Featured
-
-
Article
| Open Access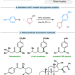 Ni-catalyzed enantioconvergent deoxygenative reductive cross-coupling of unactivated alkyl alcohols and aryl bromides
Ni-catalyzed enantioconvergent deoxygenative reductive cross-coupling of unactivated alkyl alcohols and aryl bromides
The direct reductive coupling of alkyl alcohol and aryl halide enables efficient access to valuable compounds, but the asymmetric pattern remains unknown. Here the authors describe the enantioconvergent deoxygenative reductive cross-coupling of unactivated alkyl alcohol and aryl bromide in the presence of an NHC activating agent.
- Li-Li Zhang
- , Yu-Zhong Gao
- & Ze-Peng Yang
-
Article
| Open Access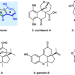 Enantioselective total synthesis of (‒)-lucidumone enabled by tandem prins cyclization/cycloetherification sequence
Enantioselective total synthesis of (‒)-lucidumone enabled by tandem prins cyclization/cycloetherification sequence
The Ganoderma meroterpenoids are a growing class of natural products with a wide range of biological activities. Here, the authors report the enantioselective total synthesis of the Ganoderma meroterpenoid (‒)-lucidumone, featuring a copper-catalyzed enantioselective silicon-tethered intramolecular Diels-Alder cycloaddition to assemble the bicyclo[2.2.2]octane framework and a domino deprotection/Prins reaction/cycloetherification/oxidation sequence to generate concurrenly the tetrahydrofuran and the fused indanone skeleton.
- Xian-Zhang Liao
- , Ran Wang
- & Guang Li
-
Article
| Open Access Asymmetric synthesis of P-stereogenic phosphindane oxides via kinetic resolution and their biological activity
Asymmetric synthesis of P-stereogenic phosphindane oxides via kinetic resolution and their biological activity
P-stereogenic heterocycles are privileged chiral ligands and bioactive compounds, but the catalytic asymmetric synthesis of P-stereogenic phosphindane derivatives is challenging. Here, the authors report a catalytic kinetic resolution of phosphindole oxides via rhodium-catalyzed diastereo- and enantioselective conjugate addition to access enantiopure Pstereogenic phosphindane and phosphindole derivatives.
- Long Yin
- , Jiajia Li
- & Dong Guo
-
Article
| Open Access Carbene organic catalytic planar enantioselective macrolactonization
Carbene organic catalytic planar enantioselective macrolactonization
Macrolactones exhibit distinct conformational and configurational properties and are widely found in natural products but catalysts to govern both macrolactone formation and stereochemical control remain largely unexplored. Here, the authors disclose an N-heterocyclic carbene (NHC)-catalyzed enantioselective synthesis of chiral macrolactones varying in ring size from sixteen to twenty members that feature distinct configurationally stable planar stereogenicity.
- Xiaokang Lv
- , Fen Su
- & Yonggui Robin Chi
-
Article
| Open Access Asymmetric formal sp2-hydrocarbonations of dienes and alkynes via palladium hydride catalysis
Asymmetric formal sp2-hydrocarbonations of dienes and alkynes via palladium hydride catalysis
Sp2 carbon nucleophiles are generally not considered for transition metal-catalyzed asymmetric hydrofunctionalizations of unsaturated bonds due to challenges in cleaving corresponding inert sp2 C-H bonds. Here, the authors report a protocol to achieve asymmetric formal sp2 hydrocarbonations, including hydroalkenylation, hydroallenylation and hydroketenimination of both 1,3-dienes and alkynes via hydroalkylation and Wittig reaction cascade
- Ming-Qiao Tang
- , Zi-Jiang Yang
- & Zhi-Tao He
-
Article
| Open Access Stereospecific synthesis of silicon-stereogenic optically active silylboranes and general synthesis of chiral silyl Anions
Stereospecific synthesis of silicon-stereogenic optically active silylboranes and general synthesis of chiral silyl Anions
Silicon-stereogenic optically active silylboranes could potentially allow the formation of chiral silyl nucleophiles as well as the synthesis of various chiral silicon compounds but the synthesis of such silicon-stereogenic silylboranes remains underdeveloped. Here, the authors report the synthesis of silicon-stereogenic optically active silylboranes via a stereospecific Si–H borylation of chiral hydrosilanes in high yield enantiospecificity
- Xihong Wang
- , Chi Feng
- & Hajime Ito
-
Article
| Open Access Enantioconvergent construction of stereogenic silicon via Lewis base-catalyzed dynamic kinetic silyletherification of racemic chlorosilanes
Enantioconvergent construction of stereogenic silicon via Lewis base-catalyzed dynamic kinetic silyletherification of racemic chlorosilanes
Organosilanes bearing an enantioenriched silicon center are important in many fields of chemistry. Here, the authors report a dynamic kinetic silyletherification racemic chlorosilanes with (S)- lactates, leading to asymmetric synthesis of silylethers.
