Research Highlight |
Featured
-
-
Brief Communication |
 Square condenser apertures for square cameras in low-dose transmission electron microscopy
Square condenser apertures for square cameras in low-dose transmission electron microscopy
Square and rectangular beams made with new condenser apertures enable more efficient image capture in low-dose transmission electron microscopy with no compromise to image quality in single-particle cryoelectron microscopy.
- Hamish G. Brown
- , Dan Smith
- & Eric Hanssen
-
Brief Communication
| Open Access Square beams for optimal tiling in transmission electron microscopy
Square beams for optimal tiling in transmission electron microscopy
A square electron beam improves imaging of large fields of view in transmission electron microscopes by facilitating montage tomography of vitrified specimens with no loss in data quality relative to conventional round beams.
- Eugene Y. D. Chua
- , Lambertus M. Alink
- & Alex de Marco
-
Article |
 DeepMainmast: integrated protocol of protein structure modeling for cryo-EM with deep learning and structure prediction
DeepMainmast: integrated protocol of protein structure modeling for cryo-EM with deep learning and structure prediction
DeepMainmast is a protein structure modeling protocol for cryo-EM that combines the strengths of a deep-learning-based de novo protein main-chain-tracing approach with AlphaFold2-based structure predictions for improved performance.
- Genki Terashi
- , Xiao Wang
- & Daisuke Kihara
-
Brief Communication |
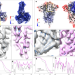 Improving resolution and resolvability of single-particle cryoEM structures using Gaussian mixture models
Improving resolution and resolvability of single-particle cryoEM structures using Gaussian mixture models
This manuscript describes a refinement protocol that extends the e2gmm method to optimize both the orientation and conformation estimation of particles to improve the alignment for flexible domains of proteins.
- Muyuan Chen
- , Michael F. Schmid
- & Wah Chiu
-
Article
| Open Access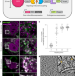 Genetically encoded multimeric tags for subcellular protein localization in cryo-EM
Genetically encoded multimeric tags for subcellular protein localization in cryo-EM
Genetically encoded multimeric particles (GEMs) are 25-nm tags with recognizable structural signatures, which can be used to label specific proteins in mammalian cells to facilitate their subcellular localization in cryo-ET.
- Herman K. H. Fung
- , Yuki Hayashi
- & Julia Mahamid
-
Article
| Open Access OPUS-DSD: deep structural disentanglement for cryo-EM single-particle analysis
OPUS-DSD: deep structural disentanglement for cryo-EM single-particle analysis
OPUS-DSD is a neural network-based algorithm that reconstructs distinct conformations or continuous dynamics of the macromolecular structural landscape, starting from single-particle cryo-EM data.
- Zhenwei Luo
- , Fengyun Ni
- & Jianpeng Ma
-
Article |
 CryoREAD: de novo structure modeling for nucleic acids in cryo-EM maps using deep learning
CryoREAD: de novo structure modeling for nucleic acids in cryo-EM maps using deep learning
Few methods for three-dimensional structure modeling of nucleic acids from cryo-EM data exist. CryoREAD, a fully automated DNA/RNA atomic structure modeling method based on deep learning, was developed to fill this gap.
- Xiao Wang
- , Genki Terashi
- & Daisuke Kihara
-
Research Briefing |
 Capturing detailed cellular landscapes by montage cryo-electron tomography
Capturing detailed cellular landscapes by montage cryo-electron tomography
To capture expansive, seamless fields of view from frozen hydrated specimens by cryo-electron tomography, we developed methods for the collection and processing of montage data. This approach enables rapid acquisition of contiguous regions of specimens using a montaged tilt series collection scheme.
-
Article
| Open Access Correlative montage parallel array cryo-tomography for in situ structural cell biology
Correlative montage parallel array cryo-tomography for in situ structural cell biology
Montage parallel array cryo-tomography adopts principles of montage tomography via regular array beam-image-shift montage acquisition and is robust for imaging large fields of view while retaining high-resolution structural information in cryo-electron tomography.
- Jie E. Yang
- , Matthew R. Larson
- & Elizabeth R. Wright
-
Correspondence |
 scNodes: a correlation and processing toolkit for super-resolution fluorescence and electron microscopy
scNodes: a correlation and processing toolkit for super-resolution fluorescence and electron microscopy
- Mart G. F. Last
- , Lenard M. Voortman
- & Thomas H. Sharp
-
Brief Communication |
 New measures of anisotropy of cryo-EM maps
New measures of anisotropy of cryo-EM maps
This paper proposes two new anisotropy metrics—the Fourier shell occupancy and the Bingham test—that can be used to understand the quality of cryogenic electron microscopy maps.
