Article
|
Open Access
Featured
-
-
Article |
 REGγ is critical for skin carcinogenesis by modulating the Wnt/β-catenin pathway
REGγ is critical for skin carcinogenesis by modulating the Wnt/β-catenin pathway
REGγ, a proteasome activator, is associated with multiple oncogenic pathways in human cancers and can promote the degradation of multiple proteins including p53. Here the authors highlight a potential role for REGγ in skin cancer and propose a molecular mechanism linking p38 MAPK and Wnt signalling.
- Lei Li
- , Yongyan Dang
- & Xiaotao Li
-
Article |
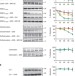 The Parkinson’s-associated protein DJ-1 regulates the 20S proteasome
The Parkinson’s-associated protein DJ-1 regulates the 20S proteasome
Mutations in the gene encoding DJ-1 are associated with early-onset familial forms of Parkinson’s disease, and several different molecular functions have been attributed to this protein. Moscovitz et al.show that DJ-1 physically binds the 20S proteasome and inhibits its degradation activity.
- Oren Moscovitz
- , Gili Ben-Nissan
- & Michal Sharon
-
Article |
 Maturation of the proteasome core particle induces an affinity switch that controls regulatory particle association
Maturation of the proteasome core particle induces an affinity switch that controls regulatory particle association
Proteasome assembly requires the coordinated association of over sixty proteins and is dependent on chaperones. Wani et al.show that the core particle undergoes a maturation-associated decrease in its affinity for the chaperone Pba1-Pba2 that ensures timely association with the regulatory particle.
- Prashant S. Wani
- , Michael A. Rowland
- & Jeroen Roelofs
-
Article |
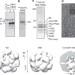 Proteasome assembly from 15S precursors involves major conformational changes and recycling of the Pba1–Pba2 chaperone
Proteasome assembly from 15S precursors involves major conformational changes and recycling of the Pba1–Pba2 chaperone
The barrel-shaped 20S proteasome core particle assembles via 15S intermediates through the action of Ump1 and Pba1–Pba2 chaperones. Using structural approaches, Kock et al. reveal conformational changes occurring upon formation of the nascent 20S particle leading to ejection of Pba1–Pba2.
- Malte Kock
- , Maria M. Nunes
- & Petra Wendler
-
Review Article |
 The role of protein clearance mechanisms in organismal ageing and age-related diseases
The role of protein clearance mechanisms in organismal ageing and age-related diseases
Proteins are subject to continuous and complex quality-control mechanisms, which ensure integrity of the proteome. Vilchez et al.review how a demise in these processes, collectively referred to as proteostasis, is linked to organismal ageing and the development of age-associated diseases.
- David Vilchez
- , Isabel Saez
- & Andrew Dillin
-
Article |
 Direct cellular delivery of human proteasomes to delay tau aggregation
Direct cellular delivery of human proteasomes to delay tau aggregation
Increasing the activity of the proteasome can prevent the accumulation of protein aggregates within the cell. Han et al.show that nanoparticle-mediated delivery of purified proteasomes to cells reduces proteotoxic stress resulting from tau overexpression, and prevents to accumulation of tau aggregates.
- Dong Hoon Han
- , Hee-Kyung Na
- & Min Jae Lee
-
Article
| Open Access Ubiquitin-proteasome system controls ciliogenesis at the initial step of axoneme extension
Ubiquitin-proteasome system controls ciliogenesis at the initial step of axoneme extension
Biogenesis of the primary cilium begins after cell cycle exit, but the regulatory steps for its formation are poorly defined. Here the authors show that proteasome-mediated removal of the ciliogenesis inhibitor, trichoplein, from mother centrioles initiates the first step of ciliogenesis.
- Kousuke Kasahara
- , Yoshitaka Kawakami
- & Masaki Inagaki
-
Article |
 Sensitive detection of proteasomal activation using the Deg-On mammalian synthetic gene circuit
Sensitive detection of proteasomal activation using the Deg-On mammalian synthetic gene circuit
Proteasome activity can be monitored using a fluorescent substrate; however, screening for proteasome activators using this technique is challenging as signal loss is intrinsically more difficult to detect. Zhao et al.design a genetic inverter circuit that reports increased proteasome activity as signal gain.
- Wenting Zhao
- , Matthew Bonem
- & Laura Segatori
-
Article |
 The 19S proteasome subunit Rpt3 regulates distribution of CENP-A by associating with centromeric chromatin
The 19S proteasome subunit Rpt3 regulates distribution of CENP-A by associating with centromeric chromatin
The histone variant CENP-A has a crucial role in defining centromeric chromatin and is required for accurate segregation of chromosomes during mitosis. Here, Kitagawa et al.identify Rpt3, a subunit of the 19S proteasome, as a novel factor regulating CENP localization.
