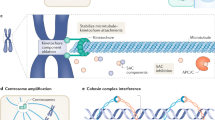
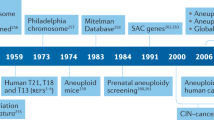
Abstract
In contrast to normal cells, aneuploidy — alterations in the number of chromosomes — is consistently observed in virtually all cancers. A growing body of evidence suggests that aneuploidy is often caused by a particular type of genetic instability, called chromosomal instability, which may reflect defects in mitotic segregation in cancer cells. A better understanding of the molecular mechanisms leading to aneuploidy holds promise for the development of cancer drugs that target this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Nowell, P. C. The clonal evolution of tumor cell populations. Science 194, 23–28 (1976).
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
Knudson, A. G. Two genetic hits (more or less) to cancer. Nature Rev. Cancer 1, 157–162 (2001).
Nowak, M. A. et al. The role of chromosomal instability in tumor initiation. Proc. Natl Acad. Sci. USA 99, 16226–16231 (2002).
Hansemann, D. Ueber asymmetrische Zelltheilung in Epithelkrebsen und deren biologische Bedeutung. Virschows Arch. Pathol. Anat. 119, 299–326 (1890).
Boveri, T. Zur Frage der Enstehung maligner Tumoren (Gustav Fischer Verlag, Jena, 1914).
Duesberg, P. et al. How aneuploidy may cause cancer and genetic instability. Anticancer Res. 19, 4887–4906 (1999).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643–649 (1998).
Hansemann, D. Ueber patologische Mitosen. Arch. Pathol. Anat. Phys. Klin. Med. 119, 299–326 (1891).
Saunders, W. S. et al. Chromosomal instability and cytoskeletal defects in oral cancer cells. Proc. Natl Acad. Sci. USA 97, 303–308 (2000).
Gisselsson, D. et al. Centrosomal abnormalities, multipolar mitoses, and chromosomal instability in head and neck tumours with dysfunctional telomeres. Br. J. Cancer 87, 202–207 (2002).
Dutrillaux, B., Gerbault-Seureau, M., Remvikos, Y., Zafrani, B. & Prieur, M. Breast cancer genetic evolution: I. Data from cytogenetics and DNA content. Breast Cancer Res. Treat. 19, 245–255 (1991).
Reid, B. J., Haggitt, R. C., Rubin, C. E. & Rabinovitch, P. S. Barrett's esophagus: correlation between flow cytometry and histology in detection of patients at risk for adenocarcinoma. Gastroenterology 93, 1–11 (1987).
Levine, D. S. et al. Distribution of aneuploid cell populations in ulcerative colitis with dysplasia or cancer. Gastroenterology 101, 1198–1210 (1991).
der-Sarkissian, H., Bacchetti, S., Cazes, L. & Londono-Vallejo, J. A. The shortest telomeres drive karyotype evolution in transformed cells. Oncogene 23, 1221–1228 (2004).
Meraldi, P., Honda, R. & Nigg, E. A. Aurora-A overexpression reveals tetraploidization as a major route to centrosome amplification in p53−/− cells. EMBO J. 21, 483–492 (2002).
Lingle, W. L. et al. Centrosome amplification drives chromosomal instability in breast tumor development. Proc. Natl Acad. Sci. USA 99, 1978–1983 (2002).
Pihan, G. A. et al. Centrosome defects and genetic instability in malignant tumors. Cancer Res. 58, 3974–3985 (1998).
Lynch, H. T. & de la Chapelle, A. Hereditary colorectal cancer. N. Engl. J. Med. 348, 919–932 (2003).
Marra, G. & Boland, C. R. Hereditary nonpolyposis colorectal cancer: the syndrome, the genes, and historical perspectives. J. Natl Cancer Inst. 87, 1114–1125 (1995).
Peltomaki, P. Deficient DNA mismatch repair: a common etiologic factor for colon cancer. Hum. Mol. Genet. 10, 735–740 (2001).
Yamamoto, H., Imai, K. & Perucho, M. Gastrointestinal cancer of the microsatellite mutator phenotype pathway. J. Gastroenterol. 37, 153–163 (2002).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997).
Heim, S. & Mitelman, F. in Cancer Cytogenetics (eds Heim, S. & Mitelman, F.) 369–388 (Wiley-Liss, New York, 1995).
Cahill, D. P. et al. Mutations of mitotic checkpoint genes in human cancers. Nature 392, 300–303 (1998).
Tighe, A., Johnson, V. L., Albertella, M. & Taylor, S. S. Aneuploid colon cancer cells have a robust spindle checkpoint. EMBO Rep. 2, 609–614 (2001).
Hoyt, M. A. A new view of the spindle checkpoint. J. Cell Biol. 154, 909–912 (2001).
Fukagawa, T. Assembly of kinetochores in vertebrate cells. Exp. Cell Res. 296, 21–27 (2004).
Cleveland, D. W., Mao, Y. & Sullivan, K. F. Centromeres and kinetochores: from epigenetics to mitotic checkpoint signaling. Cell 112, 407–421 (2003).