- Tianbao Hu
- , Chen Zhao
- & Zhenlei Song
-
Article
| Open Access Enantioselectivity of discretized helical supramolecule consisting of achiral cobalt phthalocyanines via chiral-induced spin selectivity effect
Enantioselectivity of discretized helical supramolecule consisting of achiral cobalt phthalocyanines via chiral-induced spin selectivity effect
Experimental realizations of absolute enantioselection, without chiral catalysis or chiral ingredients, has been challenging. Here, the authors obtain enantioselectivity in mesoscale helical supramolecules consisting only of achiral molecules by exploiting chiral-induced spin selectivity (CISS) effect.
- Hiroki Aizawa
- , Takuro Sato
- & Hiroshi M. Yamamoto
-
Article
| Open Access Catalytic asymmetric oxa-Diels–Alder reaction of acroleins with simple alkenes
Catalytic asymmetric oxa-Diels–Alder reaction of acroleins with simple alkenes
The asymmetric inverse-electron-demand oxa-Diels–Alder reaction (IODA) between activated α,β-unsaturated carbonyl compounds with highly polarized alkenes has been reported before but acroleins or simple alkenes were so-far rarely employed as substrates. Here, the authors study the catalytic asymmetric intermolecular IODA reaction between acroleins and simple alkenes.
- Lei Zeng
- , Shihan Liu
- & Lizhu Gao
-
Article
| Open Access Copper-catalyzed asymmetric C(sp2)–H arylation for the synthesis of P- and axially chiral phosphorus compounds
Copper-catalyzed asymmetric C(sp2)–H arylation for the synthesis of P- and axially chiral phosphorus compounds
Although transition metal-catalyzed C–H bond functionalization is a widely used method in organic synthesis, many methods rely on metals of low abundance. Here, the authors report a copper-catalyzed, asymmetric C–H arylation using diaryliodonium salts.
- Shao-Bai Yan
- , Rui Wang
- & Wei-Liang Duan
-
Article
| Open Access Copper(I)-catalyzed asymmetric alkylation of α-imino-esters
Copper(I)-catalyzed asymmetric alkylation of α-imino-esters
Asymmetric alkylation of enolates is an important method for the formation of α-stereogenic carbonyls but generally requires the introduction and then removal of chiral auxiliaries. Here, the authors report a copper-catalyzed asymmetric alkylation of α-amino esters.
- Zong-Ci Liu
- , Zi-Qing Wang
- & Liang Yin
-
Article
| Open Access An α-chloroaldehyde-based formal synthesis of eribulin
An α-chloroaldehyde-based formal synthesis of eribulin
Eribulin is an anticancer drug and the most structurally complex non-peptidic drug made by chemical synthesis. Here, the authors report a streamlined synthesis of eribulin that creates new opportunities for drug discovery and development.
- Anissa Kaghad
- , Dimitrios Panagopoulos
- & Robert Britton
-
Article
| Open Access Achiral organoiodine-functionalized helical polyisocyanides for multiple asymmetric dearomative oxidations
Achiral organoiodine-functionalized helical polyisocyanides for multiple asymmetric dearomative oxidations
Immobilizing organocatalysts onto helical polymers can facilitate catalyst recycling and boost enantioselectivity. Here, the authors prepare achiral organoiodine-functionalized single left- and right-handed helical polyisocyanides from the same monomers and demonstrate their use in three asymmetric oxidations.
- Zong-Quan Wu
- , Xue Song
- & Na Liu
-
Article
| Open Access Electricity-driven asymmetric bromocyclization enabled by chiral phosphate anion phase-transfer catalysis
Electricity-driven asymmetric bromocyclization enabled by chiral phosphate anion phase-transfer catalysis
Asymmetric electrochemical synthesis via ion-pairing catalysis is challenging due to interference of the electrolyte with the ion-pairing catalyst. Here, the authors describe a biphasic system to enable the asymmetric ion-pairing catalysis under electrochemical conditions.
