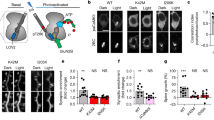
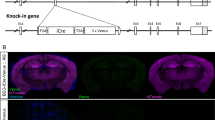
Abstract
Long-term potentiation (LTP), a well-characterized form of synaptic plasticity, has long been postulated as a cellular correlate of learning and memory. Although LTP can persist for long periods of time1, the mechanisms underlying LTP maintenance, in the midst of ongoing protein turnover and synaptic activity, remain elusive. Sustained activation of the brain-specific protein kinase C (PKC) isoform protein kinase M-ζ (PKM-ζ) has been reported to be necessary for both LTP maintenance and long-term memory2. Inhibiting PKM-ζ activity using a synthetic zeta inhibitory peptide (ZIP) based on the PKC-ζ pseudosubstrate sequence reverses established LTP in vitro and in vivo3,4. More notably, infusion of ZIP eliminates memories for a growing list of experience-dependent behaviours, including active place avoidance4, conditioned taste aversion5, fear conditioning and spatial learning6. However, most of the evidence supporting a role for PKM-ζ in LTP and memory relies heavily on pharmacological inhibition of PKM-ζ by ZIP. To further investigate the involvement of PKM-ζ in the maintenance of LTP and memory, we generated transgenic mice lacking PKC-ζ and PKM-ζ. We find that both conventional and conditional PKC-ζ/PKM-ζ knockout mice show normal synaptic transmission and LTP at Schaffer collateral–CA1 synapses, and have no deficits in several hippocampal-dependent learning and memory tasks. Notably, ZIP still reverses LTP in PKC-ζ/PKM-ζ knockout mice, indicating that the effects of ZIP are independent of PKM-ζ.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Abraham, W. C., Logan, B., Greenwood, J. M. & Dragunow, M. Induction and experience-dependent consolidation of stable long-term potentiation lasting months in the hippocampus. J. Neurosci. 22, 9626–9634 (2002)
Sacktor, T. C. PKMζ, LTP maintenance, and the dynamic molecular biology of memory storage. Prog. Brain Res. 169, 27–40 (2008)
Ling, D. S. et al. Protein kinase Mζ is necessary and sufficient for LTP maintenance. Nature Neurosci. 5, 295–296 (2002)
Pastalkova, E. et al. Storage of spatial information by the maintenance mechanism of LTP. Science 313, 1141–1144 (2006)
Shema, R., Sacktor, T. C. & Dudai, Y. Rapid erasure of long-term memory associations in the cortex by an inhibitor of PKMζ. Science 317, 951–953 (2007)
Serrano, P. et al. PKMζ maintains spatial, instrumental, and classically conditioned long-term memories. PLoS Biol. 6, e318 (2008)
Hernandez, A. I. et al. Protein kinase Mζ synthesis from a brain mRNA encoding an independent protein kinase Cζ catalytic domain. Implications for the molecular mechanism of memory. J. Biol. Chem. 278, 40305–40316 (2003)
Hirai, T., Niino, Y. S. & Chida, K. PKCζII, a small molecule of protein kinase Cζ, specifically expressed in the mouse brain. Neurosci. Lett. 348, 151–154 (2003)
Sajikumar, S. & Korte, M. Metaplasticity governs compartmentalization of synaptic tagging and capture through brain-derived neurotrophic factor (BDNF) and protein kinase Mζ (PKMζ). Proc. Natl Acad. Sci. USA 108, 2551–2556 (2011)
Erdmann, G., Schutz, G. & Berger, S. Inducible gene inactivation in neurons of the adult mouse forebrain. BMC Neurosci. 8, 63 (2007)
Lisman, J. Memory erasure by very high concentrations of ZIP may not be due to PKM-zeta. Hippocampus 22, 648–649 (2012)
Wu-Zhang, A. X., Schramm, C. L., Nabavi, S., Malinow, R. & Newton, A. C. Cellular pharmacology of protein kinase Mζ (PKMζ) contrasts with its in vitro profile. J. Biol. Chem. 287, 12879–12885 (2012)
Sacktor, T. C. & Fenton, A. A. Appropriate application of ZIP for PKMζ inhibition, LTP reversal, and memory erasure. Hippocampus 22, 645–647 (2012)
Yao, Y. et al. Matching biochemical and functional efficacies confirm ZIP as a potent competitive inhibitor of PKMζ in neurons. Neuropharmacol. 64, 37–44 (2013)
Serrano, P., Yao, Y. & Sacktor, T. C. Persistent phosphorylation by protein kinase Mζ maintains late-phase long-term potentiation. J. Neurosci. 25, 1979–1984 (2005)
Sajikumar, S., Navakkode, S., Sacktor, T. C. & Frey, J. U. Synaptic tagging and cross-tagging: the role of protein kinase Mζ in maintaining long-term potentiation but not long-term depression. J. Neurosci. 25, 5750–5756 (2005)
