Featured
-
-
Article
| Open Access CENP-E activation by Aurora A and B controls kinetochore fibrous corona disassembly
CENP-E activation by Aurora A and B controls kinetochore fibrous corona disassembly
It is unknown how the kinetochore fibrous corona is disassembled. Here, the authors reveal that Aurora A and B kinases-mediated phosphorylation activates CENP-E, which is essential to prevent the premature removal of corona proteins by dynein.
- Susana Eibes
- , Girish Rajendraprasad
- & Marin Barisic
-
Article
| Open Access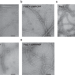 Structures of a FtsZ single protofilament and a double-helical tube in complex with a monobody
Structures of a FtsZ single protofilament and a double-helical tube in complex with a monobody
The cryoEM structures of a single protofilament of FtsZ from Klebsiella pneumoniae (KpFtsZ) in a polymerization-preferred conformation are presented and of a double-helical tube of the FtsZ–monobody complex that shows two parallel protofilaments.
- Junso Fujita
- , Hiroshi Amesaka
- & Hiroyoshi Matsumura
-
Article
| Open Access Kinetochore component function in C. elegans oocytes revealed by 4D tracking of holocentric chromosomes
Kinetochore component function in C. elegans oocytes revealed by 4D tracking of holocentric chromosomes
The exact function of kinetochore proteins in meiosis remains unclear. Using live imaging of C. elegans oocytes, the authors systematically study the contribution of each kinetochore sub-complex and describe a push-pull mechanism that confers robustness to chromosome segregation.
- Laras Pitayu-Nugroho
- , Mélanie Aubry
- & Julien Dumont
-
Article
| Open Access Rio1 downregulates centromeric RNA levels to promote the timely assembly of structurally fit kinetochores
Rio1 downregulates centromeric RNA levels to promote the timely assembly of structurally fit kinetochores
Kinetochores assemble on centromeres via histone H3 variant CENP-A and low levels of centromere transcripts (cenRNAs). Here the authors show the Rio1 kinase limits cenRNA production by reducing RNAPII accessibility and promotes cenRNA degradation by the 5’− 3’exoribonuclease Rat1.
- Ksenia Smurova
- , Michela Damizia
- & Peter De Wulf
-
Article
| Open Access Tension can directly suppress Aurora B kinase-triggered release of kinetochore-microtubule attachments
Tension can directly suppress Aurora B kinase-triggered release of kinetochore-microtubule attachments
Tension stabilizes properly attached microtubules to kinetochores during chromosome segregation, and lack of tension leads to release. Here the authors show that tension directly suppresses Aurora B kinase mediated destabilization of reconstituted kinetochore-microtubule attachments, likely ensuring accurate chromosome segregation.
- Anna K. de Regt
- , Cordell J. Clark
- & Sue Biggins
-
Article
| Open Access Recruitment of two Ndc80 complexes via the CENP-T pathway is sufficient for kinetochore functions
Recruitment of two Ndc80 complexes via the CENP-T pathway is sufficient for kinetochore functions
The kinetochores contain multiple protein interaction networks. Takenoshita et al. analyzed the complicated networks using the genetic method and revealed that two copies of Ndc80 complexes on CENP-T are sufficient for kinetochore functions.
- Yusuke Takenoshita
- , Masatoshi Hara
- & Tatsuo Fukagawa
-
Article
| Open Access Counteraction between Astrin-PP1 and Cyclin-B-CDK1 pathways protects chromosome-microtubule attachments independent of biorientation
Counteraction between Astrin-PP1 and Cyclin-B-CDK1 pathways protects chromosome-microtubule attachments independent of biorientation
Chromosome instability frequently occurs due to issues with chromosome-microtubule attachments. Here the authors show that the Astrin-PP1 and Cyclin-B-CDK1 pathways counteract each other to protect chromosome-microtubule attachments independent of biorientation.
- Xinhong Song
- , Duccio Conti
- & Viji M. Draviam
-
Article
| Open Access CENP-V is required for proper chromosome segregation through interaction with spindle microtubules in mouse oocytes
CENP-V is required for proper chromosome segregation through interaction with spindle microtubules in mouse oocytes
Chromosome segregation is essential to avoid aneuploidy, yet in mammalian oocytes it progressively fails in an age-dependent manner. Here the authors identify CENP-V as a microtubule binding and bundling protein crucial to faithful oocyte meiosis, and present Cenp-V−/− oocytes as revealing age-dependent weakening of the spindle assembly checkpoint.
