« Prev Next »
The Golgi apparatus is the central organelle mediating protein and lipid transport within the eukaryotic cell. Typically textbooks illustrate the Golgi as something resembling a stack of pita bread. However, this depiction does not adequately illustrate the dynamic nature of the Golgi compartments (called cisternae) or the variety of morphologies the Golgi manifests in different cell types. We can learn a lot by simply asking why these diverse structures even exist. Researchers do not yet fully understand how various Golgi morphologies affect its function. However, scientists are currently using the subtle variations in Golgi morphology among different cell types to ask how proteins move through the Golgi apparatus.
What Happens To Proteins As They Move Through the Golgi?
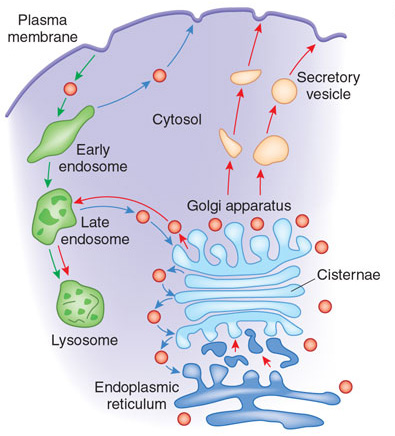
Each cisterna or region of the Golgi contains different protein modification enzymes. What do these enzymes do? The Golgi enzymes catalyze the addition or removal of sugars from cargo proteins (glycosylation), the addition of sulfate groups (sulfation), and the addition of phosphate groups (phosphorylation). Cargo proteins are modified by enzymes (called resident enzymes) located within each cisterna. The enzymes sequentially add the appropriate modifications to the cargo proteins. Some Golgi-mediated modifications act as signals to direct the proteins to their final destinations within cells, including the lysosome and the plasma membrane. What happens when there are defects in Golgi function? Defects in various aspects of Golgi function can result in congenital glycosylation disorders, some forms of muscular dystrophy, and may contribute to diabetes, cancer, and cystic fibrosis (Ungar 2009).
How do cargo proteins move between the Golgi cisternae? Scientists have proposed two possible explanations: the vesicular transport model and cisternal maturation model. Interestingly, both models account for the Golgi's steady state conditions and processes, yet they do so quite differently (Figure 2). In 2002 James Rothman and Randy Schekman won the Lasker Prize for their groundbreaking work detailing the membrane and vesicle systems that make secretion possible in eukaryotic cells. These two scientists worked independently using different model organisms and different biological approaches (Strauss 2009). Together they delivered strong evidence that there are common molecules and processes involved in membrane fusion and fission in eukaryotes. Rothman and his colleagues biochemically reconstituted mammalian Golgi membranes, isolating vesicles capable of moving from one cisterna to another. As a different approach, Schekman and his colleagues used yeast genetics to identify and characterize many of the important proteins involved in secretion in this single-celled eukaryote. Over time Rothman and Schekman's work converged on several important molecules that were involved in vesicle formation and fusion, thus leading to what came to be called the vesicular transport model.
The Vesicular Transport Model: Evidence
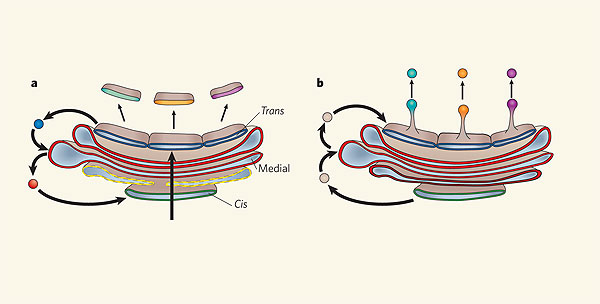
One of the principal observations by Rothman's group was that the vesicles that formed in the Golgi moved cargo proteins between cisternae from the cis face to the trans face. These observations suported the vesicular transport model originally developed and advocated by George Palade and Marilyn Farquhar (Farquhar & Palade 1998.) The vesicular trasnport model posits that the Golgi cisternae are stable compartments that house certain protein modification enzymes that function to add or remove sugars, add sulfate groups, and perform other modifications. Vesicles arrive at each cisterna carrying cargo proteins, which are then modified by the resident enzymes located within that cisterna. Next, new vesicles carrying the cargo proteins bud from the cisterna and travel to the next stable cisterna, where the next series of enzymes further processes the protein cargo (Rothman & Wieland 1996).
