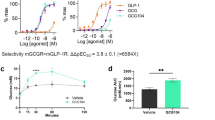
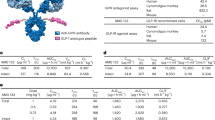
Abstract
We report the efficacy of a new peptide with agonism at the glucagon and GLP-1 receptors that has potent, sustained satiation-inducing and lipolytic effects. Selective chemical modification to glucagon resulted in a loss of specificity, with minimal change to inherent activity. The structural basis for the co-agonism appears to be a combination of local positional interactions and a change in secondary structure. Two co-agonist peptides differing from each other only in their level of glucagon receptor agonism were studied in rodent obesity models. Administration of PEGylated peptides once per week normalized adiposity and glucose tolerance in diet-induced obese mice. Reduction of body weight was achieved by a loss of body fat resulting from decreased food intake and increased energy expenditure. These preclinical studies indicate that when full GLP-1 agonism is augmented with an appropriate degree of glucagon receptor activation, body fat reduction can be substantially enhanced without any overt adverse effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ogden, C.L. et al. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA 295, 1549–1555 (2006).
Neovius, M. & Narbro, K. Cost-effectiveness of pharmacological anti-obesity treatments: a systematic review. Int. J. Obes. 32, 1752–1763 (2008).
Cvetkovic, R.S. & Plosker, G.L. Exenatide - a review of its use in patients with type 2 diabetes mellitus (as an adjunct to metformin and/or a sulfonylurea). Drugs 67, 935–954 (2007).
Gutniak, M., Orskov, C., Holst, J.J., Ahren, B. & Efendic, S. Antidiabetogenic effect of glucagon-like peptide-1 (7–36)amide in normal subjects and patients with diabetes-mellitus. N. Engl. J. Med. 326, 1316–1322 (1992).
Roth, J.D. et al. Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc. Natl. Acad. Sci. USA 105, 7257–7262 (2008).
Woods, S.C., Lutz, T.A., Geary, N. & Langhans, W. Pancreatic signals controlling food intake; insulin, glucagon and amylin. Philos. Trans. R. Soc. Lond. B Biol. Sci. 361, 1219–1235 (2006).
Geary, N., Kissileff, H.R., Pisunyer, F.X. & Hinton, V. Individual, but not simultaneous, glucagon and cholecystokinin infusions inhibit feeding in men. Am. J. Physiol. 262, R975–R980 (1992).
Geary, N. Pancreatic glucagon signals postprandial satiety. Neurosci. Biobehav. Rev. 14, 323–338 (1990).
Geary, N. Glucagon and the control of meal size. in Satiation: From Gut To Brain (ed. Smith, G.P.) 164–197 (Oxford University Press, USA, 1998).
Goodridge, A.G. & Ball, E.G. Studies on metabolism of adipose tissue. 18. In vitro effects of insulin epinephrine and glucagon on lipolysis and glycolysis in pigeon adipose tissue. Comp. Biochem. Physiol. 16, 367–381 (1965).
Heckemeyer, C.M., Barker, J., Duckworth, W.C. & Solomon, S.S. Studies of the biological effect and degradation of glucagon in the rat perifused isolated adipose cell. Endocrinology 113, 270–276 (1983).
Davidson, I.W.F., Salter, J.M. & Best, C.H. The effect of glucagon on the metabolic rate of rats. Am. J. Clin. Nutr. 8, 540–546 (1960).
Salter, J.M. Metabolic effects of glucagon in the Wistar rat. Am. J. Clin. Nutr. 8, 535–539 (1960).
Chan, E.K. et al. Suppression of weight-gain by glucagon in obese Zucker rats. Exp. Mol. Pathol. 40, 320–327 (1984).
Hjorth, S.A., Adelhorst, K., Pedersen, B.B., Kirk, O. & Schwartz, T.W. Glucagon and glucagon-like peptide 1: selective receptor recognition via distinct peptide epitopes. J. Biol. Chem. 269, 30121–30124 (1994).
Orskov, C. Glucagon-like peptide-1, a new hormone of the enteroinsular axis. Diabetologia 35, 701–711 (1992).
Wynne, K. et al. Subcutaneous oxyntomodulin reduces body weight in overweight and obese subjects - a double-blind, randomized, controlled trial. Diabetes 54, 2390–2395 (2005).
Baggio, L.L., Huang, Q.L., Brown, T.J. & Drucker, D.J. Oxyntomodulin and glucagon-like peptide-1 differentially regulate murine food intake and energy expenditure. Gastroenterology 127, 546–558 (2004).
Small, C.J. & Bloom, S.R. Gut hormones and the control of appetite. Trends Endocrinol. Metab. 15, 259–263 (2004).
Cohen, M.A. et al. Oxyntomodulin suppresses appetite and reduces food intake in humans. J. Clin. Endocrinol. Metab. 88, 4696–4701 (2003).
Billington, C.J., Bartness, T.J., Briggs, J., Levine, A.S. & Morley, J.E. Glucagon stimulation of brown adipose-tissue growth and thermogenesis. Am. J. Physiol. 252, R160–R165 (1987).
Runge, S. et al. Three distinct epitopes on the extracellular face of the glucagon receptor determine specificity for the glucagon amino terminus. J. Biol. Chem. 278, 28005–28010 (2003).