- Xuefeng Tan
- , Qingli Wang
- & Jianwei Sun
-
Article
| Open Access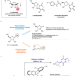 Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Nucleosides and their analogs are pharmacologically important molecules. Here, the authors report an additive-controlled stereodivergent iodocyclization method for the selective synthesis of α- or β-nucleosides.
- Qi Wang
- , Jiayi Mu
- & Fener Chen
-
Article
| Open Access An enantioselective four-component reaction via assembling two reaction intermediates
An enantioselective four-component reaction via assembling two reaction intermediates
The interception of one chemical intermediate with another intermediate is very challenging, as intermediates may be incompatible or too reactive, but could result in a streamlined method of organic synthesis. Here, the authors demonstrate enantioselective four-component reactions via the coupling of two intermediates.
- Sifan Yu
- , Wenju Chang
- & Wenhao Hu
-
Article
| Open Access Enantioselective synthesis of N-alkylindoles enabled by nickel-catalyzed C-C coupling
Enantioselective synthesis of N-alkylindoles enabled by nickel-catalyzed C-C coupling
Enantioenriched N-alkylindole compounds, in which nitrogen is bound to a stereogenic sp3 carbon, are target molecules in the fields of biological, medicinal, and organic chemistry. Here the authors show a nickel-catalyzed C-C coupling protocol to synthesize these molecules from N-indolylsubstituted alkenes.
- Lun Li
- , Jiangtao Ren
- & Deyun Qian
-
Article
| Open Access Stereoretentive cross-coupling of chiral amino acid chlorides and hydrocarbons through mechanistically controlled Ni/Ir photoredox catalysis
Stereoretentive cross-coupling of chiral amino acid chlorides and hydrocarbons through mechanistically controlled Ni/Ir photoredox catalysis
Chiral α-amino ketones are privileged motifs in bioorganic and medicinal chemistry. Here, the authors develop an efficient method to synthesize these structures via stereoretentive direct cross-coupling of amino acid chlorides with simple aliphatic substrates.
- Geun Seok Lee
- , Beomsoon Park
- & Soon Hyeok Hong
-
Article
| Open Access Collective total synthesis of C4-oxygenated securinine-type alkaloids via stereocontrolled diversifications on the piperidine core
Collective total synthesis of C4-oxygenated securinine-type alkaloids via stereocontrolled diversifications on the piperidine core
A general synthetic solution toward high-oxidation state securinega alkaloids could be useful for systematic biological studies. Here, the authors report a divergent synthesis of C4-oxygenated securinega alkaloids via stereocontrolled diversifications on the piperidine core.
- Sangbin Park
- , Gyumin Kang
- & Sunkyu Han
-
Article
| Open Access Enantioselective construction of ortho-sulfur- or nitrogen-substituted axially chiral biaryls and asymmetric synthesis of isoplagiochin D
Enantioselective construction of ortho-sulfur- or nitrogen-substituted axially chiral biaryls and asymmetric synthesis of isoplagiochin D
Ortho-heteroatom-substituted axially chiral biaryls are valuable structures in synthetic and medicinal chemistry. Here, the authors established an efficient synthesis of these chiral structures via asymmetric cross-coupling.
- He Yang
- & Wenjun Tang
-
Article
| Open Access Enyne diketones as substrate in asymmetric Nazarov cyclization for construction of chiral allene cyclopentenones
Enyne diketones as substrate in asymmetric Nazarov cyclization for construction of chiral allene cyclopentenones
Developing classes of substrates for the Nazarov reaction is an important endeavour for synthetic chemistry. Here, the authors report how enyne diketones undergo rhodium-catalyzed metal-organo relay catalysis for the enantioselective construction of chiral allene cyclopentenones via Nazarov cyclisation in good yields and enantioselectivities.
- Shengbiao Tang
- , Peng Zhang
- & Jiangtao Sun
-
Article
| Open Access Synergistic Pd/Cu-catalyzed enantioselective Csp2–F bond alkylation of fluoro-1,3-dienes with aldimine esters
Synergistic Pd/Cu-catalyzed enantioselective Csp2–F bond alkylation of fluoro-1,3-dienes with aldimine esters
Due to high bond dissociation energies of C(sp2)–F bonds, using fluorinated compounds in C(sp2)–C(sp3) cross-coupling is difficult. Here the authors report a protocol for enantioselective C(sp2)–C(sp3) coupling of dienyl fluorides with aldimine esters, enabled by synergistic copper and palladium catalysis.