The Cisternal Maturation Model
Before the work of Palade, Farquhar, Rothman and others who analyzed the vesicles moving proteins between Golgi cisternae, scientists thought that each Golgi cisterna was transient and that the cisternae themselves moved from the cis to the trans face of the Golgi, changing over time. The movement of proteins as passengers within cisternae through the Golgi stack is called the cisternal maturation model. This model proposes that the enzymes present in each individual cisterna change over time, while the cargo proteins remain inside the cisterna. Before Rothman's work on vesicles, this model had broad support. However, once scientists identified the large numbers of small transport vesicles surrounding the Golgi, researchers developed the vesicular transport model as an updated replacement. However, as often happens in science (and in fashion), old ideas sometimes come back in new ways.
What New Evidence Supports the Cisternal Maturation Model?
In the 1990s scientists studied multiple cell types to expand our understanding of the Golgi. Alberto Luini and his colleagues used cultured mammalian cells to investigate how large protein complexes moved through the Golgi. The researchers used immunoelectron microscopy to follow the pathway that rigid, 300 nm, rod-shaped, procollagen trimers took through the Golgi in mammalian fibroblasts. Luini and his colleagues observed procollagen only within Golgi cisternae, and never within the vesicles, which are normally much smaller (<100 nm diameter) than procollagen (Bonfanti et al. 1998). Other researchers, including Michael Melkonian and his colleagues, observed similar results when studying the Golgi apparatus of algae. Several types of flagellated protists construct and export scales that attach to the cell surface of these organisms. The scales have diverse but defined sizes and shapes. Researchers observed that in different species of algae that export both very large (1.5–2 mm) and more moderately sized (~40 nm) scales, the scales were consistently found within the cisternae, but not in the transport vesicles (Becker, Bolinger & Melkonian 1995; Becker & Melkonian 1996). The results from these diverse cell types support the cisternal maturation model of protein transport through the Golgi.
What were all the vesicles Rothman discovered doing
in the Golgi? The current cisternal maturation model proposes that these
vesicles are transport vehicles for Golgi enzymes rather than for protein
cargo. Retrograde vesicles that travel backward through the Golgi bud off of a cisterna
to transfer enzymes to younger cisternae. Meanwhile other vesicles, arriving
from older cisternae, carry the enzymes necessary for the next steps in protein
modification (Glick & Malhotra 1998; Pellham 1998).
Which Model Is More Accurate?
What Questions Remain about Protein Traffic through the Golgi?
Although researchers generally agree that the cisternal maturation model best fits the current data, there is still some debate over whether or not all cargo proteins take the same path. Jennifer Lippincott-Swartz and her colleagues pioneered fluorescence methods to quantitatively measure the dynamics of cellular membranes, including the Golgi. Using these methods, they learned that some cargo proteins travel through the Golgi more slowly than the rates at which the cisternae mature (Patterson et al. 2008). The researchers concluded that the cisternal maturation model could not accurately account for their data. While they do not dispute cisternal maturation, they additionally proposed a model whereby a two-phase system of membranes determines which cargo proteins and Golgi enzymes must distribute themselves during transport. Complicating the situation further, at least some cell types have connections between different cisternae within the Golgi stack (e.g. between cis and medial cisternae.) For example, Luini and colleagues observed intercisternal continuities during waves of protein traffic in mammalian cells (Trucco et al. 2004), and consequently proposed a modified version of the cisternal maturation model. Many investigators will continue to investigate and refine these new models over time.
While some aspects of protein transport through the Golgi are better understood than they used to be, there are still many unresolved issues surrounding the specifics within different organisms. Moreover, questions remain about the unifying characteristics that are shared between all Golgi. A recent gathering of prominent Golgi researchers identified several important questions to be addressed in the future, including:
- Do different types of secretory cargo follow distinct routes through the Golgi?
- What molecular mechanisms drive and regulate cisternal maturation?
- Are there specialized domains in the Golgi cisternae? How are they created, and what roles do they play in cargo sorting and export?