- Dalileh Nabi
- , Hauke Drechsler
- & Mariola Chacón
-
Article
| Open Access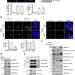 Spatiotemporal coordination of the RSF1-PLK1-Aurora B cascade establishes mitotic signaling platforms
Spatiotemporal coordination of the RSF1-PLK1-Aurora B cascade establishes mitotic signaling platforms
During cell division, chromosome alignment is engendered by connection of microtubules to kinetochores, coordinated by Aurora B and PLK1. Here, the authors show that the RSF1-PLK1 axis creates an activating phosphorylation on T236 in the GT motif of Aurora B and this is indispensable for Aurora B activation.
- Ho-Soo Lee
- , Sunwoo Min
- & Hyeseong Cho
-
Article
| Open Access Bridgin connects the outer kinetochore to centromeric chromatin
Bridgin connects the outer kinetochore to centromeric chromatin
The kinetochore is a multi-complex structure that helps attach chromosomes to spindle microtubules, ensuring accurate chromosome segregation during cell division. Kinetochores are thought to be evolutionarily conserved, but which components are conserved is unclear. Here, the authors report that some members of the fungal phylum of Basidomycota lack many conventional kinetochore linker proteins. Instead, they possess a human Ki67-like protein that bridges the outer part of the kinetochore to centromere DNA, which may compensate for the loss of a conventional linker.
- Shreyas Sridhar
- , Tetsuya Hori
- & Kaustuv Sanyal
-
Article
| Open Access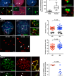 Helicase LSH/Hells regulates kinetochore function, histone H3/Thr3 phosphorylation and centromere transcription during oocyte meiosis
Helicase LSH/Hells regulates kinetochore function, histone H3/Thr3 phosphorylation and centromere transcription during oocyte meiosis
Centromeres are the sites of kinetochore and inner centromere formation, which can be epigenetically regulated. Here, the authors reveal a role for the lymphocyte specific helicase LSH/Hells associated with pericentric heterochromatin formation in centromere stability and chromosome segregation at meiotic kinetochores.
- Claudia Baumann
- , Wei Ma
- & Rabindranath De La Fuente
-
Article
| Open Access Mechanisms of microtubule dynamics and force generation examined with computational modeling and electron cryotomography
Mechanisms of microtubule dynamics and force generation examined with computational modeling and electron cryotomography
Microtubules are dynamic tubulin polymers which elongate by addition of bent guanosine triphosphate tubulin to the tips of curving protofilaments. Here authors use Brownian dynamics modeling and electron cryotomography to show that the lateral activation energy barrier in tubulin-tubulin interactions is a key parameter for this process, controlling the development of high pulling forces.
- Nikita B. Gudimchuk
- , Evgeni V. Ulyanov
- & J. Richard McIntosh
-
Article
| Open Access Long noncoding RNA SAM promotes myoblast proliferation through stabilizing Sugt1 and facilitating kinetochore assembly
Long noncoding RNA SAM promotes myoblast proliferation through stabilizing Sugt1 and facilitating kinetochore assembly
Long noncoding RNA SAM (Sugt1 associated muscle) is upregulated in the proliferating myoblast cells. Here the authors investigate SAM knockout mice and suggest that SAM binds and stabilizes Sugt1, a co-chaperone protein that regulates kinetochore assembly.
- Yuying Li
- , Jie Yuan
- & Huating Wang
-
Article
| Open Access Proper chromosome alignment depends on BRCA2 phosphorylation by PLK1
Proper chromosome alignment depends on BRCA2 phosphorylation by PLK1
The BRCA2 tumour suppressor protein is known to play an important role in homologous recombination. Here the authors reveal how the phosphorylation of BRCA2 by Polo-like kinase 1 (PLK1) contributes to the regulation of mitosis.
- Åsa Ehlén
- , Charlotte Martin
- & Aura Carreira
-
Article
| Open Access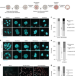 Tetraploidy causes chromosomal instability in acentriolar mouse embryos
Tetraploidy causes chromosomal instability in acentriolar mouse embryos
During cell division, tetraploidy can drive chromosomal instability (CIN) via supernumerary centrosomes, but it is unclear if this is the only route to CIN. Here the authors show that, in early mouse embryos, tetraploidy can drive chromosomal instability by altering microtubule dynamics and attachment.