Runge, S., Wulff, B.S., Madsen, K., Brauner-Osborne, H. & Knudsen, L.B. Different domains of the glucagon and glucagon-like peptide-1 receptors provide the critical determinants of ligand selectivity. Br. J. Pharmacol. 138, 787–794 (2003).
Neidigh, J.W., Fesinmeyer, R.M., Prickett, K.S. & Andersen, N.H. Exendin-4 and glucagon-like-peptide-1: NMR structural comparisons in the solution and micelle-associated states. Biochemistry 40, 13188–13200 (2001).
Runge, S., Thogersen, H., Madsen, K., Lau, J. & Rudolph, R. Crystal structure of the ligand-bound glucagon-like peptide-1 receptor extracellular domain. J. Biol. Chem. 283, 11340–11347 (2008).
Scrocchi, L.A. et al. Glucose intolerance but normal satiety in mice with a null mutation in the glucagon-like peptide 1 receptor gene. Nat. Med. 2, 1254–1258 (1996).
Rucker, D., Padwal, R., Li, S.K., Curioni, C. & Lau, D.C.W. Long term pharmacotherapy for obesity and overweight: updated meta-analysis. BMJ 335, 1194–1199 (2007).
Christensen, R., Kristensen, P.K., Bartels, E.M., Blidda, H. & Astrup, A. Efficacy and safety of the weight-loss drug rimonabant: a meta-analysis of randomised trials. Lancet 370, 1706–1713 (2007).
Griffen, L. & Anchors, M. Asymptomatic mitral and aortic valve disease is seen in half of the patients taking 'Phen-Fen'. Arch. Intern. Med. 158, 102 (1998).
Amori, R.E., Lau, J. & Pittas, A.G. Efficacy and safety of incretin therapy in type 2 diabetes - systematic review and meta-analysis. JAMA 298, 194–206 (2007).
Savitzky, A. & Golay, M.J.E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 36, 1627–1639 (1964).
Whitmore, L. & Wallace, B.A. Protein secondary structure analyses from circular dichroism spectroscopy: methods and reference databases. Biopolymers 89, 392–400 (2008).
Whitmore, L. & Wallace, B.A. DICHROWEB, an online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic Acids Res. 32, W668–W673 (2004).
Tinsley, F.C., Taicher, G.Z. & Heiman, M.L. Evaluation of a quantitative magnetic resonance method for mouse whole body composition analysis. Obes. Res. 12, 150–160 (2004).
Acknowledgements
We thank K. Brown for skilled technical assistance and R. Seeley (University of Cincinnati), D. D'Alessio (University of Cincinnati) and S. Vignati (Marcadia Biotech) for expert advice and helpful discussions. We are grateful to J. Levy for his assistance in peptide synthesis, purification and characterization; J. Karty (Indiana University) and A. Hansen (Indiana University) for their expertise and support in mass spectrometry data collection and analysis; and B. Yang (Marcadia Biotech) for providing Fmoc l-homocysteic acid.
Author information
Authors and Affiliations
Contributions
J.W.D. designed, synthesized and characterized all peptides and co-wrote the manuscript. N.O. co-planned and led all in vivo studies and co-wrote the manuscript. J.T.P., V.G., D.S. and J.G. gave advice on chemical design, interpreted biological data and co-wrote the manuscript. H.F. and D.B. performed adipocyte studies, interpreted results and co-wrote the manuscript. D.J.D. provided mouse models, interpreted data and co-wrote the manuscript. W.A. performed in vitro studies including western blotting. N.C., J. Holland, J. Hembree, E.G., J.R., H.W., H.K. and S.H.L. co-performed all in vivo pharmacology and metabolism studies as a team. S.H. performed cholesterol and lipoprotein analysis studies. S.C.W. gave advice on experimental design, interpreted data and co-wrote the manuscript. R.N., P.T.P. and D.P.-T. co-planned, co-performed and supervised all in vivo and ex vivo biology studies. R.D. and M.H.T. conceptualized, analyzed and interpreted all studies and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.D., M.T. and J.G. have partial ownership in Marcadia Biotech. Marcadia Biotech has provided partial research support for this work.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Results (PDF 6938 kb)
Rights and permissions
About this article
Cite this article
Day, J., Ottaway, N., Patterson, J. et al. A new glucagon and GLP-1 co-agonist eliminates obesity in rodents. Nat Chem Biol 5, 749–757 (2009). https://doi.org/10.1038/nchembio.209
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.209
This article is cited by
-
Polyagonists in Type 2 Diabetes Management
Current Diabetes Reports (2024)
-
Poly-Agonist Pharmacotherapies for Metabolic Diseases: Hopes and New Challenges
Drugs (2024)
-
Efficacy and safety of tirzepatide, dual GLP-1/GIP receptor agonists, in the management of type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials
Diabetology & Metabolic Syndrome (2023)
-
Effects of site-directed mutagenesis of GLP-1 and glucagon receptors on signal transduction activated by dual and triple agonists
Acta Pharmacologica Sinica (2023)
-
Gut hormone co-agonists for the treatment of obesity: from bench to bedside
Nature Metabolism (2023)