- Jose-Luis Vilas
- & Hemant D. Tagare
-
Research Briefing |
 Mapping the motion and structure of flexible proteins from cryo-EM data
Mapping the motion and structure of flexible proteins from cryo-EM data
A deep learning algorithm maps out the continuous conformational changes of flexible protein molecules from single-particle cryo-electron microscopy images, allowing the visualization of the conformational landscape of a protein with improved resolution of its moving parts.
-
Article
| Open Access 3DFlex: determining structure and motion of flexible proteins from cryo-EM
3DFlex: determining structure and motion of flexible proteins from cryo-EM
3D Flexible Refinement (3DFlex) is a generative neural network model for continuous molecular heterogeneity for cryo-EM data that can be used to determine the structure and motion of flexible biomolecules. It enables visualization of nonrigid motion and improves 3D structure resolution by aggregating information from particle images spanning the conformational landscape of the target molecule.
- Ali Punjani
- & David J. Fleet
-
Correspondence |
 DAQ-Score Database: assessment of map–model compatibility for protein structure models from cryo-EM maps
DAQ-Score Database: assessment of map–model compatibility for protein structure models from cryo-EM maps
- Tsukasa Nakamura
- , Xiao Wang
- & Daisuke Kihara
-
Technology Feature |
 Tuning in to epigenetic cross-talk
Tuning in to epigenetic cross-talk
Chemical modifications to DNA, histones and RNA make changes happen. Scientists are exploring ways to track these modifications and how they interact.
- Vivien Marx
-
Review Article |
 Cryo-electron tomography on focused ion beam lamellae transforms structural cell biology
Cryo-electron tomography on focused ion beam lamellae transforms structural cell biology
This Review describes advances in cryogenic electron tomography on focused ion beam lamellae, highlighting the key benefits of this technology for in situ structural biology and discussing important future directions.
- Casper Berger
- , Navya Premaraj
- & Peter J. Peters
-
News & Views |
 Seeing the wood for the trees
Seeing the wood for the trees
A deep learning approach called DeepPiCt facilitates segmentation and macromolecular identification in the cellular jungle of electron cryotomography data.
- Olivia E. R. Smith
- & Tanmay A. M. Bharat
-
Article
| Open Access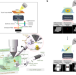 ELI trifocal microscope: a precise system to prepare target cryo-lamellae for in situ cryo-ET study
ELI trifocal microscope: a precise system to prepare target cryo-lamellae for in situ cryo-ET study
The ELI-TriScope advances cryo-CLEM by focusing light, electron and ion beams on cryopreserved samples for markedly improved preparation of cryo-lamellae containing target structures.
- Shuoguo Li
- , Ziyan Wang
- & Fei Sun
-
Article
| Open Access Uniform thin ice on ultraflat graphene for high-resolution cryo-EM
Uniform thin ice on ultraflat graphene for high-resolution cryo-EM
This paper shows that the uniformity of vitreous ice thickness relies on the surface flatness of the supporting film, and presents a method to use ultraflat graphene as the support for cryo-EM specimen preparation.
- Liming Zheng
- , Nan Liu
- & Hailin Peng
-
Research Briefing |
 Fast cryo-electron tomography data acquisition to determine molecular structures in situ
Fast cryo-electron tomography data acquisition to determine molecular structures in situ
To accelerate data acquisition for in situ cryo-electron tomography, we created a method that takes into consideration sample geometry for the robust prediction of sample movement while the microscope stage is tilted. This approach enabled the parallel collection of tens to hundreds of tilt series.
-
Article |
 Parallel cryo electron tomography on in situ lamellae
Parallel cryo electron tomography on in situ lamellae
The parallel cryo electron tomography (PACE-tomo) method increases the throughput on in situ samples by parallelizing acquisition. It maximizes the usable sample area on individual lamellae without compromising data quality.
- Fabian Eisenstein
- , Haruaki Yanagisawa
- & Radostin Danev
-
Research Briefing |
 Obtaining cryo-EM structures by scanning transmission electron microscopy
Obtaining cryo-EM structures by scanning transmission electron microscopy
Scanning transmission electron microscopy (STEM) techniques reveal atomic-resolution details of organic and inorganic materials. The application of STEM to biological vitrified specimens under low-dose cryogenic imaging conditions demonstrates that STEM also achieves near-atomic-resolution 3D structures of biological macromolecules.
-
Article
| Open Access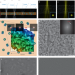 Single-particle cryo-EM structures from iDPC–STEM at near-atomic resolution
Single-particle cryo-EM structures from iDPC–STEM at near-atomic resolution
This paper explores the use of scanning transmission electron microscopy (STEM) to vitrified biological samples for biomolecular structure elucidation. Integrated differential phase contrast (iDPC)–STEM imaging of keyhole limpet hemocyanin and tobacco mosaic virus enabled cryo-EM structure determination at 6.5 and 3.5 Å resolution, respectively.