Rajagopalan, H., Nowak, M. A., Vogelstein, B. & Lengauer, C. The significance of unstable chromosomes in colorectal cancer. Nature Rev. Cancer 3, 695–701 (2003).
Bardelli, A. et al. Carcinogen-specific induction of genetic instability. Proc. Natl Acad. Sci. USA 98, 5770–5775 (2001).
Dai, W. et al. Slippage of mitotic arrest and enhanced tumor development in mice with BubR1 haploinsufficiency. Cancer Res. 64, 440–445 (2004).
Hanks, S. et al. Constitutional aneuploidy and cancer predisposition caused by biallelic BUB1B mutations. Nature Genet. advance online publication 10 October 2004 (doi:10.1038/ng1449).
Li, Y., Gorbea, C., Mahaffey, D., Rechsteiner, M. & Benezra, R. MAD2 associates with the cyclosome/anaphase-promoting complex and inhibits its activity. Proc. Natl Acad. Sci. USA 94, 12431–12436 (1997).
Michel, L. S. et al. MAD2 haplo-insufficiency causes premature anaphase and chromosome instability in mammalian cells. Nature 409, 355–359 (2001).
Hernando, E. et al. Rb inactivation promotes genomic instability by uncoupling cell cycle progression from mitotic control. Nature 430, 797–802 (2004).
Wang, Z. et al. Three classes of genes mutated in colorectal cancers with chromosomal instability. Cancer Res. 64, 2998–3001 (2004).
Zhou, H. et al. Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nature Genet. 20, 189–193 (1998).
Strohmaier, H. et al. Human F-box protein hCdc4 targets cyclin E for proteolysis and is mutated in a breast cancer cell line. Nature 413, 316–322 (2001).
Rajagopalan, H. et al. Inactivation of hCDC4 can cause chromosomal instability. Nature 428, 77–81 (2004).
Weaver, Z. et al. Mammary tumors in mice conditionally mutant for Brca1 exhibit gross genomic instability and centrosome amplification yet display a recurring distribution of genomic imbalances that is similar to human breast cancer. Oncogene 21, 5097–5107 (2002).
Tutt, A. et al. Absence of Brca2 causes genome instability by chromosome breakage and loss associated with centrosome amplification. Curr. Biol. 9, 1107–1110 (1999).
Spencer, F., Gerring, S. L., Connelly, C. & Hieter, P. Mitotic chromosome transmission fidelity mutants in Saccharomyces cerevisiae. Genetics 124, 237–249 (1990).
Hieter, P., Mann, C., Snyder, M. & Davis, R. W. Mitotic stability of yeast chromosomes: a colony color assay that measures nondisjunction and chromosome loss. Cell 40, 381–392 (1985).
Zhou, W. et al. Counting alleles to predict recurrence of early-stage colorectal cancers. Lancet 359, 219–225 (2002).
Watanabe, T. et al. Molecular predictors of survival after adjuvant chemotherapy for colon cancer. N. Engl. J. Med. 344, 1196–1206 (2001).
Risques, R. A. et al. Genetic pathways and genome-wide determinants of clinical outcome in colorectal cancer. Cancer Res. 63, 7206–7214 (2003).
Sawyers, C. L. Research on resistance to cancer drug Gleevec. Science 294, 1834 (2001).
Wang, T. L. et al. Digital karyotyping identifies thymidylate synthase amplification as a mechanism of resistance to 5-fluorouracil in metastatic colorectal cancer patients. Proc. Natl. Acad. Sci. USA 101, 3089–3094 (2004).
Kops, G. J., Foltz, D. R. & Cleveland, D. W. Lethality to human cancer cells through massive chromosome loss by inhibition of the mitotic checkpoint. Proc. Natl. Acad. Sci. USA 101, 8699–8704 (2004).
Acknowledgements
We thank the members of our laboratory for critical reading of this manuscript. We also thank D. Gisselsson and M. Höglund for sharing some of their unpublished thoughts on mitotic disturbances, and M. R. Speicher for providing the photograph for Fig. 1. We especially thank S. Kern and D. A. Dezentje for permission to report unpublished data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Rajagopalan, H., Lengauer, C. Aneuploidy and cancer. Nature 432, 338–341 (2004). https://doi.org/10.1038/nature03099
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature03099
This article is cited by
-
A unique case of rectal cancer with coexistence of multiple pathways of carcinogenesis
World Journal of Surgical Oncology (2023)
-
Current advances in noninvasive methods for the diagnosis of oral squamous cell carcinoma: a review
European Journal of Medical Research (2023)
-
SIX1 amplification modulates stemness and tumorigenesis in breast cancer
Journal of Translational Medicine (2023)
-
DNAJA2 deficiency activates cGAS-STING pathway via the induction of aberrant mitosis and chromosome instability
Nature Communications (2023)
-
Loss of phosphatase CTDNEP1 potentiates aggressive medulloblastoma by triggering MYC amplification and genomic instability
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.