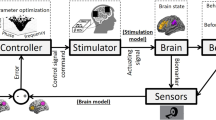
Key Points
-
Functional MRI (fMRI) has fundamentally changed the way that we can question brain systems because we can see them in action, bringing systems neuroscience to life. fMRI lets neuroscientists visualize the activity of neural networks that underlie human behaviour and development.
-
fMRI is an indirect measure of neural activity that can be used to evaluate disease states as well as drug function in awake humans and animals.
-
Current problems with the translation of animal behaviour (assays) to the human condition might be overcome by using a circuit-based approach to define objective changes in neural systems.
-
fMRI is able to dissect complex brain function in terms of temporally coordinated activation of related neurocircuits rather than dissociated events associated with disparate nuclei. fMRI has given new dimensions to many areas of complex brain function such as pain, sensory systems, psychiatric disorders and their co-morbidity with other central nervous system (CNS) conditions, cognition, language consciousness and neural mechanisms that underlie the developmental plasticity of the brain or its recovery of function after trauma.
-
fMRI can report on the functional neuroanatomy of the brain and, in combination with targeted therapeutic agents, provide novel insights into CNS neuropharmacology that could inform the drug development process from preclinical stages to clinical evaluation.
-
Whole-system effects allow for the evaluation of drug effects or disease states over time, conferring a better understanding of the long-term effects of drugs by objective measures of these circuits.
Abstract
Drug development today needs to balance agility, speed and risk in defining the probability of success for molecules, mechanisms and therapeutic concepts. New techniques in functional magnetic resonance imaging (fMRI) promise to be part of a sequence that could transform drug development for disorders of the central nervous system (CNS) by examining brain systems and their functional activation dynamically. The brain is complex and multiple transmitters and intersecting brain circuits are implicated in many CNS disorders. CNS therapeutics are designed against specific CNS targets, many of which are unprecedented. The challenge is to reveal the functional consequences of these interactions to assess therapeutic potential. fMRI can help optimize CNS drug discovery by providing a key metric that can increase confidence in early decision-making, thereby improving success rates and reducing risk, development times and costs of drug development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Cohen, C. M. A path to improved pharmaceutical productivity. Nature Rev. Drug Discov. 2, 751?753 (2003). A critical article on the evaluation of performance metrics in the pharmaceutical industry.
Kola, I. & Landis, J. Can the pharmaceutical industry reduce attrition rates? Nature Rev. Drug Discov. 3, 711?715 (2004). An evaluation of fiscal issues facing R&D and how to improve this domain in the pharmaceutical industry.
Frank, R. & Hargreaves, R. Clinical biomarkers in drug discovery and development. Nature Rev. Drug Discov. 2, 566?580 (2003). Biomarkers will become highly valued processes to characterize drugs and disease states.
Pritchard, J. F. et al. Making better drugs: Decision gates in non-clinical drug development. Nature Rev. Drug Discov. 2, 542?553 (2003).
Bleicher, K. H., Bohm, H. J., Muller, K. & Alanine, A. I. Hit and lead generation: beyond high-throughput screening. Nature Rev. Drug Discov. 2, 369?378 (2003).
Bild, A. H. et al. Oncogenic pathway signatures in human cancers as a guide to targeted therapies. Nature 439, 353?357 (2006).
DaSilva, A. F. et al. Somatotopic activation in the human trigeminal pain pathway. J. Neurosci. 22, 8183?8192 (2002). One of the first papers to describe pain activation in the spinal cord (brainstem) in humans. Imaging a complete pathway provides an approach to evaluate surrogate models and disease states affecting the trigeminal system.
Hornung, J. P. The human raphe nuclei and the serotonergic system. J. Chem. Neuroanat. 26, 331?343 (2003).
Grzanna, R. & Fritschy, J. M. Efferent projections of different subpopulations of central noradrenaline neurons. Prog. Brain. Res. 88, 89?101 (1991).
Everitt, B. J. & Robbins, T. W. Central cholinergic systems and cognition. Annu. Rev. Psychol. 48, 649?684 (1997).
Johansson, B. B. Brain plasticity in health and disease. Keio. J. Med. 53, 231?246 (2004).