- Lia Mara Gomes Paim
- & Greg FitzHarris
-
Article
| Open Access Mechanism of centromere recruitment of the CENP-A chaperone HJURP and its implications for centromere licensing
Mechanism of centromere recruitment of the CENP-A chaperone HJURP and its implications for centromere licensing
The CENP-A chaperone HJURP associates with Mis18α, Mis18β, and M18BP1 to target centromeres and deposit new CENP-A. Here the authors provide evidence that two repeats in human HJURP previously proposed to be functionally distinct are interchangeable and bind concomitantly to the 4:2:2 Mis18α:Mis18β:M18BP1 complex without dissociating it.
- Dongqing Pan
- , Kai Walstein
- & Andrea Musacchio
-
Article
| Open Access The poly-SUMO2/3 protease SENP6 enables assembly of the constitutive centromere-associated network by group deSUMOylation
The poly-SUMO2/3 protease SENP6 enables assembly of the constitutive centromere-associated network by group deSUMOylation
While the biological roles of ubiquitin chains are well studied, little is known about the functions of SUMO polymers. Here, the authors identify poly-SUMOylation substrates and provide evidence that SUMO polymers regulate the accumulation of CCAN subunits at chromatin and centromeres.
- Frauke Liebelt
- , Nicolette S. Jansen
- & Alfred C. O. Vertegaal
-
Article
| Open Access Microtubule end conversion mediated by motors and diffusing proteins with no intrinsic microtubule end-binding activity
Microtubule end conversion mediated by motors and diffusing proteins with no intrinsic microtubule end-binding activity
During cell division, it is currently unclear how kinetochores transit from lateral microtubule attachment to durable association to dynamic microtubule plus ends. Here, using in vitro reconstitution and computer modeling, the authors provide biophysical mechanism for microtubule end-conversion driven by two kinetochore components, CENP-E and Ndc80 complex
- Manas Chakraborty
- , Ekaterina V. Tarasovetc
- & Ekaterina L. Grishchuk
-
Article
| Open Access Cdk1-mediated DIAPH1 phosphorylation maintains metaphase cortical tension and inactivates the spindle assembly checkpoint at anaphase
Cdk1-mediated DIAPH1 phosphorylation maintains metaphase cortical tension and inactivates the spindle assembly checkpoint at anaphase
Cell rounding at mitosis is driven by cortical tension and maintained through metaphase, although the mechanism is unknown. Here, the authors demonstrate that Cdk1 phosphorylation of DIAPH1 is required for both cortical tension maintenance and inactivation of the spindle assembly checkpoint.
- Koutarou Nishimura
- , Yoshikazu Johmura
- & Makoto Nakanishi
-
Article
| Open Access The binding of Borealin to microtubules underlies a tension independent kinetochore-microtubule error correction pathway
The binding of Borealin to microtubules underlies a tension independent kinetochore-microtubule error correction pathway
How the chromosome passenger complex (CPC) phosphorylates the kinetochores that can be a micron away to control mitotic events is unknown. Here the authors find that the CPC directly binds microtubules near inner centromeres, which controls its ability to phosphorylate kinetochores independently of tension generated by kinetochore microtubule attachments.
- Prasad Trivedi
- , Anatoly V. Zaytsev
- & P. Todd Stukenberg
-
Article
| Open Access Complete microtubule–kinetochore occupancy favours the segregation of merotelic attachments
Complete microtubule–kinetochore occupancy favours the segregation of merotelic attachments
Single microtubules (MTs) can move chromosomes, but it is unclear why kinetochores bind up to 20 MTs. Here, the authors decrease the number of kinetochore MTs with BAL27862 and see lagging chromosomes, suggesting that numerous kinetochore MTs provide force ensuring robust chromosomal segregation.
- Damian Dudka
- , Anna Noatynska
- & Patrick Meraldi
-
Article
| Open Access Unattached kinetochores drive their own capturing by sequestering a CLASP
Unattached kinetochores drive their own capturing by sequestering a CLASP
Kinetochores (KT) that are not attached to microtubules prevent chromosome missegregation via the spindle assembly checkpoint. Here the authors show that Mps1 localizes Stu1 at unattached KTs together with Slk19, causing a reorganization of the nuclear MT network that favors the capturing of unattached KT.