- Huimin Yu
- , Qinglong Zhang
- & Weiwei Zi
-
Article
| Open Access Kinetic resolution of cyclic benzylic azides enabled by site- and enantioselective C(sp3)–H oxidation
Kinetic resolution of cyclic benzylic azides enabled by site- and enantioselective C(sp3)–H oxidation
Catalytic kinetic resolution of racemates is among the best ways to prepare enantiopure compounds, but this process is difficult with organic azides given the small size of the azido group. Here, the authors show kinetic resolution of cyclic benzylic azides through site- and enantioselective C(sp3)–H oxidation.
- Pengbo Ye
- , Aili Feng
- & Lei Liu
-
Article
| Open Access Cu-catalyzed asymmetric addition of alcohols to β,γ-alkynyl-α-imino esters for the construction of linear chiral N,O-ketals
Cu-catalyzed asymmetric addition of alcohols to β,γ-alkynyl-α-imino esters for the construction of linear chiral N,O-ketals
N,O-acetals are part of many synthetic intermediates and important skeletons of numerous natural products and pharmaceutical drugs. Here the authors show a Cu-catalyzed asymmetric addition of alcohols to β,γ-alkynyl-α-imino esters, providing the corresponding linear chiral N,O-ketals with up to 96% ee.
- Cheng Sheng
- , Zheng Ling
- & Wanbin Zhang
-
Article
| Open Access Ir-catalyzed enantioselective B−H alkenylation for asymmetric synthesis of chiral-at-cage o‑carboranes
Ir-catalyzed enantioselective B−H alkenylation for asymmetric synthesis of chiral-at-cage o‑carboranes
Chiral-at-cage o-carboranes are clusters of carbon and boron atoms that, when functionalized, can lower the symmetry of the cluster. Here the authors present a method to alkenylate B–H bonds on ocarboranes enantioselectively via iridium catalysis.
- Ruofei Cheng
- , Jie Zhang
- & Zuowei Xie
-
Article
| Open Access Catalytic enantioselective reductive domino alkyl arylation of acrylates via nickel/photoredox catalysis
Catalytic enantioselective reductive domino alkyl arylation of acrylates via nickel/photoredox catalysis
Enantioenriched α-aryl propionic acids are an important class of nonsteroidal anti-inflammatory medications. Here the authors use a visible-light-promoted photoredox/nickel catalyzed method to form such compunds via a three-component alkyl arylation of acrylates.
- Pengcheng Qian
- , Haixing Guan
- & Jianyou Mao
-
Article
| Open Access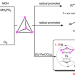 Pentaphosphaferrocene-mediated synthesis of asymmetric organo-phosphines starting from white phosphorus
Pentaphosphaferrocene-mediated synthesis of asymmetric organo-phosphines starting from white phosphorus
Asymmetrically substituted phosphine ligands play a decisive role in catalysis, but their synthesis is rather challenging and often involves hazardous chemicals such as PCl3 or PH3. Here, the authors report the one-pot preparation of asymmetric phosphines from white phosphorus via a pentaphosphaferrocene based modular system, in which the transition metal complex can be reused.
- Stephan Reichl
- , Eric Mädl
- & Manfred Scheer
-
Article
| Open Access A concise access to bridged [2,2,1] bicyclic lactones with a quaternary stereocenter via stereospecific hydroformylation
A concise access to bridged [2,2,1] bicyclic lactones with a quaternary stereocenter via stereospecific hydroformylation
Enantiomeric bridged [2,2,1] bicyclic lactone skeletons and their ring-opening products are important scaffolds widely occurring in both pharmaceutics and biology active compounds. Here the authors show an efficient method for enantioselective construction of these compounds bearing a quaternary stereocenter via Rh-catalyzed asymmetric hydroformylation/intramolecular cyclization/ pyridinium chlorochromate oxidation.
