Abstract
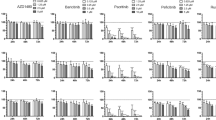
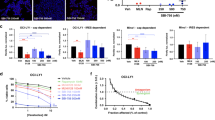
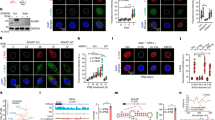
Primary effusion lymphoma (PEL) is an aggressive type of non-Hodgkin lymphoma localized predominantly in body cavities. Kaposi’s sarcoma-associated herpes virus (KSHV) is the causative agent of PEL. PEL is an incurable malignancy and has extremely poor prognosis when treated with conventional chemotherapy. Immunomodulatory drugs (IMiDs) lenalidomide and pomalidomide are Food and Drug Administration-approved drugs for the treatment of various ailments. IMiDs display pronounced antiproliferative effect against majority of PEL cell lines within their clinically achievable concentrations, by arresting cells at G0/G1 phase of cell cycle and without any induction of KSHV lytic cycle reactivation. Although microarray examination of PEL cells treated with lenalidomide revealed activation of interferon (IFN) signaling, blocking the IFN pathway did not block the anti-PEL activity of IMiDs. The anti-PEL effects of IMiDs involved cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1, but not IKZF3. Small hairpin RNA-mediated knockdown of MYC enhanced the cytotoxicity of IMiDs. Bromodomain (BRD) and extra-terminal domain (BET) proteins are epigenetic readers, which perform a vital role in chromatin remodeling and transcriptional regulation. BRD4, a widely expressed transcriptional coactivator, belongs to the BET family of proteins, which has been shown to co-occupy the super enhancers associated with MYC. Specific BRD4 inhibitors were developed, which suppress MYC transcriptionally. Lenalidomide displayed synergistic cytotoxicity with several structurally distinct BRD4 inhibitors (JQ-1, IBET151 and PFI-1). Furthermore, combined administration of lenalidomide and BRD4 inhibitor JQ-1 significantly increased the survival of PEL bearing NOD–SCID mice in an orthotopic xenograft model as compared with either agent alone. These results provide compelling evidence for clinical testing of IMiDs alone and in combination with BRD4 inhibitors for PEL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Patel S, Xiao P . Primary effusion lymphoma. Arch Pathol Lab Med 2013; 137: 1152–1154.
Cesarman E, Chang Y, Moore PS, Said JW, Knowles DM . Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 1995; 332: 1186–1191.
Kobayashi Y, Kamitsuji Y, Kuroda J, Tsunoda S, Uoshima N, Kimura S et al. Comparison of human herpes virus 8 related primary effusion lymphoma with human herpes virus 8 unrelated primary effusion lymphoma-like lymphoma on the basis of HIV: report of 2 cases and review of 212 cases in the literature. Acta Haematol 2007; 117: 132–144.
Shortt J, Hsu AK, Johnstone RW . Thalidomide-analogue biology: immunological, molecular and epigenetic targets in cancer therapy. Oncogene 2013; 32: 4191–4202.
Zhu YX, Kortuem KM, Stewart AK . Molecular mechanism of action of immune-modulatory drugs thalidomide, lenalidomide and pomalidomide in multiple myeloma. Leuk Lymphoma 2013; 54: 683–687.
Singhal S, Mehta J, Desikan R, Ayers D, Roberson P, Eddlemon P et al. Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med 1999; 341: 1565–1571.
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y et al. Identification of a primary target of thalidomide teratogenicity. Science 2010; 327: 1345–1350.
Zhu YX, Braggio E, Shi CX, Bruins LA, Schmidt JE, Van Wier S et al. Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood 2011; 118: 4771–4779.
Lopez-Girona A, Mendy D, Ito T, Miller K, Gandhi AK, Kang J et al. Cereblon is a direct protein target for immunomodulatory and antiproliferative activities of lenalidomide and pomalidomide. Leukemia 2012; 26: 2326–2335.
Lu G, Middleton RE, Sun H, Naniong M, Ott CJ, Mitsiades CS et al. The myeloma drug lenalidomide promotes the cereblon-dependent destruction of Ikaros proteins. Science 2014; 343: 305–309.
Kronke J, Udeshi ND, Narla A, Grauman P, Hurst SN, McConkey M et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science 2014; 343: 301–305.
Filippakopoulos P, Knapp S . Targeting bromodomains: epigenetic readers of lysine acetylation. Nat Rev Drug Discov 2014; 13: 337–356.
Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O et al. Selective inhibition of BET bromodomains. Nature 2010; 468: 1067–1073.
Dawson MA, Prinjha RK, Dittmann A, Giotopoulos G, Bantscheff M, Chan WI et al. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 2011; 478: 529–533.
