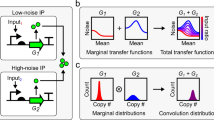
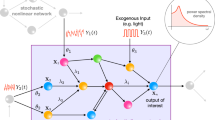
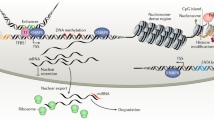
Abstract
Noise has many roles in biological function, including generation of errors in DNA replication leading to mutation and evolution, noise-driven divergence of cell fates, noise-induced amplification of signals, and maintenance of the quantitative individuality of cells. Yet there is order to the behaviour and development of cells. They operate within strict parameters and in many cases this behaviour seems robust, implying that noise is largely filtered by the system. How can we explain the use, rejection and sensitivity to noise that is found in biological systems? An exploration of the sources and consequences of noise calls for the use of stochastic models.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ko, M. S., Nakauchi, H. & Takahashi, N. The dose dependence of glucocorticoid-inducible gene expression results from changes in the number of transcriptionally active templates. EMBO J. 9, 2835–2842 (1990).
Berg, O. G. A model for the statistical fluctuations of protein numbers in a microbial population. J. Theor. Biol. 71, 587–603 (1978).
Ko, M. S. A stochastic model for gene induction. J. Theor. Biol. 153, 181–194 (1991).
McAdams, H. H. & Arkin, A. Stochastic mechanisms in gene expression. Proc. Natl Acad. Sci. USA 94, 814–819 (1997).
White, J. A., Rubinstein, J. T. & Kay, A. R. Channel noise in neurons. Trends Neurosci. 23, 131–137 (2000).
Allen, C. & Stevens, C. F. An evaluation of causes for unreliability of synaptic transmission. Proc. Natl Acad. Sci. USA 91, 10380–10383 (1994).
van Oudenaarden, A. & Theriot, J. A. Cooperative symmetry-breaking by actin polymerization in a model for cell motility. Nature Cell Biol. 1, 493–499 (1999).
Simon, S. M., Peskin, C. S. & Oster, G. F. What drives the translocation of proteins? Proc. Natl Acad. Sci. USA 89, 3770–3774 (1992).
Sternberg, P. W. & Felix, M. A. Evolution of cell lineage. Curr. Opin. Genet. Dev. 7, 543–550 (1997).
Houchmandzadeh, B., Wieschaus, E. & Leibler, S. Establishment of developmental precision and proportions in the early Drosophila embryo. Nature 415, 798–802 (2002).
Barkai, N. & Leibler, S. Robustness in simple biochemical networks. Nature 387, 913–917 (1997).
von Dassow, G., Meir, E., Munro, E. M. & Odell, G. M. The segment polarity network is a robust developmental module. Nature 406, 188–192 (2000).
Ptashne, M. A Genetic Switch : Phage Lambda and Higher Organisms (Cell Press, Blackwell Scientific Publications, Cambridge, MA, 1998).
Msadek, T. When the going gets tough: survival strategies and environmental signaling networks in Bacillus subtilis. Trends Microbiol. 7, 201–207 (1999).
Mayani, H., Dragowska, W. & Lansdorp, P. M. Lineage commitment in human hemopoiesis involves asymmetric cell division of multipotent progenitors and does not appear to be influenced by cytokines. J. Cell. Physiol. 157, 579–586 (1993).
Spudich, J. L. & Koshland, D. E. Jr Non-genetic individuality: chance in the single cell. Nature 262, 467–471 (1976).
Morgan, H. D., Sutherland, H. G., Martin, D. I. & Whitelaw, E. Epigenetic inheritance at the agouti locus in the mouse. Nature Genet. 23, 314–318 (1999).
Connell, I. et al. Type 1 fimbrial expression enhances Escherichia coli virulence for the urinary tract. Proc. Natl Acad. Sci. USA 93, 9827–9832 (1996).
Abraham, J. M., Freitag, C. S., Clements, J. R. & Eisenstein, B. I. An invertible element of DNA controls phase variation of type 1 fimbriae of Escherichia coli. Proc. Natl Acad. Sci. USA 82, 5724–5747 (1985).
Mulvey, M. A., Schilling, J. D., Martinez, J. J. & Hultgren, S. J. Bad bugs and beleaguered bladders: interplay between uropathogenic Escherichia coli and innate host defenses. Proc. Natl Acad. Sci. USA 97, 8829–8835 (2000).
Sauer, F. G., Mulvey, M. A., Schilling, J. D., Martinez, J. J. & Hultgren, S. J. Bacterial pili: molecular mechanisms of pathogenesis. Curr. Opin. Microbiol. 3, 65–72 (2000).
Mehr, I. J. & Seifert, H. S. Differential roles of homologous recombination pathways in Neisseria gonorrhoeae pilin antigenic variation, DNA transformation and DNA repair. Mol. Microbiol. 30, 697–710 (1998).
Ziebuhr, W. et al. A novel mechanism of phase variation of virulence in Staphylococcus epidermidis: evidence for control of the polysaccharide intercellular adhesin synthesis by alternating insertion and excision of the insertion sequence element IS256. Mol. Microbiol. 32, 345–356 (1999).