Raineteau, O. & Schwab, M. E. Plasticity of motor systems after incomplete spinal cord injury. Nature Rev. Neurosci. 2, 263?273 (2001).
Chen, R., Cohen, L. G. & Hallett, M. Nervous system reorganization following injury. Neuroscience 111, 761?773 (2002).
Hatten, M. E. & Heintz, N. Large-scale genomic approaches to brain development and circuitry. Annu. Rev. Neurosci. 28, 89?108 (2005).
Seitz, R. J., Kleiser, R. & Butefisch, C. M. Reorganization of cerebral circuits in human brain lesion. Acta Neurochir. Suppl. 93, 65?70 (2005).
Eden, G. F. et al. Neural changes following remediation in adult developmental dyslexia. Neuron 44, 411?422 (2004).
Temple, E. et al. Neural deficits in children with dyslexia ameliorated by behavioral remediation: evidence from functional MRI. Proc. Natl Acad. Sci. USA 100, 2860?2865 (2003).
Stoeckel, M. C., Jorgens, S., Witte, O. W. & Seitz, R. J. Reduced somatosensory hand representation in thalidomide-induced dysmelia as revealed by fMRI. Eur. J. Neurosci. 21, 556?562 (2005).
Baker, C. I., Peli, E., Knouf, N. & Kanwisher, N. G. Reorganization of visual processing in macular degeneration. J. Neurosci. 25, 614?618 (2005).
Borsook, D. et al. Acute plasticity in the human somatosensory cortex following amputation. Neuroreport 9, 1013?1017 (1998).
Apkarian, A. V. et al. Chronic back pain is associated with decreased prefrontal and thalamic gray matter density. J. Neurosci. 24, 10410?10415 (2004). A revolutionary paper and quantum leap in understanding that chronic pain is a degenerative disease affecting the CNS.
Tracey, I. Prospects for human pharmacological functional magnetic resonance imaging (phMRI). J. Clin. Pharmacol. (Suppl.), 21S?28S (2001). A good review of the potential of fMRI in pharmacology.
Borsook, D., Ploghaus, A. & Becerra, L. Utilizing brain imaging for analgesic drug development. Curr. Opin. Investig. Drugs. 3, 1342?1347 (2002).
Borsook, D. & Becerra, L. Pain imaging: future applications to integrative clinical and basic neurobiology. Adv. Drug. Deliv. Rev. 55, 967?986 (2003).
Fava, M., Mallinckrodt, C. H., Detke, M. J., Watkin, J. G. & Wohlreich, M. M. The effect of duloxetine on painful physical symptoms in depressed patients: do improvements in these symptoms result in higher remission rates? J. Clin. Psychiatry 65, 521?530 (2004).
Ohayon, M. M. Specific characteristics of the pain/depression association in the general population. J. Clin. Psychiatry 65 (Suppl. 12), 5?9 (2004).
Klein, T., Magerl, W., Rolke, R. & Treede, R. D. Human surrogate models of neuropathic pain. Pain 115, 227?233 (2005).
Bennett, G. J. & Xie, Y. K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33, 87?107 (1988).
Decosterd, I. & Woolf, C. J. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87, 149?158 (2000).
Mantyh, P. W. A mechanism-based understanding of bone cancer pain. Novartis Found. Symp. 261, 194?214 (2004).
Blackburn-Munro, G. Pain-like behaviours in animals- how human are they? Trends Pharmacol. Sci. 25, 299?305 (2004).
Mogil, J. S. & Crager, S. E. What should we be measuring in behavioral studies of chronic pain in animals? Pain 112, 12?15 (2004).
Kachergus, J. et al. Identification of a novel LRRK2 mutation linked to autosomal dominant parkinsonism: evidence of a common founder across European populations. Am. J. Hum. Genet. 76, 672?680 (2005).
Grafton, S. T. Contributions of functional imaging to understanding parkinsonian symptoms. Curr. Opin. Neurobiol. 14, 715?719 (2004). A critical review on the use of imaging to evaluate specific circuits involved in this disease that clearly has applications to other CNS disease states.
DiMasi, J. A., Hansen, R. W. & Grabowski, H. G. The price of innovation: new estimates of drug development costs. J. Health Econ. 22, 151?185 (2003).
