Featured
-
-
Article |
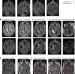 Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results
Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results
In an interim analysis of an ongoing phase 1 trial of CAR T cells targeting EGFR and IL13Ra2 in patients with multifocal, recurrent glioblastoma, intrathecal delivery is feasible and well tolerated, with some reductions seen in tumor size.
- Stephen J. Bagley
- , Meghan Logun
- & Donald M. O’Rourke
-
Research Highlight |
 MEGA CRISPR rejuvenates exhausted CAR T cells
MEGA CRISPR rejuvenates exhausted CAR T cells
MEGA is a new CRISPR-based RNA-editing platform with the ability to enhance the fitness of CAR T cells; it may also overcome certain limitations of conventional DNA-targeting CRISPR–Cas9 systems.
- Karen O’Leary
-
Article
| Open Access Locoregional delivery of IL-13Rα2-targeting CAR-T cells in recurrent high-grade glioma: a phase 1 trial
Locoregional delivery of IL-13Rα2-targeting CAR-T cells in recurrent high-grade glioma: a phase 1 trial
In a large trial of patients with recurrent high-grade gliomas, IL-13Rα2-targeting CAR-T cells were feasible to manufacture and well tolerated when delivered via intratumoral and/or intraventricular routes.
- Christine E. Brown
- , Jonathan C. Hibbard
- & Behnam Badie
-
Article |
 Combined anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal cancer: a randomized phase 2 trial
Combined anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal cancer: a randomized phase 2 trial
In a randomized phase 2 trial comparing anti-PD-1 plus anti-VEGF plus an HDAC inhibitor versus anti-PD-1 plus an HDAC inhibitor in patients with MSS/pMMR colorectal cancer, the triplet combination led to significantly better 18-week progression-free survival.
- Feng Wang
- , Ying Jin
- & Rui-Hua Xu
-
Article
| Open Access A gut microbial signature for combination immune checkpoint blockade across cancer types
A gut microbial signature for combination immune checkpoint blockade across cancer types
Efforts to identify microbial signatures of response to immune checkpoint blockade suggest that strain-level associations may be cancer type agnostic but cancer therapy specific.
- Ashray Gunjur
- , Yan Shao
- & Trevor D. Lawley
-
News & Views |
 Immunotherapy at all stages of hepatocellular carcinoma
Immunotherapy at all stages of hepatocellular carcinoma
A flurry of emerging treatment options is transforming the therapeutic landscape of hepatocellular carcinoma; two new studies highlight the complexities and gaps in knowledge, but also hint at a greater understanding of how to apply immunotherapy in early and advanced disease.
- David J. Pinato
- , Claudia A. M. Fulgenzi
- & Antonio D’Alessio
-
Research Briefing |
 Longitudinal analysis shows how microbiome changes might influence responses to immune checkpoint blockade
Longitudinal analysis shows how microbiome changes might influence responses to immune checkpoint blockade
Longitudinal analysis of stool samples reveals new insights into how the gut microbiome changes during immunotherapy. These findings could inform the application of microbiome-targeted interventions to maximize the outcomes of immune checkpoint blockade.
-
Brief Communication |
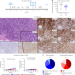 T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy
T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy
Profiling of a case of secondary T cell lymphoma following anti-CD19 CAR T cell therapy suggests that it was not caused by CAR insertional mutagenesis, with single-center analysis indicating that secondary T cell lymphoma risk after commercial CAR T cell treatment is low.
- Guido Ghilardi
- , Joseph A. Fraietta
- & Marco Ruella
-
Article
| Open Access Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial
Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial
In the final report of a phase 1/2 trial evaluating allogeneic CD19-specific CAR-NK cells armored with IL-15 in patients with CD19+ hematologic malignancies, the therapy was shown to be safe and efficacious with distinct cord blood features associated with response.
