« Prev Next »

Plant fitness revolves around the ability to gain carbon for growth and reproduction. Photosynthetic carbon is used to build structures for the acquisition of nutrients and water (roots), to compete for light (stems, leaves), and for defense against enemies (toxins, spines, etc.). Other factors being equal, greater photosynthetic carbon gain should lead to higher growth rates and reproductive output.
Photosynthetic carbon assimilation (A) can be viewed as a diffusion process whereby a concentration gradient in carbon dioxide (CO2) is multiplied by the conductance to CO2 (gCO2):
A = gCO2 (Ca - Ci)
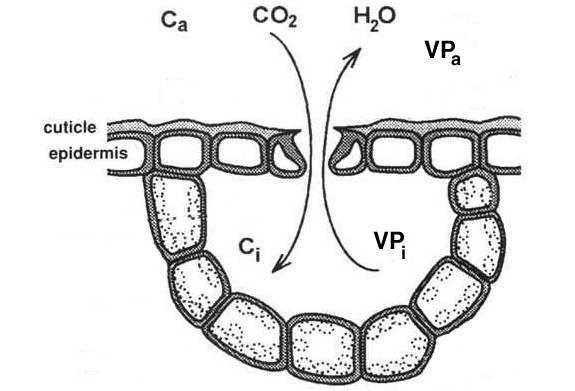
The concentration gradient is the difference in concentration between the CO2 in the surrounding air (Ca) and inside the leaf (Ci) (Figure 1). CO2 concentration inside an illuminated leaf is less than the concentration outside the leaf because of the biochemical fixation of carbon into carbohydrates during photosynthesis. Hence CO2 will diffuse down this concentration gradient into the leaf. The conductance to CO2 (gCO2) is a composite term made up of the rate of diffusion through the stomata, diffusion across the intercellular air spaces into the chloroplasts, and the rate of enzymatic fixation of carbon.
One way to maximize photosynthesis is to maximize gCO2. There are two ways plants can achieve this. First, they can minimize the resistance to diffusion of CO2 through the stomata (i.e., have lots of large, open stomata). Second, they can maximize the biochemical rate of fixation (i.e., have high concentrations of photosynthetic enzymes).
However, there are tradeoffs to maximizing the rate of photosynthesis. First, photosynthetic enzymes require nitrogen, which is often a limiting resource. So there are limits to how much carboxylating enzyme plants can make. Second, transpiration — the evaporation of water from the inside of the leaf to the surrounding air — also involves diffusion through the stomata (Figure 1). Transpiration can be expressed in much the same way as photosynthesis (where E = transpiration rate, 1.6 = the ratio of the diffusivity of water to CO2, VPa = water vapor pressure of the air outside the leaf, and VPi = water vapor pressure inside the leaf):
E = 1.6 gCO2 (VPa - VPi)
The air inside of leaves is saturated with water vapor, making VPi much higher than VPa and causing water to diffuse from inside the leaf to the outside. Maximizing gCO2 thus maximizes transpiration as well as photosynthesis. In dry habitats, this could result in dehydration and death. Terrestrial plants, therefore, face an evolutionary dilemma: how to gain carbon for growth and reproduction without first losing so much water that they dehydrate and die?
One measure of a plant's success in solving this dilemma is transpiration efficiency, the ratio of photosynthesis to transpiration (A/E). Transpiration efficiency can be calculated by combining the first two equations:
A/E = (Ca - Ci)/1.6 (VPa - VPi)
This equation highlights the point that transpiration efficiency is partially determined by the environment (VPa - VPi) — the larger the gradient between vapor pressure inside the leaf and in the surrounding air, the lower A/E will be. The difference between VPa and VPi is largest when temperatures are high and the air is dry. Thus, it is difficult for plants to achieve high A/E in such environments. Plants show a variety of adaptations to hot, dry conditions, such as vertical leaf angles and white, reflective leaves that help to decrease midday leaf temperatures. In addition, many plants reduce water loss through thick, waxy cuticles covering their leaf epidermis as well as by stomatal closure.