- Ivan Lazić
- , Maarten Wirix
- & Carsten Sachse
-
Article
| Open Access Ab initio phasing macromolecular structures using electron-counted MicroED data
Ab initio phasing macromolecular structures using electron-counted MicroED data
This article reports sub- and near-atomic structures of triclinic lysozyme and serine protease proteinase K, respectively, providing first demonstrations of ab initio phasing using electron counted MicroED data to solve macromolecular structures.
- Michael W. Martynowycz
- , Max T. B. Clabbers
- & Tamir Gonen
-
Article |
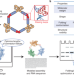 Sub-3-Å cryo-EM structure of RNA enabled by engineered homomeric self-assembly
Sub-3-Å cryo-EM structure of RNA enabled by engineered homomeric self-assembly
ROCK (RNA oligomerization-enabled cryo-EM via installing kissing loops) enables improved single-particle cryo-EM of RNAs. ROCK was used to generate high-quality structures of three diverse RNAs, including the Tetrahymena group I intron.
- Di Liu
- , François A. Thélot
- & Peng Yin
-
Editorial |
 Methods lead the way
Methods lead the way
The COVID-19 pandemic has highlighted the importance of methodological advancements in basic biological research. We believe that method development will continue to propel both fundamental and applied studies on SARS-CoV-2 and other pathogens.
-
Comment |
 Fighting SARS-CoV-2 with structural biology methods
Fighting SARS-CoV-2 with structural biology methods
High-resolution structural information is critical for rapid development of vaccines and therapeutics against emerging human pathogens. Structural biology methods have been at the forefront of research on SARS-CoV-2 since the beginning of the COVID-19 pandemic. These technologies will continue to be powerful tools to fend off future public health threats.
- Jun Zhang
- & Bing Chen
-
Article |
 CR-I-TASSER: assemble protein structures from cryo-EM density maps using deep convolutional neural networks
CR-I-TASSER: assemble protein structures from cryo-EM density maps using deep convolutional neural networks
CR-I-TASSER integrates deep neural-network learning with I-TASSER assembly simulations for automated cryo-EM structure determination.
- Xi Zhang
- , Biao Zhang
- & Yang Zhang
-
Method to Watch |
 A dynamic direction for cryo-EM
A dynamic direction for cryo-EM
Emerging algorithms are extracting information about macromolecular motions from cryo-EM data.
- Allison Doerr
-
Comment |
 A paradigm shift in structural biology
A paradigm shift in structural biology
The release of protein structure predictions from AlphaFold will increase the number of protein structural models by almost three orders of magnitude. Structural biology and bioinformatics will never be the same, and the need for incisive experimental approaches will be greater than ever. Combining these advances in structure prediction with recent advances in cryo-electron microscopy suggests a new paradigm for structural biology.
- Sriram Subramaniam
- & Gerard J. Kleywegt
-
Article |
 Deep learning improves macromolecule identification in 3D cellular cryo-electron tomograms
Deep learning improves macromolecule identification in 3D cellular cryo-electron tomograms
DeepFinder is a deep learning-based tool for identifying macromolecules in cellular cryo-electron tomograms. DeepFinder performs with an accuracy comparable to expert-supervised ground truth annotations on multiple experimental datasets.
- Emmanuel Moebel
- , Antonio Martinez-Sanchez
- & Charles Kervrann
-
News & Views |
 Neural networks learn the motions of molecular machines
Neural networks learn the motions of molecular machines
New computational approaches capture molecular motion from cryo-EM images and provide a more complete understanding of protein dynamics.
- Timothy Grant
-
Article |
 Deep learning-based mixed-dimensional Gaussian mixture model for characterizing variability in cryo-EM
Deep learning-based mixed-dimensional Gaussian mixture model for characterizing variability in cryo-EM
e2gmm uses a deep neural network with a Gaussian representation to resolve the compositional and conformational variability within biomolecules using cryo-EM data.
- Muyuan Chen
- & Steven J. Ludtke
-
Comment |
 Expanding capabilities and infrastructure for cryo-EM
Expanding capabilities and infrastructure for cryo-EM
Many challenges and considerations must be evaluated when expanding and supporting new cryo-electron microscopy facilities.
- Kutti R. Vinothkumar
-
Comment |
 RNA structure: a renaissance begins?
RNA structure: a renaissance begins?
Advances in cryo-EM technology will open a new era of RNA-only 3D structure determination.
- Rhiju Das
-
Review Article |
 Understanding the invisible hands of sample preparation for cryo-EM
Understanding the invisible hands of sample preparation for cryo-EM
The quality of structural data obtained in cryo-EM is affected by multiple factors pertaining to sample preparation. This Review discusses available techniques and current challenges.