Rawlins, M. D. Cutting the cost of drug development? Nature Rev. Drug Discov. 3, 360?364 (2004).
Littman, B. H. & Williams, S. A. The ultimate model organism: progress in experimental medicine. Nature Rev. Drug Discov. 4, 631?638 (2005).
Leslie, R. A. & James, M. F. Pharmacological magnetic resonance imaging: a new application for functional MRI. Trends Pharmacol. Sci. 21, 314?318 (2000).
Braus, D. F., Brassen, S., Weimer, E. & Tost, H. [Functional magnetic resonance imaging of psychopharmacological brain effects: an update]. Fortschr. Neurol. Psychiatr. 71, 72?83 (2003).
Honey, G. & Bullmore, E. Human pharmacological MRI. Trends Pharmacol. Sci. 25, 366?374 (2004). An outstanding review of the state of the art of pharmacological fMRI.
Shah, Y. B. & Marsden, C. A. The application of functional magnetic resonance imaging to neuropharmacology. Curr. Opin. Pharmacol. 4, 517?521 (2004).
Vanduffel, W. et al. Visual motion processing investigated using contrast agent-enhanced fMRI in awake behaving monkeys. Neuron 32, 565?577 (2001).
Febo, M. et al. Imaging cocaine-induced changes in the mesocorticolimbic dopaminergic system of conscious rats. J. Neurosci. Methods 139, 167?176 (2004).
Brewer, A. A., Press, W. A., Logothetis, N. K. & Wandell, B. A. Visual areas in macaque cortex measured using functional magnetic resonance imaging. J. Neurosci. 22, 10416?10426 (2002).
Wilkinson, D. & Halligan, P. The relevance of behavioural measures for functional-imaging studies of cognition. Nature Rev. Neurosci. 5, 67?73 (2004).
Brunner, D., Nestler, E. & Leahy, E. In need of high-throughput behavioral systems. Drug Discov. Today 7, S107?S112 (2002).
Ren, K., Thomas, D. A. & Dubner, R. Nerve growth factor alleviates a painful peripheral neuropathy in rats. Brain Res. 699, 286?292 (1995).
Apfel, S. C. et al. Efficacy and safety of recombinant human nerve growth factor in patients with diabetic polyneuropathy: A randomized controlled trial. rhNGF Clinical Investigator Group. JAMA 284, 2215?2221 (2000).
Goldstein, D. J., Wang, O., Gitter, B. D. & Iyengar, S. Dose-response study of the analgesic effect of lanepitant in patients with painful diabetic neuropathy. Clin. Neuropharmacol. 24, 16?22 (2001).
Hill, R. NK1 (substance P) receptor antagonists--why are they not analgesic in humans? Trends Pharmacol. Sci. 21, 244?246 (2000).
Duffy, R. A. Potential therapeutic targets for neurokinin-1 receptor antagonists. Expert Opin. Emerg. Drugs 9, 9?21 (2004).
Keller, M. et al. Lack of efficacy of the substance P (neurokinin(1) receptor) antagonist aprepitant in the treatment of major depressive disorder. Biol. Psychiatry 59, 216?223 (2005).
Diemunsch, P. & Grelot, L. Potential of substance P antagonists as antiemetics. Drugs 60, 533?546 (2000).
Pendergrass, K. et al. Aprepitant: an oral NK1 antagonist for the prevention of nausea and vomiting induced by highly emetogenic chemotherapy. Drugs Today (Barc.) 40, 853?863 (2004).
Ashburn, T. T. & Thor, K. B. Drug repositioning: identifying and developing new uses for existing drugs. Nature Rev. Drug Discov. 3, 673?683 (2004).
Dimasi, J. A. & Paquette, C. The economics of follow-on drug research and development: trends in entry rates and the timing of development. Pharmacoeconomics 22, 1?14 (2004).
Borras, M. C. et al. fMRI measurement of CNS responses to naloxone infusion and subsequent mild noxious thermal stimuli in healthy volunteers. J. Neurophysiol. 91, 2723?2733 (2004). One of the first studies evaluating CNS effects of a drug. Interestingly, there were no behavioural effects associated with the drug.
Becerra, L., Harter, K., Gonalez, R. G. & Borsook, D. Morphine activates neural circuits involved in reward, sedation, and endogenous analgesia in drug-naive humans. Anesthesia And Analgesia (in the press).