- Caroline Kolenda
- , Jennifer Ortiz
- & Johannes Lechner
-
Article
| Open Access Bub1 positions Mad1 close to KNL1 MELT repeats to promote checkpoint signalling
Bub1 positions Mad1 close to KNL1 MELT repeats to promote checkpoint signalling
The spindle assembly checkpoint ensures correct chromosome segregation and relies on kinetochore localization of the Bub1 and Mad1/Mad2 checkpoint proteins. Here the authors show that main function of Bub1 is to position Mad1 close to KNL1 MELT repeats in human cells.
- Gang Zhang
- , Thomas Kruse
- & Jakob Nilsson
-
Article
| Open Access Maternal age-dependent APC/C-mediated decrease in securin causes premature sister chromatid separation in meiosis II
Maternal age-dependent APC/C-mediated decrease in securin causes premature sister chromatid separation in meiosis II
Sister chromatid cohesion during meiosis II (MII), maintained by securin-mediated inhibition of separase, is reduced in aged mouse oocytes. Here the authors show that, in MII oocytes, securin levels are reduced by increased destruction by the anaphase promoting complex/cyclosome.
- Ibtissem Nabti
- , Rosanna Grimes
- & John Carroll
-
Article
| Open Access C. elegans chromosomes connect to centrosomes by anchoring into the spindle network
C. elegans chromosomes connect to centrosomes by anchoring into the spindle network
A connection between centrosomes and chromosomes is a key feature of mitotic spindles. Here the authors generate 3D reconstructions of whole mitotic spindles in earlyC. elegansembryos and show that chromosomes are anchored by the entire spindle network and that connections through kinetochore microtubules are few and likely very transient.
- Stefanie Redemann
- , Johannes Baumgart
- & Thomas Müller-Reichert
-
Article
| Open Access Epigenetic engineering reveals a balance between histone modifications and transcription in kinetochore maintenance
Epigenetic engineering reveals a balance between histone modifications and transcription in kinetochore maintenance
Centromeres are centrochromatin domains with CENP-A and H3 nucleosomes carrying transcription-associated modifications. Here the authors target synthetic modules to the centromeres to show that transcription plus histone modifications are required for CENP-A assembly and centrochromatin maintenance.
- Oscar Molina
- , Giulia Vargiu
- & William C. Earnshaw
-
Article
| Open Access Acetylation of histone H4 lysine 5 and 12 is required for CENP-A deposition into centromeres
Acetylation of histone H4 lysine 5 and 12 is required for CENP-A deposition into centromeres
The deposition of histone H3 variant CENP-A bound with histone H4 is a key feature designating the centromere region of a chromosome. Here the authors show acetylation on residues K5 and K12 in histone H4, mediated by the RbAp46/48-Hat1 complex, is required for deposition of CENP-A-H4 into centromeres.
- Wei-Hao Shang
- , Tetsuya Hori
- & Tatsuo Fukagawa
-
Article
| Open Access Chromosome biorientation produces hundreds of piconewtons at a metazoan kinetochore
Chromosome biorientation produces hundreds of piconewtons at a metazoan kinetochore
Chromosomes bind microtubules (MT) from opposite spindle poles and the generated tension stabilizes kinetochore-MT attachments. Here the authors measure kinetochore forces by engineering two force sensors and propose that kinetochore fibers exert hundreds of pNs of force to bioriented kinetochores.
- Anna A. Ye
- , Stuart Cane
- & Thomas J. Maresca
-
Article
| Open Access Two functionally distinct kinetochore pools of BubR1 ensure accurate chromosome segregation
Two functionally distinct kinetochore pools of BubR1 ensure accurate chromosome segregation
The BubR1/Bub3 complex regulates chromosome segregation to enable proper kinetochore-microtubule interactions and is also required for the spindle assembly checkpoint. Here the authors show that two distinct pools of BubR1/Bub3 exist at kinetochores to support both known functions of BubR1/Bub3.
- Gang Zhang
- , Blanca Lopez Mendez
- & Jakob Nilsson
-
Article
| Open Access Molecular requirements for the inter-subunit interaction and kinetochore recruitment of SKAP and Astrin
Molecular requirements for the inter-subunit interaction and kinetochore recruitment of SKAP and Astrin
SKAP and Astrin form a heterodimer that localizes to spindle microtubules and to mature microtubule-kinetochore attachments during mitosis. Here, the authors identify molecular requirements for the inter-subunit interaction of SKAP and Astrin and kinetochore recruitment.