- David Marin
- , Ye Li
- & Katayoun Rezvani
-
Article
| Open Access Impact of tumor microenvironment on efficacy of anti-CD19 CAR T cell therapy or chemotherapy and transplant in large B cell lymphoma
Impact of tumor microenvironment on efficacy of anti-CD19 CAR T cell therapy or chemotherapy and transplant in large B cell lymphoma
Analysis of the pivotal phase 3 ZUMA-7 trial identifies tumor gene expression signatures that are uniquely predictive of anti-CD19 CAR T cell response and event-free survival in second-line treatment for patients with relapsed or refractory large B cell lymphoma.
- Frederick L. Locke
- , Simone Filosto
- & Jérôme Galon
-
Comment |
Unanswered questions following reports of secondary malignancies after CAR-T cell therapy
Reports of T cell malignancies after CAR-T cell therapy should be investigated, but existing data from follow-up studies suggest a low risk compared with other cancer treatments.
- Bruce L. Levine
- , Marcelo C. Pasquini
- & Miguel-Angel Perales
-
Article
| Open Access Mosunetuzumab with polatuzumab vedotin in relapsed or refractory aggressive large B cell lymphoma: a phase 1b/2 trial
Mosunetuzumab with polatuzumab vedotin in relapsed or refractory aggressive large B cell lymphoma: a phase 1b/2 trial
In an ongoing phase 1/2 trial, mosunetuzumab, a CD20xCD3 bispecific antibody, plus polatuzumab vedotin, an anti-CD79B antibody–drug conjugate, had an overall response rate of 59.2%, supporting the potential of this combination for second-line treatment of patients with transplant-ineligible or CAR-T-cell-ineligible relapsed or refractory large B cell lymphoma.
- Lihua E. Budde
- , Adam J. Olszewski
- & Julio C. Chavez
-
Article
| Open Access Tislelizumab plus zanubrutinib for Richter transformation: the phase 2 RT1 trial
Tislelizumab plus zanubrutinib for Richter transformation: the phase 2 RT1 trial
In a large single-arm phase 2 trial, the anti-PD-1 inhibitor tislelizumab combined with the next-generation BTK inhibitor zanubrutinib had an overall response rate of 58.3% and was well tolerated in patients with Richter’s transformation.
- Othman Al-Sawaf
- , Rudy Ligtvoet
- & Barbara Eichhorst
-
Article |
 Association between pretreatment emotional distress and neoadjuvant immune checkpoint blockade response in melanoma
Association between pretreatment emotional distress and neoadjuvant immune checkpoint blockade response in melanoma
In a post hoc analysis of the phase 2 PRADO trial, baseline emotional distress was associated with reduced clinical responses in patients with stage III melanoma treated with neoadjuvant ipilimumab and nivolumab.
- Itske Fraterman
- , Irene L. M. Reijers
- & Lonneke V. van de Poll-Franse
-
Article
| Open Access Association between pathologic response and survival after neoadjuvant therapy in lung cancer
Association between pathologic response and survival after neoadjuvant therapy in lung cancer
Analysis of the phase 3 CheckMate 816 trial shows that the depth of pathologic response as assessed by percent residual viable tumor is correlated with event-free survival following neoadjuvant immunotherapy plus chemotherapy, supporting pathologic response as a biomarker of survival.
- Julie Stein Deutsch
- , Ashley Cimino-Mathews
- & Janis M. Taube
-
Article
| Open Access CLDN6-specific CAR-T cells plus amplifying RNA vaccine in relapsed or refractory solid tumors: the phase 1 BNT211-01 trial
CLDN6-specific CAR-T cells plus amplifying RNA vaccine in relapsed or refractory solid tumors: the phase 1 BNT211-01 trial
In the ongoing phase 1/2 BNT211-01 trial, CLDN6-specific chimeric antigen receptor (CAR)-T cells given with or without CARVac, a CAR-T cell-amplifying RNA vaccine, were well-tolerated and exhibited encouraging clinical activity in patients with relapsed or refractory CLDN6-positive solid tumors, with the highest response rate in patients with germ cell tumors.