Borsook, D. & Edwards, A. D. Antineuropathic effects of the antibiotic derivative spicamycin KRN5500. Pain Med. 5, 104?108 (2004). A paper that defines emotional circuitry in response to pain and has opened the door to understanding the potential role of pain in affecting circuitry involved in the placebo response and in functional illness.
Becerra, L., Breiter, H. C., Wise, R., Gonzalez, R. G. & Borsook, D. Reward circuitry activation by noxious thermal stimuli. Neuron 32, 927?946 (2001).
Gear, R. W., Aley, K. O. & Levine, J. D. Pain-induced analgesia mediated by mesolimbic reward circuits. J. Neurosci. 19, 7175?7181 (1999).
Perucca, E. An introduction to antiepileptic drugs. Epilepsia 46 (Suppl. 4), 31?37 (2005). This paper provides evidence that fMRI signal has a neural basis.
Becerra, L. et al. Dissecting drug efficacy for neuropathic pain in healthy subjects. Annu. Meeting Soc. Neurosci. Washington DC, A50.10 (2005).
Apkarian, A. V., Bushnell, M. C., Treede, R. D. & Zubieta, J. K. Human brain mechanisms of pain perception and regulation in health and disease. Eur. J. Pain 9, 463?484 (2005).
Ramaekers, J. G. Behavioural toxicity of medicinal drugs. Practical consequences, incidence, management and avoidance. Drug Saf. 18, 189?208 (1998).
Bazil, C. W. Effects of antiepileptic drugs on sleep structure: are all drugs equal? CNS Drugs 17, 719?728 (2003).
Hornby, P. J. Central neurocircuitry associated with emesis. Am. J. Med. 111 (Suppl. 8A), 106S?112S (2001).
Eisenach, J. C., Carpenter, R. & Curry, R. Analgesia from a peripherally active kappa-opioid receptor agonist in patients with chronic pancreatitis. Pain 101, 89?95 (2003).
DeHaven-Hudkins, D. L. & Dolle, R. E. Peripherally restricted opioid agonists as novel analgesic agents. Curr. Pharm. Des. 10, 743?757 (2004).
Dortch-Carnes, J. & Potter, D. E. Bremazocine: a kappa-opioid agonist with potent analgesic and other pharmacologic properties. CNS Drug Rev. 11, 195?212 (2005).
Flor, H. Remapping somatosensory cortex after injury. Adv. Neurol. 93, 195?204 (2003).
Muller, U. et al. Plasma level-dependent effects of methylphenidate on task-related functional magnetic resonance imaging signal changes. Psychopharmacology (Berl) 180, 624?633 (2005).
Honey, G. D. et al. Dopaminergic drug effects on physiological connectivity in a human cortico-striato-thalamic system. Brain 126, 1767?81 (2003). fMRI demonstrates functional connectivity of the human caudate nucleus using the pharmacological agent sulpiride.
Wise, R. G. et al. Combining fMRI with a pharmacokinetic model to determine which brain areas activated by painful stimulation are specifically modulated by remifentanil. Neuroimage 16, 999?1014 (2002).
Meier, C. R., Wilcock, K. & Jick, S. S. The risk of severe depression, psychosis or panic attacks with prophylactic antimalarials. Drug Saf. 27, 203?213 (2004).
Overbosch, D. et al. Atovaquone-proguanil versus mefloquine for malaria prophylaxis in nonimmune travelers: results from a randomized, double-blind study. Clin. Infect. Dis. 33, 1015?1021 (2001).
Heckers, S. The hippocampus in schizophrenia. Am. J. Psychiatry 161, 2138?2139 (2004).
Zubieta, J. K. et al. COMT val158met genotype affects mu-opioid neurotransmitter responses to a pain stressor. Science 299, 1240?1243 (2003). A paper that has defined what is yet to come: mapping phenotype (using brain imaging) with genotype/SNP mapping.
Momose, Y. et al. Association studies of multiple candidate genes for Parkinson's disease using single nucleotide polymorphisms. Ann. Neurol. 51, 133?136 (2002).
Diatchenko, L. et al. Genetic basis for individual variations in pain perception and the development of a chronic pain condition. Hum. Mol. Genet. 14, 135?143 (2005).