Transpiration efficiency is also a function of the CO2 concentration gradient, Ca - Ci. Ambient CO2 concentrations (Ca) are relatively constant, but the intercellular CO2 concentration (Ci) is determined by the balance between CO2 supply through the stomata, and CO2 uptake by photosynthesis. Thus, plants can change Ci through their physiological responses to the environment. The more stomata restrict the diffusion of CO2, the lower Ci will be, leading to higher A/E (all other factors being equal). So high Ci is associated with low A/E and low Ci is associated with high A/E, at constant vapor pressure differences. There are also tradeoffs with photosynthesis implied by this relationship. Restricting CO2 diffusion will limit photosynthesis as well as transpiration, so often high A/E in plants is associated with slow growth and low photosynthetic rates.
C3 Photosynthetic Pathway
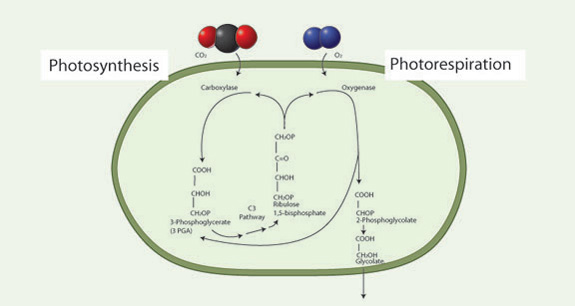
The first photosynthetic pathway that was biochemically characterized is called the C3 pathway because the initial products of carbon fixation are 3-carbon compounds (phosphoglyceric acid, PGA) (Figure 2). CO2 is combined with a 5-carbon sugar, ribulose bisphosphate (RuBP), using the enzyme ribulose bisphosphate carboxylase/oxygenase (rubisco):
RuBP + CO2 → 2PGA
Rubisco is the most common soluble protein in the world and can account for 50% of a leaf's nitrogen content. Rubisco also catalyzes a second reaction, that of oxygen with RuBP (Figure 3):
RuBP + O2 → PGA + PG
This reaction leads to no net gain of carbon for the plant. One of the products of this reaction, 2-phosphoglycolate (PG) is further metabolized in the photorespiratory carbon oxidation cycle, which leads to the release of CO2 in the light, a process called photorespiration.

Photorespiration reduces net photosynthesis in two ways. First, there is a competition between O2 and CO2 for the active site of rubisco. Those active sites that take up O2 cannot take up CO2. Second, there is the release of CO2 in the PCO cycle. These two mechanisms can reduce net photosynthesis between 35–50% depending upon environmental conditions. Photorespiration increases with increasing temperature due to the fact that the solubility of O2 decreases more slowly than the solubility of CO2 with increasing temperature. When environmental conditions lead to stomatal closure and lower Ci, O2 will gain a competitive binding advantage over CO2. Thus, warm and arid habitats lead to greater photorespiration. Scientists view this as a primary selective force in the evolution of alternative photosynthetic pathways.
C4 Photosynthetic Pathway
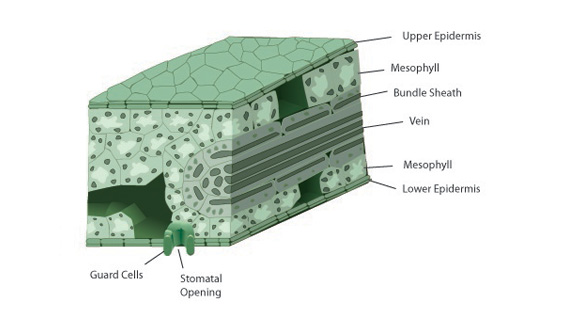
The C4 photosynthetic pathway differs from the C3 pathway in its initial carboxylating enzyme and initial products (Figure 4). The primary carboxylating enzyme in C4 plants is phosphoenolpyruvate (PEP) carboxylase. This enzyme has a higher affinity for CO2 than rubisco and has no oxygenase activity. The enzyme catalyzes the reaction of CO2 with PEP, forming a 4-carbon acid. C4 plants still have C3 biochemistry; however, there is a physical separation of enzymes. The outer, mesophyll cells have PEP carboxylase and few chloroplasts while rubisco and many chloroplasts are located in bundle sheath cells, a jacket of cells surrounding the xylem and phloem (Figure 5).
