« Prev Next »
Hard-boiled eggs. Just some eggs and water -I recommend a pinch of salt as well- and they'll come out great. Simple enough, right? Maybe it's a simple process for the cook, but the chemistry is anything but! Now that we're on the subject, take out a few eggs and we'll go through the cooking and chemistry together.
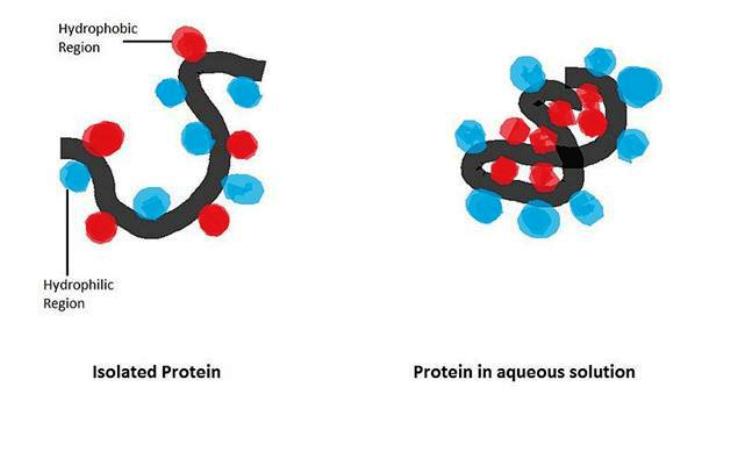
With just a raw egg in your hand, you're holding onto some really exciting chemistry. One of the main components of what you find underneath that shell is protein. The egg white is about 12 percent protein and the egg yolk itself is about 16 percent protein. This means that any alteration to the structure of these proteins will, in turn, significantly alter the structure of the entire egg. All proteins are made up of chains of amino acids, and those egg proteins are no exception. Right now, the proteins are in their normal, or native, state. When a protein is in its native state, the amino acids are folded in a very specific way that gives the protein its shape, and that shape, among other things, gives the protein its properties. A large part of what determines this native shape is how it interacts with water. Parts of the protein are hydrophilic, meaning they love water, and other parts are hydrophobic, meaning they will always avoid water. The water-loving parts will find themselves with the water on the outside of the molecule while the water-avoiding parts hide on the inside of the protein. This interaction, as well as many others, will keep the protein in its shape until you start boiling your eggs.
Now put your eggs in the pot, fill the pot with water, turn on the stove, and we can talk about denaturation. Denaturation is what happens when heat is applied to the eggs. Chemically, this is the process by which the chains of amino acids are changed from their original (or native) state. The heat coming from your stove denatures the protein by disrupting some of its bonds that held the molecule into shape. In the case of hard-boiled eggs, the proteins clump together and solidify, causing the egg white and yolk to harden.
With some more chemistry, we can now learn the science behind some common techniques used in preparation of hard-boiled eggs that are going to help us out. One common problem associated with cooking hard-boiled eggs is that their shells tend to crack, leaking out egg white. An easy fix is to add table salt or vinegar. Salt and acids (like vinegar) can also denature proteins in the same way heat does. Adding these substances speeds up the process by which the egg whites solidify and stops the seepage. In fact, some recipes call for eggs to be hard-boiled in vinegar and salt water to prevent the leaks from ever occurring. A second problem is associated with hard-boiled eggs is a green discoloration on the egg yolk. This is caused by a reaction between iron ions (Fe2+) in the yolk and hydrogen sulfide gas from the egg white, which is formed when hydrogen and sulfide react after being heated. The reaction produces a green compound, iron sulfide. Even though the green might look weird, the eggs are quite safe to eat. However, more advanced cooks may want to douse the eggs in cold water after cooking, which should mitigate the formation of the iron sulfide.
In case you forget whether you've actually hard-boiled some eggs (blame morning haze), here's a cool way to distinguish between hard-boiled eggs and raw eggs: spin them. A hard-boiled egg will spin far better than a raw one because the yolk and egg white have about the same consistency. In a raw egg, the yolk will spin at a different rate than the rest of the egg, causing it to be off balance and spin irregularly. I hope you enjoyed learning about and eating hard-boiled eggs as much as I did!
References:
Chang, R. Chemistry. New York: McGraw-Hill, 2007.
MSU Science Theater. "Hard-boiled Eggs." 1991.
TLC Cooking. "Why Do Eggs Turn Hard When You Boil Them?" 14 June 2011.
Novak, C. "A Brighter Idea: Eggs!" Virginia Tech
Image credit: Treshphrd (via Wikipedia)





















This post seems to draw on some of the information posted in other places on the web. The cool off step controls overcooking as is talked about in many websites. Overcooking is what causes the green yolk.