Becerra, L., Morris, S., Chizh, B. & Borsook, D. fMRI Activation of the trigeminal sensory pathway in trigeminal neuropathy. Annu. Meeting Soc. Neurosci. Washington DC, A53.16 (2005).
Logothetis, N. K., Pauls, J., Augath, M., Trinath, T. & Oeltermann, A. Neurophysiological investigation of the basis of the fMRI signal. Nature 412, 150?157 (2001).
Prathikanti, S. & Weinberger, D. R. Psychiatric genetics- the new era: genetic research and some clinical implications. Br. Med. Bull. 73?74, 107?122 (2005).
Winterer, G., Hariri, A. R., Goldman, D. & Weinberger, D. R. Neuroimaging and Human Genetics. Int Rev Neurobiol 67PB, 325?383 (2005).
Borsook, D., Burstein, R. & Becerra, L. Functional imaging of the human trigeminal system: opportunities for new insights into pain processing in health and disease. J. Neurobiol. 61, 107?125 (2004).
Borsook, D., DaSilva, A. F., Ploghaus, A. & Becerra, L. Specific and somatotopic functional magnetic resonance imaging activation in the trigeminal ganglion by brush and noxious heat. J. Neurosci. 23, 7897?7903 (2003).
Burstein, R. Deconstructing migraine headache into peripheral and central sensitization. Pain 89, 107?110 (2001).
Salmeron, B. J. & Stein, E. A. Pharmacological applications of magnetic resonance imaging. Psychopharmacol. Bull. 36, 102?129 (2002).
De Luca, M., Beckmann, C. F., De Stefano, N., Matthews, P. M. & Smith, S. M. fMRI resting state networks define distinct modes of long-distance interactions in the human brain. Neuroimage 29, 1359?1367 (2005).
Foss, J. M., Apkarian, A. V. & Chialvo, D. R. Dynamics of pain: fractal dimension of temporal variability of spontaneous pain differentiates between pain States. J. Neurophysiol. 95, 730?736 (2006).
Salvador, R., Suckling, J., Schwarzbauer, C. & Bullmore, E. Undirected graphs of frequency-dependent functional connectivity in whole brain networks. Philos. Trans. R. Soc. B Biol. Sci. 360, 937?946 (2005).
deCharms, R. C. et al. Control over brain activation and pain learned by using real-time functional MRI. Proc. Natl Acad. Sci. USA 102, 18626?18631 (2005).
Smith, S. M. et al. Variability in fMRI: a re-examination of inter-session differences. Hum. Brain Mapp. 24, 248?257 (2005).
Casey, B. J. et al. Reproducibility of fMRI results across four institutions using a spatial working memory task. Neuroimage 8, 249?261 (1998).
Marshall, I. et al. Repeatability of motor and working-memory tasks in healthy older volunteers: assessment at functional MR imaging. Radiology 233, 868?877 (2004).
Wei, X. et al. Functional MRI of auditory verbal working memory: long-term reproducibility analysis. Neuroimage 21, 1000?1008 (2004).
Yoo, S. S., Wei, X., Dickey, C. C., Guttmann, C. R. & Panych, L. P. Long-term reproducibility analysis of fMRI using hand motor task. Int J. Neurosci. 115, 55?77 (2005).
Friston, K. J., Holmes, A. P., Price, C. J., Buchel, C. & Worsley, K. J. Multisubject fMRI studies and conjunction analyses. Neuroimage 10, 385?396 (1999).
Brett, M., Johnsrude, I. S. & Owen, A. M. The problem of functional localization in the human brain. Nature Rev. Neurosci. 3, 243?249 (2002).
Foland, L. C., Thomason, M. E. & Glover, G. H. Calibrating functional MRI data across subjects and scan sites. Annu. Meeting Soc. Neurosci. San Diego, A693.9 (2004).
Borsook, D. & Becerra, L. Functional imaging of pain and analgesia ? a valid diagnostic tool? Pain 117, 247?250 (2005).
Davis, K. D., Wood, M. L., Crawley, A. P. & Mikulis, D. J. fMRI of human somatosensory and cingulate cortex during painful electrical nerve stimulation. Neuroreport 7, 321?325 (1995).