- Shuailong Li
- , Zhuangxing Li
- & Hui Lv
-
Article
| Open Access Enantioselective synthesis of tertiary boronic esters through catalytic asymmetric reversed hydroboration
Enantioselective synthesis of tertiary boronic esters through catalytic asymmetric reversed hydroboration
The development of conjugate boryl addition to α,β-unsaturated amide has been hampered by the intrinsic low electrophilicity of the amide group. Here the authors show a catalytic asymmetric synthesis of enantioenriched tertiary boronic esters through hydroboration of β,β-disubstituted α,β-unsaturated amides.
- Tao-Tao Gao
- , Hou-Xiang Lu
- & Bi-Jie Li
-
Article
| Open Access Unified divergent strategy towards the total synthesis of the three sub-classes of hasubanan alkaloids
Unified divergent strategy towards the total synthesis of the three sub-classes of hasubanan alkaloids
Several Hasubanan alkaloids have been synthesized in the past decades, however a divergent approach to access the 3 subclasses of such natural products has not been reported yet. Here, the authors show the enantioselective total syntheses of four representative members via a unified strategy leading to the three topologically different classes of alkaloids.
- Guang Li
- , Qian Wang
- & Jieping Zhu
-
Article
| Open Access Conversion of anilines to chiral benzylic amines via formal one-carbon insertion into aromatic C–N bonds
Conversion of anilines to chiral benzylic amines via formal one-carbon insertion into aromatic C–N bonds
Atom insertion into aromatic carbon-nitrogen (C-N) bonds is useful for the synthesis of nitrogen-containing molecules, but challenging due to the inert nature of these bonds. Here, the authors report one-carbon insertion into aromatic C-N bonds to directly convert anilines to chiral benzylic amines.
- Lei Li
- , Min Yang
- & Renhua Fan
-
Article
| Open Access Synthesis of chiral anti-1,2-diamine derivatives through copper(I)-catalyzed asymmetric α-addition of ketimines to aldimines
Synthesis of chiral anti-1,2-diamine derivatives through copper(I)-catalyzed asymmetric α-addition of ketimines to aldimines
Chiral 1,2-diamines are useful chemicals used as ligands in asymmetric catalysis. Here, the authors report a copper(I)-catalyzed asymmetric α-addition of ketimines derived from trifluoroacetophenone and 2- or 4-NO2-benzylamines to aldimines, affording a series of chiral anti-1,2-diamine derivatives.
- Xu-Cheng Gan
- , Cheng-Yuan Zhang
- & Liang Yin
-
Article
| Open Access Asymmetric total synthesis of yuzurimine-type Daphniphyllum alkaloid (+)-caldaphnidine J
Asymmetric total synthesis of yuzurimine-type Daphniphyllum alkaloid (+)-caldaphnidine J
Despite being known for more than 50 years, yuzurimine-type alkaloids have not been accessed by total synthesis. Here, the authors report the first enantioselective total synthesis of (+)-Caldaphnidine J, a highly complex yuzurimine-type Daphniphyllum alkaloid.
- Lian-Dong Guo
- , Yan Zhang
- & Jing Xu
-
Article
| Open Access Stereospecific Si-C coupling and remote control of axial chirality by enantioselective palladium-catalyzed hydrosilylation of maleimides
Stereospecific Si-C coupling and remote control of axial chirality by enantioselective palladium-catalyzed hydrosilylation of maleimides
Catalytic asymmetric hydrosilylation of internal alkenes has proven elusive due to more favourable double bond reduction or isomerization. Here, the authors show an enantioselective Si-C coupling by hydrosilylation of activated alkenes using a palladium/phosphoramidite catalyst affording axially chiral succinimides.
- Xing-Wei Gu
- , Yu-Li Sun
- & Li-Wen Xu
-
Article
| Open Access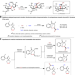 Constructing chiral bicyclo[3.2.1]octanes via palladium-catalyzed asymmetric tandem Heck/carbonylation desymmetrization of cyclopentenes
Constructing chiral bicyclo[3.2.1]octanes via palladium-catalyzed asymmetric tandem Heck/carbonylation desymmetrization of cyclopentenes
Tandem Heck/carbonylation reaction gives access to ubiquitous carbonyl molecules, however the asymmetric version is rarely studied. Here, the authors synthesize chiral bicyclo[3.2.1]octanes with a palladium-catalyzed asymmetric tandem Heck/carbonylation desymmetrization of cyclopentenes with alcohols, phenols and amines.
