases linearly with extracellular signals. Here, the authors solve this seeming contradiction by showing that nuclear translocation of ERK behaves in a switch-like manner and is controlled by ERK activity.
- Yuki Shindo
- , Kazunari Iwamoto
- & Koichi Takahashi
-
Article
| Open Access Leukotriene C4 is the major trigger of stress-induced oxidative DNA damage
Leukotriene C4 is the major trigger of stress-induced oxidative DNA damage
Chemotherapeutic agents elicit ER and oxidative stress as part of their mode of action. Here the authors show that chemotherapy and ER stress trigger MGST2-based biosynthesis of LTC4, whose inhibition abolishes chemotherapy- and ER stress-triggered oxidative stress and DNA damage, resulting in the attenuation of cell death.
- Efrat Dvash
- , Michal Har-Tal
- & Menachem Rubinstein
-
Article
| Open Access Mammalian SRP receptor switches the Sec61 translocase from Sec62 to SRP-dependent translocation
Mammalian SRP receptor switches the Sec61 translocase from Sec62 to SRP-dependent translocation
Sec62 is a membrane-bound protein that is involved in the translocation of proteins via the signal recognition particle-independent pathway. Here, the authors show that the receptor SRα displaces Sec62 from the translocon and isolate the domain on SRα that is responsible for this.
- Bhalchandra Jadhav
- , Michael McKenna
- & Martin R. Pool
-
Article
| Open Access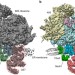 Structure of the native Sec61 protein-conducting channel
Structure of the native Sec61 protein-conducting channel
The protein-conducting channel Sec61 is responsible for protein transport and membrane insertion at the endoplasmic reticulum. Here, the authors determine the structure of ribosome-bound Sec61 in a native context, in which it adopts a laterally open conformation, irrespective of its functional state.
- Stefan Pfeffer
- , Laura Burbaum
- & Friedrich Förster
-
Article
| Open Access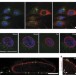 Nuclear envelope-associated endosomes deliver surface proteins to the nucleus
Nuclear envelope-associated endosomes deliver surface proteins to the nucleus
Endocytosis typically directs proteins on a recycling route back to the plasma membrane, transport to the Golgi apparatus or delivery to the lysosome. Here Chaumet et al.describe a population of vesicles that can fuse directly with the outer nuclear membrane and deliver cargo into the nuclear envelope, where it can be translocated into the nucleoplasm.
- Alexandre Chaumet
- , Graham D. Wright
- & Frederic Bard
-
Article
| Open Access Insights into the origin of the nuclear localization signals in conserved ribosomal proteins
Insights into the origin of the nuclear localization signals in conserved ribosomal proteins
Eukaryotic ribosomal proteins contain nuclear localization signals (NLSs) that their bacterial counterparts lack. Here the authors compare homologous proteins from bacterial and eukaryotic ribosomes to show how NLSs could emerge in the course of evolution, and use this knowledge to identify novel NLSs.
- Sergey Melnikov
- , Adam Ben-Shem
- & Marat Yusupov
-
Article |
 Cytosolic targeting factor AKR2A captures chloroplast outer membrane-localized client proteins at the ribosome during translation
Cytosolic targeting factor AKR2A captures chloroplast outer membrane-localized client proteins at the ribosome during translation
Post-translational import of nuclear-encoded proteins shapes the proteome of organelles. Here, Kim et al.show that AKR2A, a critical targeting factor for chloroplast outer membrane proteins, binds to client proteins co-translationally as they exit the ribosome.
- Dae Heon Kim
- , Jae-Eun Lee
- & Inhwan Hwang
-
Article
| Open Access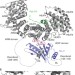 Symportin 1 chaperones 5S RNP assembly during ribosome biogenesis by occupying an essential rRNA-binding site
Symportin 1 chaperones 5S RNP assembly during ribosome biogenesis by occupying an essential rRNA-binding site
Biogenesis of the 80S ribosome involves more than 200 pre-ribosomal factors, which ensure the sequential assembly of ribosomal proteins and RNAs. Here the authors show that the nuclear transport adaptor Syo1 shields the 5S RNP-docking site on RpL11 before incorporation into the pre-60S through molecular mimicry.
- Fabiola R. Calviño
- , Satyavati Kharde
- & Irmgard Sinning
-
Article
| Open Access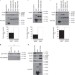 The intellectual disability protein RAB39B selectively regulates GluA2 trafficking to determine synaptic AMPAR composition
The intellectual disability protein RAB39B selectively regulates GluA2 trafficking to determine synaptic AMPAR composition
Mutations in the RAB39Bgene, which encodes a protein involved in vesicular trafficking, are associated with intellectual disability, but the impact of RAB39B loss of function on synaptic activity is not known. Here the authors show that RAB39B interacts with PICK1, and that this interaction is critical for the translocation of AMPA receptor subunits into the Golgi.