- Teppei Kitagawa
- , Kojiro Ishii
- & Tomohiro Matsumoto
-
Article |
 Quantitative live-cell imaging reveals spatio-temporal dynamics and cytoplasmic assembly of the 26S proteasome
Quantitative live-cell imaging reveals spatio-temporal dynamics and cytoplasmic assembly of the 26S proteasome
The 26S proteasome is assembled in several steps, however the extent to which this assembly occurs before or after transport into the nucleus remains unclear. Pack et al.show that full assembly can occur in the cytoplasm, and that a concatameric form of the fully assembled complex is a substrate for nuclear import.
- Chan-Gi Pack
- , Haruka Yukii
- & Yasushi Saeki
-
Article |
 Proteasomal degradation of Nck1 but not Nck2 regulates RhoA activation and actin dynamics
Proteasomal degradation of Nck1 but not Nck2 regulates RhoA activation and actin dynamics
Nck1/2 adaptor proteins control the assembly and activity of protein complexes that promote actin polymerization. Buvall et al.show that Nck1 abundance is regulated by the E3 ubiquitin ligase c-Cbl, which competes for Nck binding with the actin-binding protein synaptopodin in podocytes.
- Lisa Buvall
- , Priyanka Rashmi
- & Peter Mundel
-
Article
| Open Access Differential regulation of the REGγ–proteasome pathway by p53/TGF-β signalling and mutant p53 in cancer cells
Differential regulation of the REGγ–proteasome pathway by p53/TGF-β signalling and mutant p53 in cancer cells
REGγ is a proteasome activator and is frequently overexpressed in cancer cells. Here Ali et al. demonstrate that p53/TGF-β signalling inhibits REGγ expression, whereas p53 mutations increase REGγ transcription, identifying a gain of function for mutant p53.
- Amjad Ali
- , Zhuo Wang
- & Xiaotao Li
-
Article |
 Involvement of Bag6 and the TRC pathway in proteasome assembly
Involvement of Bag6 and the TRC pathway in proteasome assembly
The 26S proteasome comprises over 33 different subunits that must be correctly assembled by dedicated chaperones for efficient protein degradation. Here the authors find that general chaperone proteins are also vital for proper proteasome assembly.
- Takashi Akahane
- , Kazutaka Sahara
- & Shigeo Murata
-
Article |
 FAT10 and NUB1L bind to the VWA domain of Rpn10 and Rpn1 to enable proteasome-mediated proteolysis
FAT10 and NUB1L bind to the VWA domain of Rpn10 and Rpn1 to enable proteasome-mediated proteolysis
FAT-10 targets proteins for degradation in a ubiquitin-independent manner and NUB1L accelerates this process. In this study, both FAT-10 and NUB1L are shown to interact with the VWA domain of the Rpn10 subunit of the 26S proteasome, suggesting that this is a receptor site for ubiquitin-like modifiers.
- Neha Rani
- , Annette Aichem
- & Marcus Groettrup
-
Article |
 A photoconvertible fluorescent reporter to track chaperone-mediated autophagy
A photoconvertible fluorescent reporter to track chaperone-mediated autophagy
Soluble cytosolic proteins can be degraded in lysosomes by chaperone-mediated autophagy, however, the current method to measure this process requires isolation of lysosomes. Now, a fluorescent reporter is described that can measure this type of autophagy in intact cells.
- Hiroshi Koga
- , Marta Martinez-Vicente
- & Ana Maria Cuervo
-
Article |
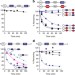 Rad23 escapes degradation because it lacks a proteasome initiation region
Rad23 escapes degradation because it lacks a proteasome initiation region
Rad23 accompanies ubiquitinated substrates to the proteasome for destruction but manages to avoid degradation. In this study, Fishbainet al.show that Rad23 escapes because it lacks an effective initiation region; therefore, the proteasome is unable to engage the protein and unfold it.
- Susan Fishbain
- , Sumit Prakash
- & Andreas Matouschek
-
Article
| Open Access C-terminal UBA domains protect ubiquitin receptors by preventing initiation of protein degradation
C-terminal UBA domains protect ubiquitin receptors by preventing initiation of protein degradation
Rad23 and Dsk2 bind polyubiquitylated proteins and escort them to the proteasome for destruction. In this study, Heinenet al.investigate the molecular mechanisms that protect the C-terminal UBA domains of Rad23 and Dsk2 from proteasomal destruction.
- Christian Heinen
- , Klàra Ács
- & Nico P. Dantuma

 Thymoproteasomes produce unique peptide motifs for positive selection of CD8+ T cells
Thymoproteasomes produce unique peptide motifs for positive selection of CD8+ T cells