- Andreas Mackensen
- , John B.A.G. Haanen
- & Uğur Şahin
-
Article
| Open Access The PD-1- and LAG-3-targeting bispecific molecule tebotelimab in solid tumors and hematologic cancers: a phase 1 trial
The PD-1- and LAG-3-targeting bispecific molecule tebotelimab in solid tumors and hematologic cancers: a phase 1 trial
The bispecific molecule tebotelimab, which blocks both PD-1 and LAG-3, is well tolerated as a monotherapy and in combination with the anti-HER-2 antibody margetuximab and elicits encouraging clinical activity in solid tumors with high LAG-3 levels and/or expression of IFN-γ-regulated genes.
- Jason J. Luke
- , Manish R. Patel
- & Paul A. Moore
-
Brief Communication |
 Neoplasia risk in patients with Lynch syndrome treated with immune checkpoint blockade
Neoplasia risk in patients with Lynch syndrome treated with immune checkpoint blockade
In a retrospective analysis of patients with Lynch syndrome and primary cancers treated with immune checkpoint inhibitors, 12% developed subsequent malignancies, suggesting that this treatment may not eliminate risk in individuals predisposed to mismatch repair-deficient cancers, and ongoing surveillance is warranted.
- Emily C. Harrold
- , Michael B. Foote
- & Zsofia K. Stadler
-
Article
| Open Access Adjuvant nivolumab in resected stage IIB/C melanoma: primary results from the randomized, phase 3 CheckMate 76K trial
Adjuvant nivolumab in resected stage IIB/C melanoma: primary results from the randomized, phase 3 CheckMate 76K trial
In this pre-specified interim analysis, patients with resected stage IIB/C melanoma who received adjuvant nivolumab had significantly prolonged recurrence-free survival compared to placebo-treated patients, providing another treatment option for this population.
- John M. Kirkwood
- , Michele Del Vecchio
- & Georgina V. Long
-
Article
| Open Access Gemcitabine and cisplatin plus nivolumab as organ-sparing treatment for muscle-invasive bladder cancer: a phase 2 trial
Gemcitabine and cisplatin plus nivolumab as organ-sparing treatment for muscle-invasive bladder cancer: a phase 2 trial
In a phase 2 trial, the combination of gemcitabine, cisplatin and anti-PD-1 led to a clinical complete response in 43% of patients with muscle-invasive bladder cancer, which facilitated bladder sparing and was associated with long-term bladder-intact metastasis-free survival.
- Matthew D. Galsky
- , Siamak Daneshmand
- & Sumanta K. Pal
-
Article
| Open Access Axicabtagene ciloleucel as second-line therapy in large B cell lymphoma ineligible for autologous stem cell transplantation: a phase 2 trial
Axicabtagene ciloleucel as second-line therapy in large B cell lymphoma ineligible for autologous stem cell transplantation: a phase 2 trial
In the ALYCANTE trial, second-line treatment with axicabtagene ciloleucel, a CD19-targeting CAR T-cell therapy, resulted in high response rates and durable remissions in patients with high-risk large B-cell lymphoma deemed ineligible for autologous stem cell transplantation.
- Roch Houot
- , Emmanuel Bachy
- & François Lemonnier
-
Article |
 Nivolumab plus ipilimumab in advanced salivary gland cancer: a phase 2 trial
Nivolumab plus ipilimumab in advanced salivary gland cancer: a phase 2 trial
Treatment of patients with metastatic salivary gland cancer with anti-PD-1 and anti-CTLA-4 led to encouraging clinical benefit in certain histologic subtypes, with translational analyses showing pre-existing T cell clonal expansion in responding tumors.
- Joris L. Vos
- , Bharat Burman
- & Luc G. T. Morris
-
Article
| Open Access Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results
Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results
In a pivotal phase 2 trial, elranatamab, a bispecific antibody targeting B-cell maturation antigen (BCMA) and CD3, had a confirmed objective response rate of 61% in patients with relapsed or refractory multiple myeloma who had not previously received BCMA-directed therapy.
- Alexander M. Lesokhin
- , Michael H. Tomasson
- & Mohamad Mohty
-
Article
| Open Access First-line durvalumab and tremelimumab with chemotherapy in RAS-mutated metastatic colorectal cancer: a phase 1b/2 trial
First-line durvalumab and tremelimumab with chemotherapy in RAS-mutated metastatic colorectal cancer: a phase 1b/2 trial
Treatment with anti-PDL1 and anti-CTLA4 in combination with mFOLFOX6 chemotherapy for patients with treatment-naive microsatellite stable metastatic colorectal cancer is safe, shows encouraging progression-free survival and induces a tumor-specific immune response.