- Giulia Weissenberger
- , Rene J. M. Henderikx
- & Peter J. Peters
-
News & Views |
 Improving cryo-EM structure validation
Improving cryo-EM structure validation
A community-wide challenge yields recommendations for improving cryo-EM structure validation.
- Alexis Rohou
-
Article |
 CryoDRGN: reconstruction of heterogeneous cryo-EM structures using neural networks
CryoDRGN: reconstruction of heterogeneous cryo-EM structures using neural networks
CryoDRGN is an unsupervised machine learning algorithm that reconstructs continuous distributions of three-dimensional density maps from heterogeneous single-particle cryo-EM data.
- Ellen D. Zhong
- , Tristan Bepler
- & Joseph H. Davis
-
Analysis
| Open Access Cryo-EM model validation recommendations based on outcomes of the 2019 EMDataResource challenge
Cryo-EM model validation recommendations based on outcomes of the 2019 EMDataResource challenge
A multi-laboratory study in the form of a community challenge assesses the quality of models that can be produced from cryo-EM maps using different software tools, the reproducibility of models generated by different users and the performance of metrics used for model validation.
- Catherine L. Lawson
- , Andriy Kryshtafovych
- & Wah Chiu
-
Article |
 A ‘Build and Retrieve’ methodology to simultaneously solve cryo-EM structures of membrane proteins
A ‘Build and Retrieve’ methodology to simultaneously solve cryo-EM structures of membrane proteins
The iterative Build and Retrieve (BaR) methodology facilitates the solving of cryo-EM structures of multiple membrane (and soluble) proteins simultaneously, including small and low-abundance membrane proteins.
- Chih-Chia Su
- , Meinan Lyu
- & Edward W. Yu
-
Research Highlight |
 Cryo-EM goes atomic
Cryo-EM goes atomic
Technological advances push the limits of single-particle cryo-EM to achieve atomic resolution.
- Rita Strack
-
Article |
 Non-uniform refinement: adaptive regularization improves single-particle cryo-EM reconstruction
Non-uniform refinement: adaptive regularization improves single-particle cryo-EM reconstruction
Membrane proteins exhibit spatial variation in rigidity and disorder, which poses a challenge for traditional cryo-EM reconstruction algorithms. Non-uniform refinement accounts for this spatial variability, yielding improved 3D reconstruction quality even for small membrane proteins.
- Ali Punjani
- , Haowei Zhang
- & David J. Fleet
-
Research Highlight |
 A dual-constriction biological nanopore
A dual-constriction biological nanopore
Addition of a second constriction to CsgG-family nanopores improves the accuracy of homopolymer sequencing.
- Arunima Singh
-
Article |
 Improvement of cryo-EM maps by density modification
Improvement of cryo-EM maps by density modification
A method based on maximum likelihood density modification, adapted from X-ray crystallography, improves cryo-EM maps.
- Thomas C. Terwilliger
- , Steven J. Ludtke
- & Pavel V. Afonine
-
Brief Communication |
 Time-resolved cryo-EM using Spotiton
Time-resolved cryo-EM using Spotiton
A spraying-mixing approach for preparing cryo-EM grids using the Spotiton robot allows time-resolved observations of short-lived biomolecular states.
- Venkata P. Dandey
- , William C. Budell
- & Bridget Carragher
-
-
Article |
 Accelerated cryo-EM-guided determination of three-dimensional RNA-only structures
Accelerated cryo-EM-guided determination of three-dimensional RNA-only structures
The Ribosolve pipeline combines single-particle cryo-EM, M2-seq biochemical analysis and Rosetta auto-DRRAFTER modeling to guide three-dimensional RNA structure determination.
- Kalli Kappel
- , Kaiming Zhang
- & Rhiju Das
-
Article |
 Measurement of atom resolvability in cryo-EM maps with Q-scores
Measurement of atom resolvability in cryo-EM maps with Q-scores
Q-scores provide a quantitative metric for resolvability in cryo-EM maps, and can be used at the atom, residue or macromolecule scale.
- Grigore Pintilie
- , Kaiming Zhang
- & Wah Chiu
-
Article |
 In situ structure determination at nanometer resolution using TYGRESS
In situ structure determination at nanometer resolution using TYGRESS
The unique advantages of single-particle cryo-electron microscopy and cryo-electron tomography are combined in a method called TYGRESS, here applied to determine the structure of the intact ciliary axoneme at a resolution of 12 Å.
- Kangkang Song
- , Zhiguo Shang
- & Daniela Nicastro

 Graphene sandwich for cryo-EM
Graphene sandwich for cryo-EM