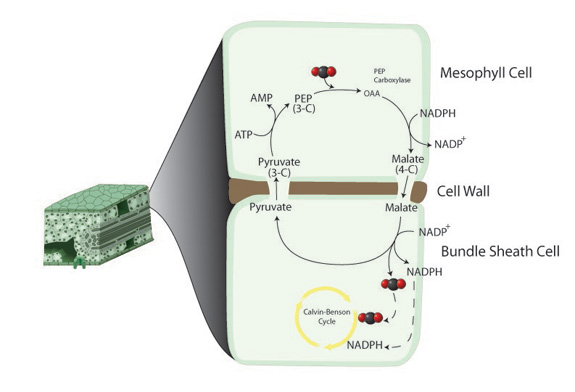
The physical separation of biochemical activities acts as a CO2 concentrating mechanism for rubisco. After CO2 is combined with PEP, the C4 acid is transferred to the bundle sheath cells, where it is decarboxylated. CO2 is released next to rubisco in the bundle sheath cells, providing it with a competitive advantage over O2. C4 plants show no photorespiration in intact leaves, although their rubisco still has oxygenase activity. Any CO2 that is released in the bundle sheath cells is fixed by PEP carboxylase in the mesophyll cells before it can diffuse out of the leaf.
Reduced photorespiration gives a photosynthetic advantage to C4 plants relative to C3 plants in conditions where photorespiration is high (i.e., in low CO2 concentrations and/or in high temperatures). Because PEP carboxylase has a higher affinity for CO2, it can work efficiently at low intercellular concentrations of CO2. Thus, for a given photosynthetic rate, stomatal conductance can be lower, decreasing transpiration and increasing transpiration efficiency.
Crassulacean Acid Metabolism (CAM) Photosynthesis
The biochemistry of the CAM photosynthetic pathway is similar to that of C4 plants. However, rather than a physical separation of biochemistry, enzyme activities are separated in time (Figure 6). In this pathway, stomata open at night, which allows CO2 to diffuse into the leaf to be combined with PEP and form malate. This acid is then stored in large central vacuoles until daytime. During the day, malate is released from the vacuoles and decarboxylated. Rubisco then combines the released CO2 with RuBP in the C3 pathway. In CAM plants, photosynthesis is proportional to vacuolar storage capacity, so CAM plants usually have thick and fleshy water-storing leaves or stems (i.e., they are succulents).

Ecological Significance Of Pathway Variation
Because of the biochemical and morphological differences among the three photosynthetic pathways, each has its own unique set of advantages and disadvantages. These differences result in differential performance in different environments.
The C3 pathway is the oldest (originating around 2,800 million years ago) and is also the most widespread, both taxonomically and environmentally. C3 plants can be found in the warmest deserts and the coldest arctic habitats.
C4 photosynthesis has evolved independently approximately fifty times but is most widespread in grasses where about 50% of the species are C4 photosynthesizers (Edwards and Smith 2010). C4 photosynthesis occurs in 3% of vascular plant species, but accounts for 25% of terrestrial photosynthesis (Sage,2004). It is thus a relatively recent innovation, with the earliest appearances occurring during the low atmospheric CO2 levels that occurred in the mid-Oligocene (approximately thirty million years ago). C4 plants reach their peak of productivity in high-light and warm climates with summer rains (Figure 7), dominating tropical and subtropical grasslands and savannas whereas C3 grasses dominate cooler, temperate grasslands. Because C4 photosynthesis is saturated at lower CO2 concentrations and allows lower stomatal conductance for a given photosynthetic rate, C4 plants also perform well in saline and dry habitats. Edwards and Smith (2010) found that eighteen of twenty evolutionary origins of C4 grasses were correlated with reductions in mean annual precipitation, consistent with a shift out of tropical forests into open, high-light tropical savannas as the global climate became drier in the Oligocene. Australia is now one of the driest continents in the world, and a survey of over a thousand grass species found that C4 grasses dominate 80–85% of the continent (Hattersley 1983). But even within Australia there are differences in pathway distribution, with C4 species being most numerous where the summer is hot and wet while C3 species are most common where the spring is cool and wet (extreme southern Australia and Tasmania). Studies along environmental gradients of temperature and precipitation have often highlighted differences between C3 and C4 species. For example, Tieszen et al. (1979) used an altitudinal gradient to show that the percent of grass species with the C3 pathway in African savannas increased with altitude as precipitation increased and temperature decreased. Cabido et al. (1997) found a similar pattern in the temperate grasslands of Argentina (Table 1).