- Marion Thibaudin
- , Jean-David Fumet
- & François Ghiringhelli
-
Article
| Open Access Mesothelin-targeting T cell receptor fusion construct cell therapy in refractory solid tumors: phase 1/2 trial interim results
Mesothelin-targeting T cell receptor fusion construct cell therapy in refractory solid tumors: phase 1/2 trial interim results
T cells containing an anti-mesothelin single-domain antibody fused to a component of the endogenous T cell receptor signaling complex exhibit notable toxicities but encouraging clinical responses in patients with treatment-refractory mesothelioma and ovarian cancer.
- Raffit Hassan
- , Marcus Butler
- & David Hong
-
Article
| Open Access Neoadjuvant adebrelimab in locally advanced resectable esophageal squamous cell carcinoma: a phase 1b trial
Neoadjuvant adebrelimab in locally advanced resectable esophageal squamous cell carcinoma: a phase 1b trial
In the NATION-1907 trial, treatment of patients with resectable esophageal squamous cell carcinoma with the anti-PD-L1 agent adebrelimab was safe and showed preliminary overall survival efficacy, with responders exhibiting an immune-enriched tumor microenvironment phenotype at baseline.
- Jun Yin
- , Jingnan Yuan
- & Qing Zhou
-
Article |
 CD19 CAR T-cell therapy and prophylactic anakinra in relapsed or refractory lymphoma: phase 2 trial interim results
CD19 CAR T-cell therapy and prophylactic anakinra in relapsed or refractory lymphoma: phase 2 trial interim results
Prophylactic subcutaneous administration of anakinra, an IL-1 receptor inhibitor, reduced severe immune effector cell-associated neurotoxicity syndrome incidence in patients with relapsed or refractory large B-cell and mantle cell lymphoma treated with anti-CD19 chimeric antigen receptor (CAR) T cells.
- Jae H. Park
- , Karthik Nath
- & Michel Sadelain
-
Research Highlight |
 Base-edited CAR T cells for pediatric leukemia
Base-edited CAR T cells for pediatric leukemia
Researchers used base editing — a type of CRISPR technology — to create a cell-therapy product with potent activity in children with relapsed T-ALL.
- Karen O’Leary
-
Comment |
 Neoadjuvant immunotherapy for melanoma is now ready for clinical practice
Neoadjuvant immunotherapy for melanoma is now ready for clinical practice
Individuals with cutaneous melanoma who are treated with neoadjuvant immunotherapy show a substantial improvement; this should be incorporated into standard care.
- Claus Garbe
- , Reinhard Dummer
- & Georgina V. Long
-
Research Highlight |
 CAR T cells show promise in neuroblastoma
CAR T cells show promise in neuroblastoma
Using CAR T cells to treat solid tumors is notoriously challenging, but a new study shows impressive responses rates in children with high-risk neuroblastoma.
- Karen O’Leary
-
Perspective |
 Tumor inflammation-associated neurotoxicity
Tumor inflammation-associated neurotoxicity
The authors describe a localized toxicity syndrome that is associated with immunotherapy treatment for CNS tumors and propose a new grading scale—with the goal of promoting research and standardizing both reporting and management.
- Jasia Mahdi
- , Jorg Dietrich
- & Michelle Monje
-
Article
| Open Access Concurrent intrathecal and intravenous nivolumab in leptomeningeal disease: phase 1 trial interim results
Concurrent intrathecal and intravenous nivolumab in leptomeningeal disease: phase 1 trial interim results
In an interim analysis of a phase 1 trial of concurrent intrathecal and intravenous anti-PD1 delivery in patients with leptomeningeal disease and melanoma, treatment was feasible and well-tolerated with no dose-limiting toxicities.