Picaud S, Da Costa D, Thanasopoulou A, Filippakopoulos P, Fish PV, Philpott M et al. PFI-1, a highly selective protein interaction inhibitor, targeting BET Bromodomains. Cancer Res 2013; 73: 3336–3346.
Delmore JE, Issa GC, Lemieux ME, Rahl PB, Shi J, Jacobs HM et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 2011; 146: 904–917.
Mertz JA, Conery AR, Bryant BM, Sandy P, Balasubramanian S, Mele DA et al. Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc Natl Acad Sci USA 2011; 108: 16669–16674.
Tolani B, Gopalakrishnan R, Punj V, Matta H, Chaudhary PM . Targeting Myc in KSHV-associated primary effusion lymphoma with BET bromodomain inhibitors. Oncogene 2014; 33: 2928–2937.
Chen N, Lau H, Kong L, Kumar G, Zeldis JB, Knight R et al. Pharmacokinetics of lenalidomide in subjects with various degrees of renal impairment and in subjects on hemodialysis. J Clin Pharmacol 2007; 47: 1466–1475.
Kasserra C, Assaf M, Hoffmann M, Li Y, Liu L, Wang X et al. Pomalidomide: evaluation of cytochrome P450 and transporter-mediated drug-drug interaction potential in vitro and in healthy subjects. J Clin Pharmacol 2015; 55: 168–178.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Sun R, Lin SF, Gradoville L, Yuan Y, Zhu F, Miller G . A viral gene that activates lytic cycle expression of Kaposi’s sarcoma-associated herpesvirus. Proc Natl Acad Sci USA 1998; 95: 10866–10871.
Zhao J, Punj V, Matta H, Mazzacurati L, Schamus S, Yang Y et al. K13 blocks KSHV lytic replication and deregulates vIL6 and hIL6 expression: a model of lytic replication induced clonal selection in viral oncogenesis. PLoS One 2007; 2: e1067.
Shaffer AL, Emre NC, Lamy L, Ngo VN, Wright G, Xiao W et al. IRF4 addiction in multiple myeloma. Nature 2008; 454: 226–231.
Yang Y, Shaffer 3rd AL, Emre NC, Ceribelli M, Zhang M, Wright G et al. Exploiting synthetic lethality for the therapy of ABC diffuse large B cell lymphoma. Cancer Cell 2012; 21: 723–737.
Lopez-Girona A, Heintel D, Zhang LH, Mendy D, Gaidarova S, Brady H et al. Lenalidomide downregulates the cell survival factor, interferon regulatory factor-4, providing a potential mechanistic link for predicting response. Br J Haematol 2011; 154: 325–336.
Liu L, Eby MT, Rathore N, Sinha SK, Kumar A, Chaudhary PM . The human herpes virus 8-encoded viral FLICE inhibitory protein physically associates with and persistently activates the Ikappa B kinase complex. J Biol Chem 2002; 277: 13745–13751.
Matta H, Chaudhary PM . Activation of alternative NF-kappa B pathway by human herpes virus 8-encoded Fas-associated death domain-like IL-1 beta-converting enzyme inhibitory protein (vFLIP). Proc Natl Acad Sci USA 2004; 101: 9399–9404.
Keller SA, Schattner EJ, Cesarman E . Inhibition of NF-kappaB induces apoptosis of KSHV-infected primary effusion lymphoma cells. Blood 2000; 96: 2537–2542.
Shaffer AL, Emre NC, Romesser PB, Staudt LM . IRF4: Immunity. Malignancy! Therapy? Clin Cancer Res 2009; 15: 2954–2961.
Libermann TA, Baltimore D . Activation of interleukin-6 gene expression through the NF-kappa B transcription factor. Mol Cell Biol 1990; 10: 2327–2334.
John LB, Ward AC . The Ikaros gene family: transcriptional regulators of hematopoiesis and immunity. Mol Immunol 2011; 48: 1272–1278.
Chou TC, Talalay P . Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 1984; 22: 27–55.
Qin Z, Dai L, Trillo-Tinoco J, Senkal C, Wang W, Reske T et al. Targeting sphingosine kinase induces apoptosis and tumor regression for KSHV-associated primary effusion lymphoma. Mol Cancer Ther 2014; 13: 154–164.
Dimopoulos MA, Terpos E, Niesvizky R . How lenalidomide is changing the treatment of patients with multiple myeloma. Crit Rev Oncol Hematol 2013; 88 (Suppl 1): S23–S35.
Richardson PG, Mark TM, Lacy MQ . Pomalidomide: new immunomodulatory agent with potent antiproliferative effects. Crit Rev Oncol Hematol 2013; 88 (Suppl 1): S36–S44.