Peak, I. R., Jennings, M. P., Hood, D. W., Bisercic, M. & Moxon, E. R. Tetrameric repeat units associated with virulence factor phase variation in Haemophilus also occur in Neisseria spp. and Moraxella catarrhalis. FEMS Microbiol. Lett. 137, 109–114 (1996).
Wright, A. C., Powell, J. L., Kaper, J. B. & Morris, J. G. Jr Identification of a group 1-like capsular polysaccharide operon for Vibrio vulnificus. Infect. Immun. 69, 6893–6901 (2001).
Hallet, B. Playing Dr Jekyll and Mr Hyde: combined mechanisms of phase variation in bacteria. Curr. Opin. Microbiol. 4, 570–581 (2001).
Arkin, A. P. Synthetic cell biology. Curr. Opin. Biotechnol. 12, 638–644 (2001).
Slepchenko, B. M., Schaff, J. C., Carson, J. H. & Loew, L. M. Computational cell biology: spatiotemporal simulation of cellular events. Annu. Rev. Biophys. Biomol. Struct. 31, 423–441 (2002).
Gardiner, C. W. Handbook of Stochastic Methods forPhysics, Chemistry, and the Natural Sciences (Springer, Berlin, 1990).
Kloeden, P. E. & Platen, E. Numerical Solution of Stochastic Differential Equations (Springer, Berlin, 1992).
Gillespie, D. T. The chemical Langevin equation. J. Chem. Phys. 113, 297–306 (2000).
Gillespie, D. T. The chemical Langevin equation and Fokker-Planck equation for the reverisble isomerization reaction. J. Phys. Chem. A 106, 5063–5071 (2002).
Kurtz, T. G. Approximation of Population Processes (SIAM, Philadelphia, 1981).
Kohn, K. W. Molecular interaction map of the mammalian cell cycle control and DNA repair systems. Mol. Biol. Cell 10, 2703–2734 (1999).
Gillespie, D. T. Exact stochastic simulation of coupled chemical reactions. J. Phys. Chem. 81, 2340–2361 (1977).
Le Novere, N. & Shimizu, T. S. STOCHSIM: modelling of stochastic biomolecular processes. Bioinformatics 17, 575–576 (2001).
Shimizu, T. S. & Bray, D. in Foundations of Systems Biology (ed. Kitano, H.) 213–232 (MIT Press, Cambridge, MA, 2001).
Gillespie, D. T. Approximate accelerated stochastic simulation of chemically reacting systems. J. Chem. Phys. 115, 1716–1733 (2001).
Gibson, M. A. & Bruck, J. Exact stochastic simulation of chemical systems with many species and many channels. J. Phys. Chem. A 105, 1876–1889 (2000).
Rao, C. V. & Arkin, A. Stochastic chemical kinetics and the quasi steady-state assumption: application to the Gillespie algorithm. J. Chem. Phys. (in the press).
Haseltine, E. L. & Rawlings, J. B. Approximate simulation of coupled fast and slow reactions for stochastic chemical kinetics. J. Chem. Phys. 117, 6958–6969 (2002).
Arkin, A., Ross, J. & McAdams, H. H. Stochastic kinetic analysis of developmental pathway bifurcation in phage lambda-infected Escherichia coli cells. Genetics 149, 1633–1648 (1998).
Thattai, M. & van Oudenaarden, A. Intrinsic noise in gene regulatory networks. Proc. Natl Acad. Sci. USA 98, 8614–8619 (2001).
Kierzek, A. M., Zaim, J. & Zielenkiewicz, P. The effect of transcription and translation initiation frequencies on the stochastic fluctuations in prokaryotic gene expression. J. Biol. Chem. 276, 8165–8172 (2001).
Kepler, T. B. & Elston, T. C. Stochasticity in transcriptional regulation: origins, consequences, and mathematical representations. Biophys J. 81, 3116–3136 (2001).
Ozbudak, E. M., Thattai, M., Kurtser, I., Grossman, A. D. & van Oudenaarden, A. Regulation of noise in the expression of a single gene. Nature Genet. 31, 69–73 (2002).
Elowitz, M. B., Levine, A. J., Siggia, E. D. & Swain, P. S. Stochastic gene expression in a single cell. Science 297, 1183–1186 (2002).
Arkin, A. P. in Self-organized Biological Dynamics and Nonlineaer Control (ed. Walleczek, J.) 112–144 (Cambridge Univ. Press, London, 2000).
Samoilov, M., Arkin, A. & Ross, J. Signal processing by simple chemical systems. J. Phys. Chem. A (in the press).
Detwiler, P. B., Ramanathan, S., Sengupta, A. & Shraiman, B. I. Engineering aspects of enzymatic signal transduction: photoreceptors in the retina. Biophys. J. 79, 2801–2817 (2000).
Thattai, M. & Van Oudenaarden, A. Attenuation of noise in ultrasensitive signaling cascades. Biophys. J. 82, 2943–2950 (2002).