C4 plant species also have a potential advantage over C3 species in low-nutrient habitats. Rubisco can account for 25–30% of leaf nitrogen in C3 plants, but C4 plants contain three to six times less rubisco. The higher affinity of PEP carboxylase for CO2 means that C4 plants don't need as much enzyme, so overall leaf nitrogen is often less than half that found in C3 plants. However, this reduction in leaf nitrogen does not result in lower maximal photosynthetic rates; thus, C4 plants have higher photosynthetic-nitrogen-use efficiency (ratio of photosynthesis to leaf nitrogen) than C3 plants. This gives them an advantage in high-light, low-nutrient environments.
|
Altitude (m) |
350 |
600 |
1,000 |
1,400 |
1,600 |
1,800 |
1,900 |
2,100 |
| % of C3 species | 2.3 | 10.0 |
35.8 |
39.2 |
60.6 | 65.6 | 69.2 | 80.0 |
|
% of C4 species |
97.7 | 90.0 | 64.2 | 60.8 | 38.2 |
34.4 |
30.8 |
20.0 |
|
Table 1: The percentage of C3 and C4 grass species at different elevations in central Argentina
Source: Cabido et al. 1997 | ||||||||
Despite the advantages outlined above for C4 species, there are several disadvantages to this form of photosynthesis. For example, the regeneration of PEP in the C4 pathway (Figure 4) leads to higher costs for ATP than in the C3 pathway. ATP for photosynthetic biochemistry is supplied by the light reactions. The extra cost of C4 biochemistry is a disadvantage where low light occurs, as found in inner-canopy competitive situations in cool, wet forests. It is rare to find C4 plants in such environments.
CAM photosynthesis is found in more than 7% of vascular plant species, and has evolved independently several times. Due to their stomata being open at night when the vapor pressure differences between the leaf and the surrounding air are lowest (reducing transpiration), CAM photosynthetic plants have higher transpiration efficiencies than either C3 or C4 plants. Distribution patterns for CAM plants reflect this and are dominated by habitat aridity. Water storing cacti are adapted to enduring long periods with no precipitation. This is because they close their stomata, even at night during prolonged periods of drought and can refix CO2 lost in respiration before it diffuses out of the leaf or stem. Consequently, some cactus species lose very little biomass over months without rain.
An interesting aspect of CAM distribution is the high number of CAM species found in tropical environments. Epiphytes are plants that do not have their roots in the ground but instead grow on other plants (primarily trees). Thus, they frequently lack a ready supply of liquid water. Many epiphytic species in tropical and subtropical regions, such as orchids and bromeliads, therefore exhibit the CAM pathway.
A disadvantage for CAM plants is that they often have low photosynthetic capacity, slow growth, and low competitive abilities because their photosynthetic rates are limited by vacuolar storage capacity and by greater ATP costs, similar to those for C4 species. However, there is great plasticity in the expression of CAM photosynthesis. Many CAM plants can function in a C3 mode with stomata open during the day when water is available, so low photosynthetic and growth rates are not always limiting factors.
An interesting example that illustrates another advantage for the nighttime activity of CAM photosynthesis is found in aquatic plants such as Isoetes howellii. This plant is found in temporary vernal ponds in the western United States in which CO2 is depleted on warm, sunny days by other submerged aquatic plants. Since CO2 diffuses very slowly in water and its solubility decreases with increasing temperature, these habitats can easily become CO2 depleted. Isoetes howellii, with its CAM photosynthesis, takes advantage of higher CO2 concentrations in the water at night when other plants are releasing CO2 through respiration. In a similar fashion, the advantage of C4 photosynthesis in low CO2 conditions is illustrated by some submerged aquatic plants, such as Hydrilla verticillata. Hydrilla uses C4 photosynthesis to cope with the reduced CO2 concentrations that occur in warm, high-light aquatic environments in the summer.