Klein U, Gloghini A, Gaidano G, Chadburn A, Cesarman E, Dalla-Favera R et al. Gene expression profile analysis of AIDS-related primary effusion lymphoma (PEL) suggests a plasmablastic derivation and identifies PEL-specific transcripts. Blood 2003; 101: 4115–4121.
Zhang LH, Kosek J, Wang M, Heise C, Schafer PH, Chopra R . Lenalidomide efficacy in activated B-cell-like subtype diffuse large B-cell lymphoma is dependent upon IRF4 and cereblon expression. Br J Haematol 2013; 160: 487–502.
Davis RE, Ngo VN, Lenz G, Tolar P, Young RM, Romesser PB et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature 2010; 463: 88–92.
Ngo VN, Young RM, Schmitz R, Jhavar S, Xiao W, Lim KH et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011; 470: 115–119.
Lenardo MJ, Fan CM, Maniatis T, Baltimore D . The involvement of NF-kappa B in beta-interferon gene regulation reveals its role as widely inducible mediator of signal transduction. Cell 1989; 57: 287–294.
Guasparri I, Keller SA, Cesarman E . KSHV vFLIP is essential for the survival of infected lymphoma cells. J Exp Med 2004; 199: 993–1003.
Nador RG, Cesarman E, Chadburn A, Dawson DB, Ansari MQ, Sald J et al. Primary effusion lymphoma: a distinct clinicopathologic entity associated with the Kaposi's sarcoma-associated herpes virus. Blood 1996; 88: 645–656.
Li X, Chen S, Feng J, Deng H, Sun R . Myc is required for the maintenance of Kaposi’s sarcoma-associated herpesvirus latency. J Virol 2010; 84: 8945–8948.
Zhang H, Neely L, Lundgren K, Yang YC, Lough R, Timple N et al. BIIB021, a synthetic Hsp90 inhibitor, has broad application against tumors with acquired multidrug resistance. Int J Cancer 2010; 126: 1226–1234.
Antar A, El Hajj H, Jabbour M, Khalifeh I, El-Merhi F, Mahfouz R et al. Primary effusion lymphoma in an elderly patient effectively treated by lenalidomide: case report and review of literature. Blood Cancer J 2014; 4: e190.
Gopalakrishnan R, Matta H, Chaudhary PM . A purine scaffold HSP90 inhibitor BIIB021 has selective activity against KSHV-associated primary effusion lymphoma and blocks vFLIP K13-induced NF-kappaB. Clin Cancer Res 2013; 19: 5016–5026.
Tolani B, Matta H, Gopalakrishnan R, Punj V, Chaudhary PM . NEMO is essential for Kaposi's sarcoma-associated herpesvirus-encoded vFLIP K13-induced gene expression and protection against death receptor-induced cell death, and its N-terminal 251 residues are sufficient for this process. J Virol 2014; 88: 6345–6354.
Yang Y, Groshong JS, Matta H, Gopalakrishnan R, Yi H, Chaudhary PM . Constitutive NF-kappaB activation confers interleukin 6 (IL6) independence and resistance to dexamethasone and janus kinase inhibitor INCB018424 in murine plasmacytoma cells. J Biol Chem 2011; 286: 27988–27997.
Matta H, Gopalakrishnan R, Graham C, Tolani B, Khanna A, Yi H et al. Kaposi’s sarcoma associated herpesvirus encoded viral FLICE inhibitory protein K13 activates NF-kappaB pathway independent of TRAF6, TAK1 and LUBAC. PLoS One 2012; 7: e36601.
Matta H, Gopalakrishnan R, Punj V, Yi H, Suo Y, Chaudhary PM . A20 is induced by Kaposi sarcoma-associated herpesvirus-encoded viral FLICE inhibitory protein (vFLIP) K13 and blocks K13-induced nuclear factor-kappaB in a negative feedback manner. J Biol Chem 2011; 286: 21555–21564.
Arvanitakis L, Mesri EA, Nador RG, Said JW, Asch AS, Knowles DM et al. Establishment and characterization of a primary effusion (body cavity- based) lymphoma cell line (BC-3) harboring kaposi's sarcoma-associated herpesvirus (KSHV/HHV-8) in the absence of Epstein-Barr virus. Blood 1996; 88: 2648–2654.
Renne R, Zhong W, Herndier B, McGrath M, Abbey N, Kedes D et al. Lytic growth of Kaposi’s sarcoma-associated herpesvirus (human herpesvirus 8) in culture. Nat Med 1996; 2: 342–346.
Cannon JS, Ciufo D, Hawkins AL, Griffin CA, Borowitz MJ, Hayward GS et al. A new primary effusion lymphoma-derived cell line yields a highly infectious Kaposi’s sarcoma herpesvirus-containing supernatant. J Virol 2000; 74: 10187–10193.