- Zhenbo Yuan
- , Yuye Zeng
- & Hequan Yao
-
Article
| Open Access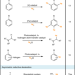 Bringing biocatalytic deuteration into the toolbox of asymmetric isotopic labelling techniques
Bringing biocatalytic deuteration into the toolbox of asymmetric isotopic labelling techniques
Deuterated chemicals are widely exploited in analytical chemistry and increasingly in the synthesis of pharmaceutical compounds. Here, the authors developed a mild biocatalytic method for the selective asymmetric reductive deuteration of organic compounds, by using H2 and 2H2O to generate 2H-NADH.
- J. S. Rowbotham
- , M. A. Ramirez
- & K. A. Vincent
-
Article
| Open Access Copper-catalyzed enantioselective Sonogashira-type oxidative cross-coupling of unactivated C(sp3)−H bonds with alkynes
Copper-catalyzed enantioselective Sonogashira-type oxidative cross-coupling of unactivated C(sp3)−H bonds with alkynes
Enantioselective oxidative cross-coupling of unactivated C(sp3)−H bonds and terminal alkynes is challenging. Here, the authors developed a copper/cinchona alkaloid catalyst for the asymmetric Sonogashira-type alkynylation of C(sp3)-H bonds via radical intermediates.
- Zhen-Hua Zhang
- , Xiao-Yang Dong
- & Xin-Yuan Liu
-
Article
| Open Access Chiral donor–acceptor azetines as powerful reactants for synthesis of amino acid derivatives
Chiral donor–acceptor azetines as powerful reactants for synthesis of amino acid derivatives
Chiral 3-azetidinones are structural analogues of medicinally relevant β-lactams, however their synthesis and reactivity are underexplored. Here, the authors show a highly enantioselective copper-catalyzed [3 + 1]-cycloaddition generating 2-azetidines, which react with nucleophiles yielding amino acids via 3-azetidinones.
- Kostiantyn O. Marichev
- , Kuiyong Dong
- & Michael P. Doyle
-
Article
| Open Access Synergetic iridium and amine catalysis enables asymmetric [4+2] cycloadditions of vinyl aminoalcohols with carbonyls
Synergetic iridium and amine catalysis enables asymmetric [4+2] cycloadditions of vinyl aminoalcohols with carbonyls
Inverse electron-demand cycloadditions in palladium catalysis are inherently limited by the linear selectivity in the allylic substitution step. To overcome this issue, the authors report a synergetic iridium (branched-selective) and amine catalysis in [4+2] cycloadditions of vinyl aminoalcohols with carbonyls.
- Mao-Mao Zhang
- , Ya-Ni Wang
- & Wen-Jing Xiao
-
Article
| Open Access Asymmetric synthesis of tetrazole and dihydroisoquinoline derivatives by isocyanide-based multicomponent reactions
Asymmetric synthesis of tetrazole and dihydroisoquinoline derivatives by isocyanide-based multicomponent reactions
Asymmetric isocyanide-based multicomponent reactions are elegant, yet challenging, strategies to access valuable N-heterocycles. Here, the authors employ a chiral Mg(II) -N,N′-dioxide catalyst in three- or four-component reactions to obtain chiral tetrazoles and devise a dearomative [3+2] annulation reaction of isoquinolines.
- Qian Xiong
- , Shunxi Dong
- & Xiaoming Feng
-
Article
| Open Access A unified strategy toward total syntheses of lindenane sesquiterpenoid [4 + 2] dimers
A unified strategy toward total syntheses of lindenane sesquiterpenoid [4 + 2] dimers
Of the 3 classes of dimeric lindenane sesquiterpenoids, only type I And II have been targeted in total synthesis. Here, the authors report a unified strategy to synthetically access the 3 classes of lindenane sesquiterpenoids dimers by developing a [4 + 2] cycloaddition between a common furyl diene and various dienophiles.
- Biao Du
- , Zhengsong Huang
- & Bo Liu
-
Article
| Open Access Catalytic enantioselective oxidative coupling of saturated ethers with carboxylic acid derivatives
Catalytic enantioselective oxidative coupling of saturated ethers with carboxylic acid derivatives
Cross-dehydrogenative coupling (CDC) is a powerful method for C-C bond formation however the enantioselective variant is underdeveloped. Here, the authors show a formal enantioselective CDC method involving unactivated ethers and carboxylic acid derivatives allowing for the rapid preparation of biologically active molecules.
