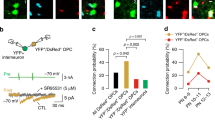
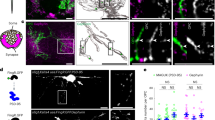
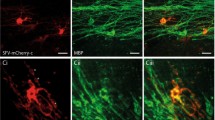
Abstract
A defining feature of glial cells has been their inability to generate action potentials. We show here that there are two distinct types of morphologically identical oligodendrocyte precursor glial cells (OPCs) in situ in rat CNS white matter. One type expresses voltage-gated sodium and potassium channels, generates action potentials when depolarized and senses its environment by receiving excitatory and inhibitory synaptic input from axons. The other type lacks action potentials and synaptic input. We found that when OPCs suffered glutamate-mediated damage, as occurs in cerebral palsy, stroke and spinal cord injury, the action potential–generating OPCs were preferentially damaged, as they expressed more glutamate receptors, and received increased spontaneous glutamatergic synaptic input in ischemia. These data challenge the idea that only neurons generate action potentials in the CNS and imply that the development of therapies for demyelinating disorders will require defining which OPC type can carry out remyelination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
21 May 2008
In the version of this article initially published, the units for the values reported for resistance in Figure 2i,j were incorrect. The correct unit should be gigaohms. In addition, the authors did not state that the cell death experiments in Figure 6e,f were carried out using a physiological extracellular magnesium concentration of 1 mM. This error has been corrected in the HTML and PDF versions of the article.
References
Nishiyama, A., Lin, X.-H., Giese, N., Heldin, C.-H. & Stallcup, W.B. Colocalization of NG2 proteoglycan and PDGF alpha receptor on O2A progenitor cells in the developing rat brain. J. Neurosci. Res. 43, 299–314 (1996).
Nishiyama, A., Watanabe, M., Yang, Z. & Bu, J. Identity, distribution and development of polydendrocytes: NG2-expressing glial cells. J. Neurocytol. 31, 437–455 (2002).
Stallcup, W.B. The NG2 proteoglycan: past insights and future prospects. J. Neurocytol. 31, 423–435 (2002).
Horner, P.J. et al. Proliferation and differentiation of progenitor cells throughout the intact adult rat spinal cord. J. Neurosci. 20, 2218–2228 (2000).
Dawson, M.R., Polito, A., Levine, J.M. & Reynolds, R. NG2-expressing glial progenitor cells: an abundant and widespread population of cycling cells in the adult rat CNS. Mol. Cell. Neurosci. 24, 476–488 (2003).
Volpe, J.J. Neurobiology of periventricular leukomalacia in the premature infant. Pediatr. Res. 50, 553–562 (2001).
Levine, J.M. Increased expression of the NG2 chondroitin-sulfate proteoglycan after brain injury. J. Neurosci. 14, 4716–4730 (1994).
Keirstead, H.S., Levine, J.M. & Blakemore, W. Response of the oligodendrocyte progenitor cell population (defined by NG2 labelling) to demyelination of the adult spinal cord. Glia 22, 161–170 (1998).
Gensert, J.M. & Goldman, J.E. Endogenous progenitors remyelinate demyelinated axons in the adult CNS. Neuron 19, 197–203 (1997).
McTigue, D.M., Wei, P. & Stokes, B.T. Proliferation of NG2-positive cells and altered oligodendrocyte numbers in the contused rat spinal cord. J. Neurosci. 21, 3392–3400 (2001).
Horner, P.J., Thallmair, M. & Gage, F.H. Defining the NG2-expressing cell of the adult CNS. J. Neurocytol. 31, 469–480 (2002).
Levine, J.M., Reynolds, R. & Fawcett, J.W. The oligodendrocyte precursor cell in health and disease. Trends Neurosci. 24, 39–47 (2001).
Groves, A.K. et al. Repair of demyelinated lesions by transplantation of purified O-2A progenitor cells. Nature 362, 453–455 (1993).
Kondo, T. & Raff, M. Oligodendrocyte precursor cells reprogrammed to become multipotential CNS stem cells. Science 289, 1754–1757 (2000).
Peppiatt, C.M., Howarth, C., Mobbs, P. & Attwell, D. Bidirectional control of CNS capillary diameter by pericytes. Nature 443, 700–704 (2006).
Ligon, K.L. et al. The oligodendroglial lineage marker Olig2 is universally expressed in diffuse gliomas. J. Neuropathol. Exp. Neurol. 63, 499–509 (2004).
Ligon, K.L. et al. Developmental of NG2 neural progenitor cells requires Olig gene function. Proc. Natl. Acad. Sci. USA 103, 7853–7858 (2006).
Káradóttir, R., Cavelier, P., Bergersen, L. & Attwell, D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438, 1162–1168 (2005).
Káradóttir, R. & Attwell, D. Combining patch-clamping of cells in brain slices with immunocytochemical labelling to define cell type and developmental stage. Nat. Protoc. 1, 1977–1986 (2006).
Dawson, M.R.L., Levine, J.M. & Reynolds, R. NG2-expressing cells in the central nervous system: are they oligodendroglial progenitors? J. Neurosci. Res. 61, 471–479 (2000).
Alonso, G. NG2 proteoglycan-expressing cells of the adult rat brain: possible involvement in the formation of glial scar astrocytes following stab wound. Glia 49, 318–338 (2005).
Chittajallu, R., Aguirre, A. & Gallo, V. NG2-positive cells in the mouse white and grey matter display distinct physiological properties. J. Physiol. (Lond.) 561, 109–122 (2004).