- Isabella C. Glitza Oliva
- , Sherise D. Ferguson
- & Michael A. Davies
-
Research Highlight |
 Earlier appears better for immunotherapy in melanoma
Earlier appears better for immunotherapy in melanoma
For patients with advanced, resectable melanoma, treatment with pembrolizumab before as well as after surgery led to improved outcomes — without additional toxicity.
- Karen O’Leary
-
Research Highlight |
 Mediterranean diet linked to immunotherapy response
Mediterranean diet linked to immunotherapy response
In patients with advanced melanoma, adherence to a habitual Mediterranean diet was associated with a higher probability of responding to immunotherapy.
- Karen O’Leary
-
Article
| Open Access First-line serplulimab or placebo plus chemotherapy in PD-L1-positive esophageal squamous cell carcinoma: a randomized, double-blind phase 3 trial
First-line serplulimab or placebo plus chemotherapy in PD-L1-positive esophageal squamous cell carcinoma: a randomized, double-blind phase 3 trial
The combination of anti-PD-1 treatment with cisplatin and 5-fluorouracil in patients with previously untreated, PD-L1-positive esophageal squamous cell carcinoma improved progression-free survival and overall survival.
- Yan Song
- , Bo Zhang
- & Jing Huang
-
Article
| Open Access Combined PD-1, BRAF and MEK inhibition in BRAFV600E colorectal cancer: a phase 2 trial
Combined PD-1, BRAF and MEK inhibition in BRAFV600E colorectal cancer: a phase 2 trial
Patients with BRAFV600E-mutated colorectal cancer have encouraging overall response rates to inhibition of PD-1, BRAF and MEK, with translational analyses suggesting that induction of tumor-intrinsic programs and immune programs contributes to improved outcomes via MAPK inhibition.
- Jun Tian
- , Jonathan H. Chen
- & Ryan B. Corcoran
-
Article |
 Allogeneic BCMA-targeting CAR T cells in relapsed/refractory multiple myeloma: phase 1 UNIVERSAL trial interim results
Allogeneic BCMA-targeting CAR T cells in relapsed/refractory multiple myeloma: phase 1 UNIVERSAL trial interim results
In an interim phase 1 trial analysis, escalating doses of off-the-shelf allogeneic anti-BCMA CAR T cells, in combination with an anti-CD52 antibody-containing lymphodepletion regimen, were feasible to administer and exhibited an encouraging safety profile in patients with relapsed or refractory multiple myeloma
- Sham Mailankody
- , Jeffrey V. Matous
- & Shaji K. Kumar
-
Article
| Open Access Autologous T cell therapy for MAGE-A4+ solid cancers in HLA-A*02+ patients: a phase 1 trial
Autologous T cell therapy for MAGE-A4+ solid cancers in HLA-A*02+ patients: a phase 1 trial
In a phase 1 dose-escalation trial in patients with nine different types of solid tumors, MAGE-A4-specific T cells had an acceptable safety profile and exhibited an encouraging overall response rate in patients with synovial sarcoma.
- David S. Hong
- , Brian A. Van Tine
- & Marcus O. Butler
-
Article |
 Germline variants associated with toxicity to immune checkpoint blockade
Germline variants associated with toxicity to immune checkpoint blockade
A genome-wide association study in large cohorts of patients with different types of cancer treated with immune checkpoint inhibitors identifies genetic variants associated with immune-related adverse events.
- Stefan Groha
- , Sarah Abou Alaiwi
- & Alexander Gusev
-
Article
| Open Access Atezolizumab plus anthracycline-based chemotherapy in metastatic triple-negative breast cancer: the randomized, double-blind phase 2b ALICE trial
Atezolizumab plus anthracycline-based chemotherapy in metastatic triple-negative breast cancer: the randomized, double-blind phase 2b ALICE trial
In patients with metastatic triple-negative breast cancer, atezolizumab combined with immunogenic, anthracycline-based metronomic chemotherapy improves progression-free survival, including in patients with PD-L1negative disease.