Jones D, Ballestas ME, Kaye KM, Gulizia JM, Winters GL, Fletcher J et al. Primary-effusion lymphoma and Kaposi’s sarcoma in a cardiac-transplant recipient. N Engl J Med 1998; 339: 444–449.
Sarosiek KA, Cavallin LE, Bhatt S, Toomey NL, Natkunam Y, Blasini W et al. Efficacy of bortezomib in a direct xenograft model of primary effusion lymphoma. Proc Natl Acad Sci USA 2010; 107: 13069–13074.
Bhatt S, Ashlock BM, Natkunam Y, Sujoy V, Chapman JR, Ramos JC et al. CD30 targeting with brentuximab vedotin: a novel therapeutic approach to primary effusion lymphoma. Blood 2013; 122: 1233–1242.
Cesarman E, Moore PS, Rao PH, Inghirami G, Knowles DM, Chang Y . In vitro establishment and characterization of two acquired immunodeficiency syndrome-related lymphoma cell lines (BC-1 and BC-2) containing Kaposi’s sarcoma-associated herpesvirus-like (KSHV) DNA sequences. Blood 1995; 86: 2708–2714.
Gao SJ, Kingsley L, Li M, Zheng W, Parravicini C, Ziegler J et al. KSHV antibodies among Americans, Italians and Ugandans with and without Kaposi’s sarcoma. Nat Med 1996; 2: 925–928.
Lee BS, Connole M, Tang Z, Harris NL, Jung JU . Structural analysis of the Kaposi’s sarcoma-associated herpesvirus K1 protein. J Virol 2003; 77: 8072–8086.
Acknowledgements
We thank the following investigators for their generous gift of cell lines: Dr Jae Jung (BC-3, BCBL-1, JSC-1, BC-1, BCP-1, VG-1 and APK-1); Drs Izidore Lossos and Juan Ramos (UMPEL-1 and UMPEL-3); Dr Art Shaffer (TMD8, U-2932, HBL-1, OCI-Ly7, OCI-Ly8 and OCI-Ly19); Dr Alan Epstein (DG-75); Randall Rossi (SUDHL-4, SUDHL-6, Granta, Toledo, KG-1 and MV-4-11); Dr Markus Mapara (L428, L540, L1236 and KM-H2); Dr Irene Ghobrial (BCWM.1 and WMCL-1); and Dr Alan Lichenstein (MM.1S and RPMI8226). We are grateful to Dr William Kaelin Jr for providing the shRNA constructs (CRBN, IKZF1-1 and IKZF1-2), Dr James Bradner for his generous contribution of (+) and (−) JQ-1, Dr Peter Howley for BRD4 antibody and Dr Gary Hayward for KSHV replication and transcription activator antibody. This work was supported by grants from the National Institutes of Health (CA139119, DE019811, SC CTSI UL1TR000130 and P30CA014089) and Stop Cancer Foundation. Flow Cytometry was analyzed in the USC Flow Cytometry Core Facility that is supported in part by the National Cancer Institute Cancer Center Shared Grant award P30CA014089 and the USC Provost Office Dean’s Development Funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
RG and PMC are inventors on a patent application (No. 62/031,053) filed to US patent office pertaining to the compositions and methods for treating primary effusion lymphoma. All other authors declare no conflict of interest.
Additional information
Accession codes
Microarray gene expression data has been deposited under accession number GSE60618 at the website Gene Expression Omnibus (http://www.ncbi.nlm.nih.gov/geo/).
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Gopalakrishnan, R., Matta, H., Tolani, B. et al. Immunomodulatory drugs target IKZF1-IRF4-MYC axis in primary effusion lymphoma in a cereblon-dependent manner and display synergistic cytotoxicity with BRD4 inhibitors. Oncogene 35, 1797–1810 (2016). https://doi.org/10.1038/onc.2015.245
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.245
This article is cited by
-
A non-randomized risk-adjusted comparison of lenalidomide + R-CHOP versus R-CHOP for MYC-rearranged DLBCL patients
Blood Cancer Journal (2023)
-
IRF4 drives clonal evolution and lineage choice in a zebrafish model of T-cell lymphoma
Nature Communications (2022)
-
Key regulators of sensitivity to immunomodulatory drugs in cancer treatment
Biomarker Research (2021)
-
KSHV transactivator-derived small peptide traps coactivators to attenuate MYC and inhibits leukemia and lymphoma cell growth
Communications Biology (2021)
-
A phase 1b dose-escalation/expansion study of BET inhibitor RO6870810 in patients with advanced multiple myeloma
Blood Cancer Journal (2021)