- How are the Golgi compartments constructed and remodeled?
-
Is Golgi
stacking fundamentally important for membrane traffic? If so, how do organisms
such as S. cerevisiae bypass this
requirement?" (Emr et al. 2009)
Summary
The structure of the Golgi apparatus varies in different cell types. The dispersed nature of Golgi cisternae in the yeast Saccharomyces cerevisiae allowed researchers to resolve individual cisternae. By observing fluorescently labeled proteins that normal reside within different cisternae, researchers found convincing evidence that the Golgi cisternae change over time, supporting the cisternal maturation model of protein movement through the Golgi apparatus. However, there is clearly much left to discover about the Golgi.
References and Recommended Reading
Alberts, B. et al. Molecular Biology of the Cell, 5th ed. New York: Garland Science, 2008.
Becker, B. & Melkonian, M. The secretory pathway of protists: Spatial and functional organization and evolution. Microbiological Reviews 60, 697–721 (1996).
Becker, B., Bolinger, B. & Melkonian, M. Anterograde transport of algal scales through the Golgi complex is not mediated by vesicles. Trends in Cell Biology 5, 305–307 (1995) doi: 10.1016/S0962-8924(00)89047-9.
Bonfanti, L. et al. Procollagen traverses the Golgi stack without leaving the lumen of cisternae: Evidence for cisternal maturation. Cell 95, 993–1003 (1998) doi:10.1016/S0092-8674(00)81723-7.
Emr, S. et al. Journeys through the Golgi — Taking stock in a new era. Journal of Cell Biology 187, 449–453 (2009) doi: 10.1083/jcb.200909011.
Farquhar, M. G. & Palade, G. E. The Golgis apparatus: 100 years of progress and controversy. Trends in Cell Biology 8, 2–10 (1998) doi: 10.1016/S0962-8924(97)01187-2.
Glick, B. S. & Malhotra, V. The curious status of the Golgi apparatus. Cell 95, 883–889 (1998) doi:10.1016/S0092-8674(00)81713-4.
Glick, B. S. & Nakano, A. Membrane traffic within the Golgi apparatus. Annual Review of Cell and Developmental Biology 25, 113–132 (2009) doi:10.1146/annurev.cellbio.24.110707.175421.
Karp, G. Cell and Molecular Biology: Concepts and Experiments, 6th ed. New York: John Wiley and Sons, 2009.
Losev, E. et al. Golgi maturation visualized in living yeast. Nature 22, 1002–1006 (2006) doi:10.1038/nature04717.
Malhotra, V. & Mayor, S. Cell biology: The Golgi grows up. Nature 441, 939–940 (2006) doi:10.1038/441939a.
Matsuura-Tokita, K. et al. Live imaging of yeast Golgi cisternal maturation. Nature 22, 1007–1010 (2006) doi:10.1038/nature04737.
Patterson, G. H. et al. Transport through the Golgi apparatus by rapid partitioning within a two-phase membrane system. Cell 133, 1055–1067 (2008) doi: 10.1016/j.cell.2008.04.044.
Pelham, H. R. B. Getting through the Golgi complex. Trends in Cell Biology 8, 45–49 (1998) doi:10.1016/S0962-8924(97)01185-9.
Rothman, J. E. & Wieland, F. T. Protein sorting by transport vesicles. Science 272, 227–234 (1996) doi: 10.1126/science.272.5259.227.
Strauss, E. Albert Lasker Basic Medical Research Award 2002 - Award Description — James Rothman and Randy Schekman. Lasker Foundation Website (2009).
Trucco, A. et al. Secretory traffic triggers the formation of tubular continuities across Golgi sub-compartments. Nature Cell Biology 6, 1071–1081 (2004) doi: 10.1038/ncb1180.
Ungar, D. Golgi linked protein glycosylation and associated diseases. Seminars in Cell and Developmental Biology 20, 762–769 (2009).
Xu, D. & Esko, J. D. A Golgi-on-a-chip for glycan synthesis. Nature Chemical Biology 5, 612–613 (2009) doi: 10.1038/nchembio0909-612.






























