Featured
-
-
Research Highlights |
Prion-like immune trigger
-
Letter |
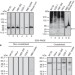 α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation
α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation
- Tim Bartels
- , Joanna G. Choi
- & Dennis J. Selkoe
-
Review Article |
 Molecular chaperones in protein folding and proteostasis
Molecular chaperones in protein folding and proteostasis
- F. Ulrich Hartl
- , Andreas Bracher
- & Manajit Hayer-Hartl
-
Outlook |
 Amyloid: Little proteins, big clues
Amyloid: Little proteins, big clues
After a quarter of a century, the amyloid hypothesis for Alzheimer's disease is reconnecting to its roots in prion research.
- Jim Schnabel
-
Letter |
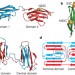 Single-molecule fluorescence reveals sequence-specific misfolding in multidomain proteins
Single-molecule fluorescence reveals sequence-specific misfolding in multidomain proteins
- Madeleine B. Borgia
- , Alessandro Borgia
- & Jane Clarke
-
Letter |
 Structural basis for recognition of centromere histone variant CenH3 by the chaperone Scm3
Structural basis for recognition of centromere histone variant CenH3 by the chaperone Scm3
- Zheng Zhou
- , Hanqiao Feng
- & Yawen Bai
-
News & Views |
 Protection from the outside
Protection from the outside
Protein folding is a high-stakes process, with cell dysfunction and death being the unforgiving penalties for failure. Work in bacteria hints that organisms manage this process beyond the boundaries of the cytoplasm — and even the cell.
- Evan T. Powers
- & William E. Balch
-
Research Highlights |
Toxic clumps trap many proteins
-
News & Views |
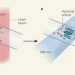 Weighing up protein folding
Weighing up protein folding
Labelling molecules by fast oxidation allows mass spectrometry to study protein folding at submillisecond time resolution. The method also brings a wealth of structural information about protein folding within reach.
- Martin Gruebele
-
News |
 Supercomputer sets protein-folding record
Supercomputer sets protein-folding record
Faster simulations follow protein movements for longer.
- Heidi Ledford
-
Research Highlights |
Ageing: Proteins clump with age
-
Brief Communications Arising |
Laurén et al. reply
- Juha Laurén
- , David A. Gimbel
- & Stephen M. Strittmatter
-
News Feature |
 Citizen science: People power
Citizen science: People power
Networks of human minds are taking citizen science to a new level, reports Eric Hand.
- Eric Hand
-
Letter |
 The folding cooperativity of a protein is controlled by its chain topology
The folding cooperativity of a protein is controlled by its chain topology
Proteins often comprise domains that can be distinguished as relatively separate regions in the three-dimensional structure. Communication between these domains is important for catalysis, regulation and folding, but how they communicate is largely unclear. Here, single-molecule optical tweezers were used to pull on a protein while monitoring the energetics of unfolding and refolding events in disparate regions. By comparing topological variations of the same protein, new rules of cooperation between domains were derived.
- Elizabeth A. Shank
- , Ciro Cecconi
- & Carlos Bustamante
-
Research Highlights |
Pathology: Bent out of shape
-
Research Highlights |
Neurobiology: Entangled diseases
-
News Feature |
 Protein folding: The dark side of proteins
Protein folding: The dark side of proteins
Almost every human protein has segments that can form amyloids, the sticky aggregates known for their role in disease. Yet cells have evolved some elaborate defences, finds Jim Schnabel.
- Jim Schnabel
-
Research Highlights |
Cell biology: Lost in the mail
-
Letter |
 Experimental evidence for a frustrated energy landscape in a three-helix-bundle protein family
Experimental evidence for a frustrated energy landscape in a three-helix-bundle protein family
The primary sequence of a protein defines its free-energy landscape and thus determines the rate constants of folding and unfolding, with theory suggesting that roughness in the energy landscape leads to slower folding. However, obtaining experimental descriptions of this landscape is challenging. Landscape roughness is now shown to be responsible for the slower folding and unfolding times observed in the R16 and R17 domains of α-spectrin relative to the similar R15 domain.
- Beth G. Wensley
- , Sarah Batey
- & Jane Clarke
-
Research Highlights |
Neurobiology: Prions at work
-
News |
 Healthy prions protect nerves
Healthy prions protect nerves
The proteins that can cause CJD have a vital role in the nervous system.
- Alison Abbott
-
Letter |
 Mechanism of folding chamber closure in a group II chaperonin
Mechanism of folding chamber closure in a group II chaperonin
Group II chaperonins are present in eukaryotes and archaea and are essential mediators of cellular protein folding. This process is critically dependent on the closure of a built-in lid, which is triggered by ATP hydrolysis, but the structural rearrangements and molecular events leading to lid closure are unknown. Using cryo-electron microscopy, the structures of an archaeal group II chaperonin in the open and closed states are now reported, providing details of this mechanism.
- Junjie Zhang
- , Matthew L. Baker
- & Wah Chiu
-
Article |
 Coupled chaperone action in folding and assembly of hexadecameric Rubisco
Coupled chaperone action in folding and assembly of hexadecameric Rubisco
Form I Rubisco, one of the most abundant proteins in nature, catalyses the fixation of atmospheric CO2 in photosynthesis. The limited catalytic efficiency of Rubisco has sparked extensive efforts to re-engineer the enzyme to enhance agricultural productivity. To bring this goal closer, the formation of cyanobacterial form I Rubisco is now analysed by in vitro reconstitution and cryo-electron microscopy.
- Cuimin Liu
- , Anna L. Young
- & Manajit Hayer-Hartl

 Excitation-induced ataxin-3 aggregation in neurons from patients with Machado–Joseph disease
Excitation-induced ataxin-3 aggregation in neurons from patients with Machado–Joseph disease