- Gang Wang
- , Xiaodong Xin
- & Lei Liu
-
Article
| Open Access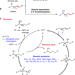 Asymmetric remote C-H borylation of internal alkenes via alkene isomerization
Asymmetric remote C-H borylation of internal alkenes via alkene isomerization
Sequential alkene isomerization/functionalization enables enantioselective transformations of remote C–H bonds. Here, the authors report a chiral cobalt catalytic system for the highly enantioselective, remote C–H borylation of internal alkenes via an isomerization/hydroboration sequence.
- Xu Chen
- , Zhaoyang Cheng
- & Zhan Lu
-
Article
| Open Access Symmetry breakdown of 4,4″-diamino-p-terphenyl on a Cu(111) surface by lattice mismatch
Symmetry breakdown of 4,4″-diamino-p-terphenyl on a Cu(111) surface by lattice mismatch
In a symmetric molecule with identical functional groups, selective activation of only one site is challenging. Here, the authors show that 4,4″-diamino-p-terphenyl adsorbs asymmetrically to a metal surface, leading to a change in binding affinity of one of its amine groups.
- Qigang Zhong
- , Daniel Ebeling
- & André Schirmeisen
-
Article
| Open Access Silicon-oriented regio- and enantioselective rhodium-catalyzed hydroformylation
Silicon-oriented regio- and enantioselective rhodium-catalyzed hydroformylation
Hydroformylation of 1,2-disubstituted alkenes usually occurs at the α position of the directing heteroatom. Here, the authors report the asymmetric rhodium-catalyzed hydroformylation of 1,2-disubstituted alkenylsilanes with excellent regioselectivity at the β position (relative to the silicon heteroatom) and enantioselectivity.
- Cai You
- , Xiuxiu Li
- & Xumu Zhang
-
Article
| Open Access Enantioselective reduction of sulfur-containing cyclic imines through biocatalysis
Enantioselective reduction of sulfur-containing cyclic imines through biocatalysis
The 3-thiazolidine ring, a pharmaceutically interesting cyclic structural element found e.g. in some antibiotics, is hard to obtain via currently used approaches. Here, the authors developed a straightforward method to efficiently synthesize a variety of defined, pure 3-thiazolidines.
- Nadine Zumbrägel
- , Christian Merten
- & Harald Gröger
-
Article
| Open Access Dual catalysis for enantioselective convergent synthesis of enantiopure vicinal amino alcohols
Dual catalysis for enantioselective convergent synthesis of enantiopure vicinal amino alcohols
Chiral vicinal amino alcohols are found in many bioactive compounds and may serve as chiral ligands. Here, the authors report a photocatalytic enantioselective cross-coupling of nitrones with aromatic aldehydes with a chiral ligand-coordinated rare earth ion synergistically producing enantiopure vicinal amino alcohols.
- Chen-Xi Ye
- , Yared Yohannes Melcamu
- & Pei-Qiang Huang
-
Article
| Open Access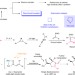 Catalytic asymmetric synthesis of a nitrogen heterocycle through stereocontrolled direct photoreaction from electronically excited state
Catalytic asymmetric synthesis of a nitrogen heterocycle through stereocontrolled direct photoreaction from electronically excited state
Despite intensive research on photoexcited molecules, stereocontrol of direct bond formation upon photoexcitation remains limited. Here the authors expand the research on stereocontrolled bond forming photochemistry and introduce the catalytic asymmetric synthesis of chiral nitrogen heterocycles.
- Xiaoqiang Huang
- , Xinyao Li
- & Eric Meggers
-
Article
| Open Access Pulsed electroconversion for highly selective enantiomer synthesis
Pulsed electroconversion for highly selective enantiomer synthesis
Synthesis of chiral molecules mostly relies on asymmetric catalysis. Here, the authors developed a pulsed electrochemical method to convert a prochiral ketone into the corresponding chiral alcohols with very high enantioselectivity on chiral-imprinted mesoporous platinum.
- Chularat Wattanakit
- , Thittaya Yutthalekha
- & Alexander Kuhn

 Palladium-catalyzed asymmetric carbene coupling en route to inherently chiral heptagon-containing polyarenes
Palladium-catalyzed asymmetric carbene coupling en route to inherently chiral heptagon-containing polyarenes