- Alexandra Friese
- , Alex C. Faesen
- & Andrea Musacchio
-
Article
| Open Access Cep57 is a Mis12-interacting kinetochore protein involved in kinetochore targeting of Mad1–Mad2
Cep57 is a Mis12-interacting kinetochore protein involved in kinetochore targeting of Mad1–Mad2
The spindle assembly checkpoint relies on the accumulation of Mad1-Mad2 at kinetochores, but the mechanism of regulation is not known. Here Zhou et al. show that the centrosomal protein Cep57 interacts with the kinetochore proteins Mis12 and Mad1, and regulates the recruitment of Mad1/Mad2 to kinetochores.
- Haining Zhou
- , Tianning Wang
- & Jianguo Chen
-
Article
| Open Access Overlap microtubules link sister k-fibres and balance the forces on bi-oriented kinetochores
Overlap microtubules link sister k-fibres and balance the forces on bi-oriented kinetochores
During metaphase, k-fibre microtubules exert force on kinetochores, but there are also non-kinetochore microtubules close to kinetochores without a known function. Here the authors show that these microtubules, which they call bridging fibres, balance interkinetochore tension by bridging sister k-fibres.
- Janko Kajtez
- , Anastasia Solomatina
- & Iva M. Tolić
-
Article
| Open Access Kinetochore–microtubule attachment is sufficient to satisfy the human spindle assembly checkpoint
Kinetochore–microtubule attachment is sufficient to satisfy the human spindle assembly checkpoint
The spindle assembly checkpoint protects against premature chromosome segregation during mitosis but it is not known whether microtubule attachment to the kinetochore, or force generated from this interaction, is being monitored. Here the authors uncouple these processes and show that microtubule attachment is sufficient to satisfy the checkpoint.
- Banafsheh Etemad
- , Timo E. F. Kuijt
- & Geert J. P. L. Kops
-
Article
| Open Access Stable kinetochore–microtubule attachment is sufficient to silence the spindle assembly checkpoint in human cells
Stable kinetochore–microtubule attachment is sufficient to silence the spindle assembly checkpoint in human cells
The spindle assembly checkpoint prevents mitotic progression when chromosomes are not properly attached to the mitotic spindle. Here Tauchman et al.show that stable microtubule attachment to the kinetochore, and not tension generated from this interaction, is sufficient to silence the checkpoint.
- Eric C. Tauchman
- , Frederick J. Boehm
- & Jennifer G. DeLuca
-
Article
| Open Access Bub1 autophosphorylation feeds back to regulate kinetochore docking and promote localized substrate phosphorylation
Bub1 autophosphorylation feeds back to regulate kinetochore docking and promote localized substrate phosphorylation
Bub1 kinase phosphorylates histone H2A-T120 at the centromere to recruit shugoshin proteins and promote sister chromatid cohesion during mitosis. Here the authors show that Bub1 autophosphorylation on T589 influences Bub1 dynamics at the kinetochore and restricts H2A-T120 phosphorylation to centromeres.
- Adeel Asghar
- , Audrey Lajeunesse
- & Sabine Elowe
-
Article
| Open Access A quantitative description of Ndc80 complex linkage to human kinetochores
A quantitative description of Ndc80 complex linkage to human kinetochores
The Ndc80 complex acts as an interface between kinetochores and spindle microtubules, and is recruited to the kinetochore by several different proteins. Suzukiet al. use protein counting techniques and RNA interference to quantify the interaction stoichiometries within this intricate cellular structure.
- Aussie Suzuki
- , Benjamin L. Badger
- & Edward D. Salmon
-
Article
| Open Access The chromatin remodeller RSF1 is essential for PLK1 deposition and function at mitotic kinetochores
The chromatin remodeller RSF1 is essential for PLK1 deposition and function at mitotic kinetochores
Polo-like kinase 1 (PLK1) is recruited to kinetochores during mitosis, where it is required for proper chromosome alignment. Leeet al. show that the chromatin-remodelling factor RSF1 is required for PLK1 recruitment, and that this function depends on phosphorylation of RSF1 by the mitotic kinase CDK1.
- Ho-Soo Lee
- , Yong-Yea Park
- & Hyeseong Cho
-
Article
| Open Access Distinct domains in Bub1 localize RZZ and BubR1 to kinetochores to regulate the checkpoint
Distinct domains in Bub1 localize RZZ and BubR1 to kinetochores to regulate the checkpoint
The spindle assembly checkpoint (SAC) depends on the recruitment of specific protein complexes to the kinetochore. Here Zhang et al. show that Bub1 recruits the RZZ complex and BubR1 to the kinetochore, and loss of the BubR1 binding sequence enhances checkpoint activity suggesting both SAC activating and silencing roles.