Kwapis, J. L., Jarome, T., Lonegran, M. & Helmstetter, F. Protein kinase Mzeta maintains fear memory in the amygdala but not in the hippocampus. Behav. Neurosci. 123, 844–850 (2009)
Mei, F., Nagappan, G., Ke, Y., Sacktor, T. C. & Lu, B. BDNF facilitates L–LTP maintenance in the absence of protein synthesis through PKMζ. PLoS ONE 6, e21568 (2011)
Krotova, K. et al. Peptides modified by myristoylation activate eNOS in endothelial cells through Akt phosphorylation. Br. J. Pharmacol. 148, 732–740 (2006)
Lim, S. et al. A myristoylated pseudosubstrate peptide of PKC-ζ induces degranulation in HMC-1 cells independently of PKC-ζ activity. Life Sci. 82, 733–740 (2008)
Denny, J. B., Polan-Curtain, J., Rodriguez, S., Wayner, M. J. & Armstrong, D. L. Evidence that protein kinase M does not maintain long-term potentiation. Brain Res. 534, 201–208 (1990)
Huang, Y. Y. & Kandel, E. R. Recruitment of long-lasting and protein kinase A-dependent long-term potentiation in the CA1 region of hippocampus requires repeated tetanization. Learn. Mem. 1, 74–82 (1994)
Kim, S. J. & Linden, D. J. Ubiquitous plasticity and memory storage. Neuron 56, 582–592 (2007)
McEchron, M. D., Bouwmeester, H., Tseng, W., Weiss, C. & Disterhoft, J. F. Hippocampectomy disrupts auditory trace fear conditioning and contextual fear conditioning in the rat. Hippocampus 8, 638–646 (1998)
Ling, D. S., Benardo, L. S. & Sacktor, T. C. Protein kinase Mζ enhances excitatory synaptic transmission by increasing the number of active postsynaptic AMPA receptors. Hippocampus 16, 443–452 (2006)
Shema, R. et al. Enhancement of consolidated long-term memory by overexpression of protein kinase Mζ in the neocortex. Science 331, 1207–1210 (2011)
Makuch, L. et al. Regulation of AMPA receptor function by the human memory-associated gene KIBRA. Neuron 71, 1022–1029 (2011)
Rodríguez, C. I. et al. High-efficiency deleter mice show that FLPe is an alternative to Cre-loxP. Nature Genet. 25, 139–140 (2000)
Acknowledgements
We thank G. Schütz for providing the CaMKII Cre-ERT2 mice, M. Gallagher and D. Smith for advice on behaviour and M. Coulter for technical support. We also thank all members of the Huganir lab for discussion and support. This work was supported by grants from the National Institute of Health (NS36715) and the Howard Hughes Medical Institute (to R.L.H.). L.J.V. is supported by a training grant from the National Institute of Health (T32MH15330).
Author information
Authors and Affiliations
Contributions
L.J.V., J.L.B. and R.L.H. designed experiments. L.J.V. and J.L.B performed experiments and analysed data. R.J. designed and generated the knockout animals. Y.Y. assisted in mating and genotyping mouse lines. L.J.V., J.L.B. and R.L.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Under a licensing agreement between Millipore Corporation and The Johns Hopkins University, R.L.H. is entitled to a share of royalties received by the University on sales of products described in this article. R.L.H. is a paid consultant to Millipore Corporation. The terms of this arrangement are being managed by The Johns Hopkins University in accordance with its conflict-of-interest policies.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-6. (PDF 771 kb)
Rights and permissions
About this article
Cite this article
Volk, L., Bachman, J., Johnson, R. et al. PKM-ζ is not required for hippocampal synaptic plasticity, learning and memory. Nature 493, 420–423 (2013). https://doi.org/10.1038/nature11802
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11802
This article is cited by
-
Sex differences in pre- and post-synaptic glutamate signaling in the nucleus accumbens core
Biology of Sex Differences (2023)
-
Modeling suggests combined-drug treatments for disorders impairing synaptic plasticity via shared signaling pathways
Journal of Computational Neuroscience (2021)
-
Zeta Inhibitory Peptide attenuates learning and memory by inducing NO-mediated downregulation of AMPA receptors
Nature Communications (2020)
-
GSK-3β activation is required for ZIP-induced disruption of learned fear
Scientific Reports (2020)
-
Neuronal Adenylyl Cyclase Targeting Central Plasticity for the Treatment of Chronic Pain
Neurotherapeutics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.