Becerra, L. R. et al. Human brain activation under controlled thermal stimulation and habituation to noxious heat: an fMRI study. Magn. Reson. Med. 41, 1044?1057 (1999).
Tuor, U. I., McKenzie, E. & Tomanek, B. Functional magnetic resonance imaging of tonic pain and vasopressor effects in rats. Magn. Reson. Imaging 20, 707?712 (2002).
Malisza, K. L. & Docherty, J. C. Capsaicin as a source for painful stimulation in functional MRI. J. Magn. Reson. Imaging 14, 341?347 (2001).
Chang, C. & Shyu, B. C. A fMRI study of brain activations during non-noxious and noxious electrical stimulation of the sciatic nerve of rats. Brain Res. 897, 71?81 (2001).
Tuor, U. I. et al. Functional magnetic resonance imaging in rats subjected to intense electrical and noxious chemical stimulation of the forepaw. Pain 87, 315?324 (2000).
Lebel, A. et al. fMRI of mechanical allodynia in children with complex regional (leg) pain syndrome (CRPS). Soc. Neurosci. Conf. Washington DC, A53.17 (2005). An important model that demonstrates the use of fMRI in assessing drug effects.
Peyron, R. et al. An fMRI study of cortical representation of mechanical allodynia in patients with neuropathic pain. Neurology 63, 1838?1846 (2004).
Maihofner, C. & Handwerker, H. O. Differential coding of hyperalgesia in the human brain: a functional MRI study. Neuroimage 28, 996?1006 (2005).
Baron, R., Baron, Y., Disbrow, E. & Roberts, T. P. Brain processing of capsaicin-induced secondary hyperalgesia: a functional MRI study. Neurology 53, 548?557 (1999).
Zambreanu, L., Wise, R. G., Brooks, J. C., Iannetti, G. D. & Tracey, I. A role for the brainstem in central sensitisation in humans. Evidence from functional magnetic resonance imaging. Pain 114, 397?407 (2005).
Giesecke, T. et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 50, 613?623 (2004).
Gracely, R. H. et al. Pain catastrophizing and neural responses to pain among persons with fibromyalgia. Brain 127, 835?843 (2004). This study provided novel insights into CNS processing in a disease about which we have had little understanding.
Cook, D. B. et al. Functional imaging of pain in patients with primary fibromyalgia. J. Rheumatol. 31, 364?378 (2004).
Alkire, M. T., White, N. S., Hsieh, R. & Haier, R. J. Dissociable brain activation responses to 5-Hz electrical pain stimulation: a high-field functional magnetic resonance imaging study. Anesthesiology 100, 939?946 (2004).
DaSilva, A. F. et al. Somatotopic activation in the human trigeminal pain pathway. J. Neurosci. 22, 8183?8192 (2002).
Brooks, J. C., Zambreanu, L., Godinez, A., Craig, A. D. & Tracey, I. Somatotopic organisation of the human insula to painful heat studied with high resolution functional imaging. Neuroimage 27, 201?209 (2005).
Moulton, E. A., Keaser, M. L., Gullapalli, R. P. & Greenspan, J. D. Regional intensive and temporal patterns of functional MRI activation distinguishing noxious and innocuous contact heat. J. Neurophysiol. 93, 2183?2193 (2005).
Bornhovd, K. et al. Painful stimuli evoke different stimulus-response functions in the amygdala, prefrontal, insula and somatosensory cortex: a single-trial fMRI study. Brain 125, 1326?1336 (2002).
Apkarian, A. V., Darbar, A., Krauss, B. R., Gelnar, P. A. & Szeverenyi, N. M. Differentiating cortical areas related to pain perception from stimulus identification: temporal analysis of fMRI activity. J. Neurophysiol. 81, 2956?2963 (1999).
Davis, K. D., Kwan, C. L., Crawley, A. P. & Mikulis, D. J. Functional MRI study of thalamic and cortical activations evoked by cutaneous heat, cold, and tactile stimuli. J. Neurophysiol. 80, 1533?1546 (1998).
Tracey, I. et al. Noxious hot and cold stimulation produce common patterns of brain activation in humans: a functional magnetic resonance imaging study. Neurosci. Lett. 288, 159?162 (2000).