Photosynthetic Pathway Plasticity
There are many plants that can switch between a C3 mode and a CAM mode. This is called facultative CAM. For example, Mesembryanthemum crystallinum (the ice plant) is an annual plant that switches photosynthetic mode in response to water and/or salt stress. It starts life using the C3 pathway but then switches to CAM later during flowering or under stress. This plant is native to Mediterranean type climates with cool, wet springs and hot, dry summers. This plasticity allows facultative CAM species to utilize the C3 pathway when water is available, which increases photosynthetic rates and growth, and then increase their stress tolerance during drier time periods through the CAM pathway.
In summary, there is considerable variety in the biochemistry of photosynthesis in the plant kingdom. Plants have evolved multiple photosynthetic pathways in response to low CO2 concentrations, high temperatures, and limitations in the availability of water. This physiological variation is combined with many other morphological and anatomical adaptations to allow plants to exploit a wide variety of aquatic and terrestrial habitats around the world.
References and Recommended Reading
Bloom, A. J. & Troughton, J. H. High productivity and photosynthetic flexibility in a CAM plant. Oecologia 38, 35–43 (1979).
Cabido, M. et al. Distribution of C3 and C4 grasses along an altitudinal gradient in central Argentina. Journal of Biogeography 24, 197–204 (1997).
Crayn, D. M. et al. Multiple origins of crassulacean acid metabolism and the epiphytic habit in the Neotropical family Bromeliaceae. Proceedings of the National Academy of Sciences 101, 3703–3708 (2004).
Dodd, A. N. et al. Crassulacean acid metabolism: plastic, fantastic. Journal of Experimental Botany 53, 569–580 (2002).
Edwards, E. J. & Smith, S. A. Phylogenetic analyses reveal the shady history of C4 grasses. Proceedings of the National Academy of Sciences 107, 2532–2537 (2010).
Edwards, E. J. & Still, C. J. Climate, phylogeny, and the ecological distribution of C4 grasses. Ecology Letters 11, 266–276 (2008).
Ehleringer, J. R. et al. C-4 photosynthesis, atmospheric CO2 and climate. Oecologia 112, 285–299 (1997).
Ehleringer, J. R. & Monson, R. K. Evolutionary and ecological aspects of photosynthetic pathway variation. Annual Review of Ecology and Systematics 24, 411–439 (1993).
Hattersley, P. W. The distribution of C3 and C4 grasses in Australia in relation to climate. Oecologia 57, 113–128 (1983).
Osborne, C. P. Atmosphere, ecology and evolution: what drove the Miocene expansion of C4 grasslands? Journal of Ecology 96, 35–45 (2008).
Osborne, C. P. & Freckleton, R. P. Ecological selection pressures for C4 photosynthesis in the grasses. Proceedings of the Royal Society B 276, 1753–1760 (2009).
Reiskind, J. B. et al. Evidence that inducible C4-type photosynthesis is a chloroplastic CO2-concentrating mechanism in Hydrilla, a submersed monocot. Plant Cell and Environment 20, 211–220 (1997).
Sage, R. F. The evolution of C-4 photosynthesis. New Phytologist 161, 341–370 (2004).
Teeri, J. A. & Stowe, L. G. Climatic patterns and distribution of C4 grasses in North America. Oecologia 23, 1–12 (1976).
Tieszen, L. L. et al. The distribution of C3 and C4 grasses and carbon isotope discrimination along an altitudinal and moisture gradient in Kenya. Oecologia 37, 337–350 (1979).
Vicentini, A. et al. The age of the grasses and clusters of origins of C-4 photosynthesis. Global Change Biology 14, 2963–2977 (2008).
Winter, K. et al. Seasonal shift from C3 photosynthesis to Crassulacean acid metabolism in Mesembryanthemum crystallinum growing in its natural environment. Oecologia 34, 225–237 (1978).