Dayer, A.G., Cleaver, K.M., Abouantoun, T. & Cameron, H.A. New GABAergic interneurons in the adult neocortex and striatum are generated from different precursors. J. Cell Biol. 168, 415–427 (2005).
Kukley, M., Capetillo-Zarate, E. & Dietrich, D. Vesicular release of glutamate from axons in white matter. Nat. Neurosci. 10, 311–320 (2007).
Ziskin, J.L., Nishiyama, A., Rubio, M., Fukaya, M. & Bergles, D.E. Vesicular release of glutamate from unmyelinated axons in white matter. Nat. Neurosci. 10, 321–330 (2007).
Follett, P.L., Rosenberg, P.A., Volpe, J.J. & Jensen, F.E. NBQX attenuates excitotoxic injury in developing white matter. J. Neurosci. 20, 9235–9241 (2000).
Káradóttir, R. & Attwell, D. Neurotransmitter receptors in the life and death of oligodendrocytes. Neuroscience 145, 1426–1438 (2007).
Allen, N.J. & Attwell, D. The effect of simulated ischaemia on spontaneous GABA release in area CA1 of the juvenile rat hippocampus. J. Physiol. (Lond.) 561, 485–498 (2004).
Li, S., Mealing, G.A., Morley, P. & Stys, P.K. Novel injury mechanism in anoxia and trauma of spinal cord white matter: glutamate release via reverse Na+-dependent glutamate transport. J. Neurosci. 19, RC16 (1999).
Barres, B.A., Koroshetz, W.J., Swartz, K.J., Chun, L.L. & Corey, D.P. Ion channel expression by white matter glia: the O-2A glial progenitor cell. Neuron 4, 507–524 (1990).
Newell, E.W. & Schlichter, L.C. Integration of K+ and Cl− currents regulate steady-state and dynamic membrane potentials in cultured rat microglia. J. Physiol. (Lond.) 567, 869–890 (2005).
Chittajallu, R., Aguirre, A.A. & Gallo, V. Downregulation of platelet-derived growth factor alpha receptor–mediated tyrosine kinase activity as a cellular mechanism for K+-channel regulation during oligodendrocyte development in situ. J. Neurosci. 25, 8601–8610 (2005).
Back, S.A. et al. Late oligodendrocyte progenitors coincide with the developmental window of vulnerability for human perinatal white matter injury. J. Neurosci. 21, 1302–1312 (2001).
Kawasaki, K., Traynelis, S.F. & Dingledine, R. Different responses of CA1 and CA3 regions to hypoxia in rat hippocampal slice. J. Neurophysiol. 63, 385–394 (1990).
Vornov, J.J., Tasker, R.C. & Coyle, J.T. Direct observation of the agonist-specific regional vulnerability to glutamate, NMDA, and kainate neurotoxicity in organotypic hippocampal cultures. Exp. Neurol. 114, 11–22 (1991).
Fukuda, A. et al. NMDA receptor–mediated differential laminar susceptibility to the intracellular Ca2+ accumulation induced by oxygen-glucose deprivation in rat neocortical slices. J. Neurophysiol. 79, 430–438 (1998).
Patt, S. et al. Neuron-like physiological properties of cells from human oligodendroglial tumors. Neuroscience 71, 601–611 (1996).
Marie, Y. et al. Olig2 as a specific marker of oligodendroglial tumour cells. Lancet 358, 298–300 (2001).
Allen, N.J., Káradóttir, R. & Attwell, D. A preferential role for glycolysis in preventing the anoxic depolarization of rat hippocampal area CA1 pyramidal cells. J. Neurosci. 25, 848–859 (2005).
Kondo, R.P., Wang, S.Y., John, S.A., Weiss, J.N. & Goldhaber, J.I. Metabolic inhibition activates a non-selective current through connexin hemichannels in isolated ventricular myocytes. J. Mol. Cell. Cardiol. 32, 1859–1872 (2000).
Acknowledgements
We thank W. Stallcup (Burnham Institute) for NG2 antibody, D. Rowitch, C.D. Stiles and J. Alberta (Harvard University) for Olig2 antibody, and R. Mirsky, K. Jessen, L. Jimenes-Diaz, S. Rakic, C. Eder, P. Mobbs, G. Frugier, A. Silver, A. Gibb and S. Bolsover for other antibodies, tissue and advice. This work was supported by the Wellcome Trust, a Wolfson-Royal Society Award to D.A. and a Royal Society Dorothy Hodgkin Fellowship to R.K.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Káradóttir, R., Hamilton, N., Bakiri, Y. et al. Spiking and nonspiking classes of oligodendrocyte precursor glia in CNS white matter. Nat Neurosci 11, 450–456 (2008). https://doi.org/10.1038/nn2060
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn2060
This article is cited by
-
Clemastine and metformin extend the window of NMDA receptor surface expression in ageing oligodendrocyte precursor cells
Scientific Reports (2024)
-
Characterization of a new mouse line triggering transient oligodendrocyte progenitor depletion
Scientific Reports (2023)
-
Insights into Alzheimer’s disease from single-cell genomic approaches
Nature Neuroscience (2023)
-
A Narrative Review on Axonal Neuroprotection in Multiple Sclerosis
Neurology and Therapy (2022)
-
Neurodevelopmental toxicity assessment of flame retardants using a human DNT in vitro testing battery
Cell Biology and Toxicology (2022)