- Maria Lidia Mignogna
- , Maila Giannandrea
- & Patrizia D’Adamo
-
Article
| Open Access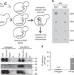 OM14 is a mitochondrial receptor for cytosolic ribosomes that supports co-translational import into mitochondria
OM14 is a mitochondrial receptor for cytosolic ribosomes that supports co-translational import into mitochondria
Mitochondrial proteins can be imported post-translationally; however, a role for co-translational import has recently provoked renewed interest. Lesnik et al.identify OM14 as a mitochondrial ribosome receptor required for efficient co-translational import of mitochondrial proteins.
- Chen Lesnik
- , Yifat Cohen
- & Yoav Arava
-
Article |
 Localized light-induced protein dimerization in living cells using a photocaged dimerizer
Localized light-induced protein dimerization in living cells using a photocaged dimerizer
Protein localization in cells can yield much information about the spatial arrangement of cellular processes and the participating groups. Here, the authors present a membrane-permeable and photoactive agent for localized protein dimerization in cells.
- Edward R. Ballister
- , Chanat Aonbangkhen
- & David M. Chenoweth
-
Article |
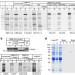 Versatile in vitro system to study translocation and functional integration of bacterial outer membrane proteins
Versatile in vitro system to study translocation and functional integration of bacterial outer membrane proteins
The mechanisms of protein translocation across and integration into bacterial outer membranes are poorly understood. Here, Norell et al. reconstitute type-V secretion and β-barrel protein biogenesis in proteoliposomes providing a versatile cell-free system to study integration and translocation.
- Derrick Norell
- , Alexander Heuck
- & Enguo Fan
-
Article |
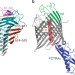 Translocation path of a substrate protein through its Omp85 transporter
Translocation path of a substrate protein through its Omp85 transporter
The two-partner secretion system transports proteins across the bacterial outer membrane but the molecular mechanisms involved are poorly understood. Here, Baud et al. use site-specific crosslinking to track the path of a protein substrate through the β-barrel of its Omp85 transporter.
- Catherine Baud
- , Jérémy Guérin
- & Françoise Jacob-Dubuisson
-
Article |
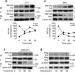 Receptor-interacting protein 140 attenuates endoplasmic reticulum stress in neurons and protects against cell death
Receptor-interacting protein 140 attenuates endoplasmic reticulum stress in neurons and protects against cell death
Uncontrolled calcium release from the endoplasmic reticulum results in cell death and toxicity. Here, the authors show that in neurons, stress induces the export of receptor-interacting protein 140 from the nucleus to the cytosol where it interacts with IP3Receptor, preventing its opening and the detrimental effects of calcium release.
- Xudong Feng
- , Kelly A. Krogh
- & Li-Na Wei
-
Article
| Open Access Retrieving the intracellular topology from multi-scale protein mobility mapping in living cells
Retrieving the intracellular topology from multi-scale protein mobility mapping in living cells
Numerous obstacles posed by cellular subcompartments and structures constrain protein transport in the cell. Here, Baum et al.map the intracellular topology from a diffusing protein’s point of view by measuring the diffusive movements of fluorescently labelled reporter proteins in living cells on multiple time and length scales.
- Michael Baum
- , Fabian Erdel
- & Karsten Rippe
-
Article |
 Remodelling of the active presequence translocase drives motor-dependent mitochondrial protein translocation
Remodelling of the active presequence translocase drives motor-dependent mitochondrial protein translocation
The ATPase activity of the chaperone Hsp70 drives protein translocation across the mitochondrial membrane. Schulz and Rehling show that continuous recruitment and dissociation of the Hsp70 ATPase activator Pam18 is required to regulate the activity of the import motor.
- Christian Schulz
- & Peter Rehling
-
Article |
 A mortise–tenon joint in the transmembrane domain modulates autotransporter assembly into bacterial outer membranes
A mortise–tenon joint in the transmembrane domain modulates autotransporter assembly into bacterial outer membranes
Bacterial autotransporters are folded in a process that entraps segments of their N-terminal passenger domain. Here, Leyton et al. identify glycine-aromatic mortise and tenon motifs that mediate the passenger domain’s translocation to the bacterial cell surface, and show that the motifs are evolutionarily conserved.
- Denisse L. Leyton
- , Matthew D. Johnson
- & Trevor Lithgow
-
Article
| Open Access Contact of cis-Golgi with ER exit sites executes cargo capture and delivery from the ER
Contact of cis-Golgi with ER exit sites executes cargo capture and delivery from the ER
Protein traffic from the endoplasmic reticulum (ER) to the Golgi is mediated by COPII-coated vesicles that bud from ER exit sites and fuse with the cis-Golgi. Kurokawa et al. show that in budding yeast, the cis-Golgi reaches out to ER exit sites in a ‘hug-and-kiss’ mechanism to facilitate cargo transfer.