- Gang Zhang
- , Tiziana Lischetti
- & Jakob Nilsson
-
Article |
 Borealin dimerization mediates optimal CPC checkpoint function by enhancing localization to centromeres and kinetochores
Borealin dimerization mediates optimal CPC checkpoint function by enhancing localization to centromeres and kinetochores
Borealin is a subunit of the chromosomal passenger complex that prevents premature mitosis before spindle assembly is complete. Bekier et al.show that Borealin mediates recruitment of this complex to both kinetochores and centromeres via distinct mechanisms, both of which depend on Borealin dimerization.
- Michael E. Bekier
- , Travis Mazur
- & William R. Taylor
-
Article |
 The internal Cdc20 binding site in BubR1 facilitates both spindle assembly checkpoint signalling and silencing
The internal Cdc20 binding site in BubR1 facilitates both spindle assembly checkpoint signalling and silencing
Kinetochores that fail to form bipolar attachments to the mitotic spindle delay chromosome segregation by BubR1 mediated inhibition of Cdc20. Lischetti et al.show that BubR1 recruits Cdc20 to the kinetochore via a domain that mediates both checkpoint activation and silencing.
- Tiziana Lischetti
- , Gang Zhang
- & Jakob Nilsson
-
Article |
 Kinetochores require oligomerization of Dam1 complex to maintain microtubule attachments against tension and promote biorientation
Kinetochores require oligomerization of Dam1 complex to maintain microtubule attachments against tension and promote biorientation
The oligomeric Dam1 complex mediates microtubule attachment to kinetochores during mitosis; however, the significance of its oligomeric structure remains unclear. Umbreit et al. show that Dam1 oligomerization is required for microtubules to form attachments that are robust against tension.
- Neil T. Umbreit
- , Matthew P. Miller
- & Trisha N. Davis
-
Article |
 The condensin component NCAPG2 regulates microtubule–kinetochore attachment through recruitment of Polo-like kinase 1 to kinetochores
The condensin component NCAPG2 regulates microtubule–kinetochore attachment through recruitment of Polo-like kinase 1 to kinetochores
Polo-like kinase 1 (PLK1) regulates the spindle assembly checkpoint and is recruited to prometaphase kinetochores. Kim et al.show that the condensin subunit NCAPG2 is required for stable interaction of PLK1 with kinetochores and for proper chromosome segregation.
- Jae Hyeong Kim
- , Jaegal Shim
- & Kyung-Tae Kim
-
Article
| Open Access Structural basis for microtubule recognition by the human kinetochore Ska complex
Structural basis for microtubule recognition by the human kinetochore Ska complex
Kinetochores must interact with both polymerizing (straight) and depolymerizing (curved) microtubules to ensure correct mitotic chromosome segregation. Abad et al. reveal how this flexibility is achieved through structural characterization of the interactions between microtubules and the kinetochore protein Ska1.
- Maria Alba Abad
- , Bethan Medina
- & A. Arockia Jeyaprakash
-
Article
| Open Access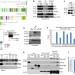 Bod1 regulates protein phosphatase 2A at mitotic kinetochores
Bod1 regulates protein phosphatase 2A at mitotic kinetochores
PP2A-B56 regulates the stability of kinetochore-microtubule attachments by dephosphorylating several kinetochore proteins. Porter et al. identify Bod1 as a specific inhibitor of PP2A-B56 phosphatase activity and show that this activity is required for proper chromosome alignment during mitosis.
- Iain M. Porter
- , Katharina Schleicher
- & Jason R. Swedlow
-
Article
| Open Access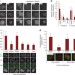 Aurora B potentiates Mps1 activation to ensure rapid checkpoint establishment at the onset of mitosis
Aurora B potentiates Mps1 activation to ensure rapid checkpoint establishment at the onset of mitosis
Mitotic exit is controlled by a cell division checkpoint that prevents premature degradation of cyclin B by the anaphase-promoting complex. Saurinet al. show that Aurora B directly regulates timely establishment of this checkpoint by facilitating activation of Mps1 kinase at unattached kinetochores.
- Adrian T. Saurin
- , Maike S. van der Waal
- & Geert J.P.L. Kops

 Plasmodium ARK2 and EB1 drive unconventional spindle dynamics, during chromosome segregation in sexual transmission stages
Plasmodium ARK2 and EB1 drive unconventional spindle dynamics, during chromosome segregation in sexual transmission stages