- Andreas Hagen Røssevold
- , Nikolai Kragøe Andresen
- & Jon Amund Kyte
-
Article
| Open Access Whole-body CD8+ T cell visualization before and during cancer immunotherapy: a phase 1/2 trial
Whole-body CD8+ T cell visualization before and during cancer immunotherapy: a phase 1/2 trial
A CD8-specific, one-armed antibody positron emission tomography tracer enables the visualization of the immune response in patients with solid tumors before and after starting immunotherapy.
- Laura Kist de Ruijter
- , Pim P. van de Donk
- & Elisabeth G. E. de Vries
-
Research Highlight |
 Cancer immunotherapy gets personal
Cancer immunotherapy gets personal
In a first in-human study, researchers used an array of technologies to generate highly personalized cell therapy products for patients with solid tumors.
- Karen O’Leary
-
Correspondence |
Unresolved questions in the second-line use of CAR-T cells for diffuse large B cell lymphoma
- Edward R. Scheffer Cliff
- , Reid W. Merryman
- & Caron A. Jacobson
-
Correspondence |
Optimizing the dose and schedule of immune checkpoint inhibitors in cancer to allow global access
- Ian Hirsch
- , Daniel A. Goldstein
- & Duncan C. Gilbert
-
Research Briefing |
 Trogocytosis-mediated tumor relapse after CAR-NK cell therapy prevented by inhibitory CARs
Trogocytosis-mediated tumor relapse after CAR-NK cell therapy prevented by inhibitory CARs
CAR-mediated trogocytosis contributes to dysfunction of natural killer (NK) cells by promoting fratricide and is an important mechanism of relapse after CAR-NK cell therapy. This problem can be overcome using a dual-CAR strategy that incorporates a dominant-negative signal to prevent NK cell fratricide and exhaustion, while sparing the CAR-activating signal against tumor targets.
-
Article |
 KIR-based inhibitory CARs overcome CAR-NK cell trogocytosis-mediated fratricide and tumor escape
KIR-based inhibitory CARs overcome CAR-NK cell trogocytosis-mediated fratricide and tumor escape
A new dual-chimeric antigen receptor (CAR) system enhances the antitumor activity of CAR natural killer cells and makes them less susceptible to therapeutic resistance in preclinical models.
- Ye Li
- , Rafet Basar
- & Katayoun Rezvani
-
Article
| Open Access A real-world comparison of tisagenlecleucel and axicabtagene ciloleucel CAR T cells in relapsed or refractory diffuse large B cell lymphoma
A real-world comparison of tisagenlecleucel and axicabtagene ciloleucel CAR T cells in relapsed or refractory diffuse large B cell lymphoma
Analysis of outcomes of over 800 patients with relapsed/refractory diffuse large B cell lymphoma, treated with commercially available CAR T cell therapy, supports higher efficacy and also a higher toxicity of axicabtagene ciloleucel compared to tisagenlecleucel as the third or more treatment line for this type of tumor.
- Emmanuel Bachy
- , Steven Le Gouill
- & Franck Morschhauser
-
Research Highlight |
 Impact of cancer immunotherapy on female fertility
Impact of cancer immunotherapy on female fertility
Mouse models reveal diminished ovarian function and fertility following checkpoint inhibitor therapy, highlighting a major clinical research gap in the cancer immunotherapy field.
- Karen O’Leary
-
Article
| Open Access Cemiplimab plus chemotherapy versus chemotherapy alone in non-small cell lung cancer: a randomized, controlled, double-blind phase 3 trial
Cemiplimab plus chemotherapy versus chemotherapy alone in non-small cell lung cancer: a randomized, controlled, double-blind phase 3 trial
Results from EMPOWER-Lung 3 demonstrate increased overall survival with cemiplimab plus platinum-doublet chemotherapy compared to cemiplimab as first-line treatment in patients with advanced non-small cell lung cancer, irrespective of PD-L1 expression.
- Miranda Gogishvili
- , Tamar Melkadze
- & Petra Rietschel

 A shared neoantigen vaccine combined with immune checkpoint blockade for advanced metastatic solid tumors: phase 1 trial interim results
A shared neoantigen vaccine combined with immune checkpoint blockade for advanced metastatic solid tumors: phase 1 trial interim results