Strigo, I. A., Albanese, M. C., Bushnell, M. C. & Duncan, G. H. Visceral and cutaneous pain representation in parasylvian cortex. Neurosci. Lett. 384, 54?59 (2005).
Dunckley, P. et al. Cortical processing of visceral and somatic stimulation: differentiating pain intensity from unpleasantness. Neuroscience 133, 533?542 (2005).
Lu, C. L. et al. Neuronal correlates of gastric pain induced by fundus distension: a 3T-fMRI study. Neurogastroenterol. Motil. 16, 575?587 (2004).
Yaguez, L. et al. Brain response to visceral aversive conditioning: a functional magnetic resonance imaging study. Gastroenterology 128, 1819?1829 (2005).
Hobson, A. R. et al. Real-time imaging of human cortical activity evoked by painful esophageal stimulation. Gastroenterology 128, 610?619 (2005).
Verne, G. N. et al. Central representation of visceral and cutaneous hypersensitivity in the irritable bowel syndrome. Pain 103, 99?110 (2003).
Bonaz, B. et al. Central processing of rectal pain in patients with irritable bowel syndrome: an fMRI study. Am. J. Gastroenterol. 97, 654?661 (2002).
Chen, Y. C. et al. Detection of dopaminergic neurotransmitter activity using pharmacologic MRI: correlation with PET, microdialysis, and behavioral data. Magn. Reson. Med. 38, 389?398 (1997).
Chen, Y. C., Choi, J. K., Andersen, S. L., Rosen, B. R. & Jenkins, B. G. Mapping dopamine D2/D3 receptor function using pharmacological magnetic resonance imaging. Psychopharmacology (Berl) 180, 705?715 (2005).
Dixon, A. L. et al. Dopamine antagonist modulation of amphetamine response as detected using pharmacological MRI. Neuropharmacology 48, 236?245 (2005).
Hagino, H. et al. Effects of D2 dopamine receptor agonist and antagonist on brain activity in the rat assessed by functional magnetic resonance imaging. Brain Res. 813, 367?373 (1998).
Ireland, M. D. et al. Mapping the effects of the selective dopamine D2/D3 receptor agonist quinelorane using pharmacological magnetic resonance imaging. Neuroscience 133, 315?326 (2005).
Febo, M. et al. The neural consequences of repeated cocaine exposure revealed by functional MRI in awake rats. Neuropsychopharmacology 30, 936?943 (2005).
Schwarz, A. J. et al. Concurrent pharmacological MRI and in situ microdialysis of cocaine reveal a complex relationship between the central hemodynamic response and local dopamine concentration. Neuroimage 23, 296?304 (2004).
Marota, J. J. et al. Cocaine activation discriminates dopaminergic projections by temporal response: an fMRI study in Rat. Neuroimage 11, 13?23 (2000).
Reese, T. et al. Impaired functionality of reperfused brain tissue following short transient focal ischemia in rats. Magn. Reson. Imaging 20, 447?454 (2002).
Jones, N., O'Neill, M. J., Tricklebank, M., Libri, V. & Williams, S. C. Examining the neural targets of the AMPA receptor potentiator LY404187 in the rat brain using pharmacological magnetic resonance imaging. Psychopharmacology (Berl) 180, 743?751 (2005).
Gozzi, A. et al. Functional magnetic resonance mapping of intracerebroventricular infusion of a neuroactive peptide in the anaesthetised rat. J. Neurosci. Methods 142, 115?124 (2005).
Henderson, L. A. et al. Functional magnetic resonance signal changes in neural structures to baroreceptor reflex activation. J. Appl. Physiol. 96, 693?703 (2004).
Rausch, M., Gentsch, C., Enz, A., Baumann, D. & Rudin, M. A study paradigm allowing comparison of multiple high-resolution rCBV-maps for the examination of drug effects. NMR Biomed 18, 260?268 (2005).
Shoaib, M., Lowe, A. S. & Williams, S. C. Imaging localised dynamic changes in the nucleus accumbens following nicotine withdrawal in rats. Neuroimage 22, 847?854 (2004).
Xu, H. et al. Heroin-induced neuronal activation in rat brain assessed by functional MRI. Neuroreport 11, 1085?1092 (2000).