- Kazuo Kurokawa
- , Michiyo Okamoto
- & Akihiko Nakano
-
Article |
 Bifurcation of the endocytic pathway into Rab5-dependent and -independent transport to the vacuole
Bifurcation of the endocytic pathway into Rab5-dependent and -independent transport to the vacuole
Proteins from the trans-Golgi network and the plasma membrane are both targeted to the vacuole in a Rab5-dependent manner; however, it remains unclear where these two routes converge. Toshima et al.show that convergence occurs upstream of Rab5, and in doing so discover a Rab5-independent endocytic pathway.
- Junko Y. Toshima
- , Show Nishinoaki
- & Jiro Toshima
-
Article |
 Structure of the mammalian oligosaccharyl-transferase complex in the native ER protein translocon
Structure of the mammalian oligosaccharyl-transferase complex in the native ER protein translocon
Proteins that are translocated across the endoplasmic reticulum (ER) membrane may be subject to glycosylation during transport. Using cryoelectron microscopy of native ER membranes, Pfeffer et al.map the location of oligosaccharyl-transferase within the translocon, providing insight into how these processes are coupled.
- Stefan Pfeffer
- , Johanna Dudek
- & Friedrich Förster
-
Article |
 CARMIL leading edge localization depends on a non-canonical PH domain and dimerization
CARMIL leading edge localization depends on a non-canonical PH domain and dimerization
CARMIL regulates actin capping proteins at the leading edge of migrating cells. By solving the crystal structure of the CARMIL N-terminal region, Zwolak et al. discover that this region encodes a cryptic pleckstrin homology domain, and show that it mediates association with the plasma membrane.
- Adam Zwolak
- , Changsong Yang
- & Roberto Dominguez
-
Article |
 Regulation of protein glycosylation and sorting by the Golgi matrix proteins GRASP55/65
Regulation of protein glycosylation and sorting by the Golgi matrix proteins GRASP55/65
GRASP proteins are thought to play a role in maintaining the stacked structure of the Golgi complex. Xiang et al. discover that depletion of GRASPs accelerates Golgi traffic, but reduces the complexity of Golgi protein glycosylation.
- Yi Xiang
- , Xiaoyan Zhang
- & Yanzhuang Wang
-
Article
| Open Access Spatial association with PTEX complexes defines regions for effector export into Plasmodium falciparum-infected erythrocytes
Spatial association with PTEX complexes defines regions for effector export into Plasmodium falciparum-infected erythrocytes
During red blood cell infection, malaria parasites export hundreds of proteins that remodel the host cell surface. Cowman and colleagues identify a putative protein translocator complex spatially associated with exported proteins, revealing the cellular domains involved in protein export.
- David T. Riglar
- , Kelly L. Rogers
- & Alan F. Cowman
-
Article
| Open Access Rab9 and retromer regulate retrograde trafficking of luminal protein required for epithelial tube length control
Rab9 and retromer regulate retrograde trafficking of luminal protein required for epithelial tube length control
The development of biological tubes is regulated by mutual interactions between cells and luminal extracellular matrix. Donget al. show that retrograde recycling of luminal chitin deacetylase regulates Drosophila tracheal tubule geometry by restricting length independently of diameter.
- Bo Dong
- , Ken Kakihara
- & Shigeo Hayashi
-
Article
| Open Access Transmembrane insertion of twin-arginine signal peptides is driven by TatC and regulated by TatB
Transmembrane insertion of twin-arginine signal peptides is driven by TatC and regulated by TatB
TatA, B and C act together to translocate folded proteins across bacterial and chloroplast membranes, however the precise mechanism remains unclear. Fröbel and colleagues discover that TatC has unforeseen membrane insertase activity, while TatB prevents premature cleavage before translocation.
- Julia Fröbel
- , Patrick Rose
- & Matthias Müller
-
Article
| Open Access MPIase is a glycolipozyme essential for membrane protein integration
MPIase is a glycolipozyme essential for membrane protein integration
Proteins are integrated into cellular membranes either co-translationally through Sec/SRP or post-translationally by chaperones. These authors show that an integration-dedicated chaperone inE. coli, MPIase, is a glycolipid and facilitates protein insertion into the inner membrane of the bacterium.
- Ken-ichi Nishiyama
- , Masahide Maeda
- & Keiko Shimamoto

 Structural assembly of the megadalton-sized receptor for intestinal vitamin B12 uptake and kidney protein reabsorption
Structural assembly of the megadalton-sized receptor for intestinal vitamin B12 uptake and kidney protein reabsorption