Lowe, A. S., Williams, S. C., Symms, M. R., Stolerman, I. P. & Shoaib, M. Functional magnetic resonance neuro-imaging of drug dependence: naloxone-precipitated morphine withdrawal. Neuroimage 17, 902?910 (2002).
Shah, Y. B., Prior, M. J., Dixon, A. L., Morris, P. G. & Marsden, C. A. Detection of cannabinoid agonist evoked increase in BOLD contrast in rats using functional magnetic resonance imaging. Neuropharmacology 46, 379?387 (2004).
Borras, M. C. et al. fMRI measurement of CNS responses to naloxone infusion and subsequent mild noxious thermal stimuli in healthy volunteers. J. Neurophysiol. 91, 2723?2733 (2004).
Vollm, B. A. et al. Methamphetamine activates reward circuitry in drug naive human subjects. Neuropsychopharmacology 29, 1715?1722 (2004).
Breiter, H. C. et al. Acute effects of cocaine on human brain activity and emotion. Neuron 19, 591?611 (1997). A pivotal study in drug addiction and reward.
Stein, E. A. et al. Nicotine-induced limbic cortical activation in the human brain: a functional MRI study. Am. J. Psychiatry 155, 1009?1015 (1998).
Logothetis, N. K., Pauls, J., Augath, M., Trinath, T. & Oeltermann, A. Neurophysiological investigation of the basis of the fMRI signal. Nature 412, 150?157 (2001).
Acknowledgements
We would like to thank K. Moldoff (www.galeriekirk.com) for kind permission to reproduce some of his graphics for this review.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
D.B. and L.B. have received unrestricted grant support from Merck Research Laboratories and a grant from the National Institutes of Neurological Diseases and Stroke. R.H. is a full-time employee of Merck & Co. Inc.
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
Glossary
- Functional magnetic resonance imaging
-
An MRI technique that uses metabolic-induced capillary blood flow changes caused by neuronal activity to produce images reflecting such activity. Activation can be correlated with brain structures as determined by these images.
- Molecular imaging
-
An imaging technique in which cellular/molecular processes have been tagged in such a way that they can be non-invasively imaged.
- Positron-emission tomography
-
(PET). A dual-photon nuclear imaging technique in which radioactive tracers are administered in non-pharmacologically active doses to subjects and images are created that reveal brain blood flow, glucose metabolism or fractional receptor binding by drugs.
- Single-photon-emission computed tomography
-
(SPECT). A nuclear imaging technique in which radioactive tracers generating single photons of a specific energy are administered to subjects to produce images. SPECT can give information about blood flow to tissues, molecular targets, and chemical reactions (metabolism) in the body.
- Neuroinformatics
-
A field that deals with data structure and software tools devoted to the analysis and integration of neuroscience.
- Gyrencephalic species
-
Mammalian species that have developed cerebrums in which gyri and sulci can be defined.
- Functional neuropathomics
-
Intended to define the underlying pathophysiology in neural conditions at a systems level.
- Functional neuromics
-
Defining the functional components of normal neural function at a systems level.
- Functional classifiers
-
Drugs can act on neural networks or systems in a particular fashion ? for example, produce sedation, euphoria or analgesia. Activation patterns that show specific regions of the brain known to be involved in a particular function allow for the segregation or functional classification of drug action.
Rights and permissions
About this article
Cite this article
Borsook, D., Becerra, L. & Hargreaves, R. A role for fMRI in optimizing CNS drug development. Nat Rev Drug Discov 5, 411–425 (2006). https://doi.org/10.1038/nrd2027
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd2027
This article is cited by
-
Neuroimaging in psychedelic drug development: past, present, and future
Molecular Psychiatry (2023)
-
Cannabidiol has a unique effect on global brain activity: a pharmacological, functional MRI study in awake mice
Journal of Translational Medicine (2021)
-
Lessons learned from using fMRI in the early clinical development of a mu-opioid receptor antagonist for disorders of compulsive consumption
Psychopharmacology (2021)
-
Bidirectional Associations among Nicotine and Tobacco Smoke, NeuroHIV, and Antiretroviral Therapy
Journal of Neuroimmune Pharmacology (2020)
-
Opioid receptors: drivers to addiction?
Nature Reviews Neuroscience (2018)