Smolen, P., Baxter, D. A. & Byrne, J. H. Modeling transcriptional control in gene networks—methods, recent results, and future directions. Bull. Math. Biol. 62, 247–292 (2000).
Fell, D. Understanding the Control of Metabolism (Portland, London, 1997).
Heinrich, R. & Schuster, S. The Regulation of Cellular Systems (Portland, London, 1996).
Becskei, A. & Serrano, L. Engineering stability in gene networks by autoregulation. Nature 405, 590–593 (2000).
Yi, T. M., Huang, Y., Simon, M. I. & Doyle, J. Robust perfect adaptation in bacterial chemotaxis through integral feedback control. Proc. Natl Acad. Sci. USA 97, 4649–4653 (2000).
McAdams, H. H. & Arkin, A. It's a noisy business! Genetic regulation at the nanomolar scale. Trends Genet. 15, 65–69 (1999).
Cook, D. L., Gerber, A. N. & Tapscott, S. J. Modeling stochastic gene expression: implications for haploinsufficiency. Proc. Natl Acad. Sci. USA 95, 15641–15646 (1998).
Hartwell, L. H. & Weinert, T. A. Checkpoints: controls that ensure the order of cell cycle events. Science 246, 629–634 (1989).
Rodnina, M. V. & Wintermeyer, W. Ribosome fidelity: tRNA discrimination, proofreading and induced fit. Trends Biochem. Sci. 26, 124–130 (2001).
Wolf, D. M. & Arkin, A. P. Fifteen minutes of fim: control of type 1 pili expression in E. coli. Omics 6, 91–114 (2002).
Morton-Firth, C. J. & Bray, D. Predicting temporal fluctuations in an intracellular signalling pathway. J. Theor. Biol. 192, 117–128 (1998).
Bren, A. & Eisenbach, M. Changing the direction of flagellar rotation in bacteria by modulating the ratio between the rotational states of the switch protein FliM. J. Mol. Biol. 312, 699–709 (2001).
Ferrell, J. E. Self-perpetuating states in signal transduction: positive feedback, double-negative feedback and bistability. Curr. Opin. Cell Biol. 14, 140–148 (2002).
Hasty, J., Pradines, J., Dolnik, M. & Collins, J. J. Noise-based switches and amplifiers for gene expression. Proc. Natl Acad. Sci. USA 97, 2075–2080 (2000).
Becskei, A., Seraphin, B. & Serrano, L. Positive feedback in eukaryotic gene networks: cell differentiation by graded to binary response conversion. EMBO J. 20, 2528–2535 (2001).
Gammaitoni, L., Hanggi, P., Jung, P. & Marchesoni, F. Stochastic resonance. Rev. Mod. Phys. 70, 223–287 (1998).
Russell, D. F., Wilkens, L. A. & Moss, F. Use of behavioural stochastic resonance by paddle fish for feeding. Nature 402, 291–294 (1999).
Douglass, J. K., Wilkens, L., Pantazelou, E. & Moss, F. Noise enhancement of information transfer in crayfish mechanoreceptors by stochastic resonance. Nature 365, 337–340 (1993).
Levin, J. E. & Miller, J. P. Broadband neural encoding in the cricket cercal sensory system enhanced by stochastic resonance. Nature 380, 165–168 (1996).
Paulsson, J., Berg, O. G. & Ehrenberg, M. Stochastic focusing: fluctuation-enhanced sensitivity of intracellular regulation. Proc. Natl Acad. Sci. USA 97, 7148–7153 (2000).
Elowitz, M. B. & Leibler, S. A synthetic oscillatory network of transcriptional regulators. Nature 403, 335–338 (2000).
Gardner, T. S., Cantor, C. R. & Collins, J. J. Construction of a genetic toggle switch in Escherichia coli. Nature 403, 339–342 (2000).
Barkai, N. & Leibler, S. Circadian clocks limited by noise. Nature 403, 267–268 (2000).
Gonze, D., Halloy, J. & Goldbeter, A. Robustness of circadian rhythms with respect to molecular noise. Proc. Natl Acad. Sci. USA 99, 673–678 (2002).
Vilar, J. M., Kueh, H. Y., Barkai, N. & Leibler, S. Mechanisms of noise-resistance in genetic oscillators. Proc. Natl Acad. Sci. USA 99, 5988–5992 (2002).
Smolen, P., Baxter, D. A. & Byrne, J. H. Modeling circadian oscillations with interlocking positive and negative feedback loops. J. Neurosci. 21, 6644–6656 (2001).
Kastner, J., Solomon, J. & Fraser, S. Modeling a hox gene network in silico using a stochastic simulation algorithm. Dev. Biol. 246, 122–131 (2002).
Levin, M. D., Morton-Firth, C. J., Abouhamad, W. N., Bourret, R. B. & Bray, D. Origins of individual swimming behavior in bacteria. Biophys. J. 74, 175–181 (1998).
Morton-Firth, C. J., Shimizu, T. S. & Bray, D. A free-energy-based stochastic simulation of the Tar receptor complex. J. Mol. Biol. 286, 1059–1074 (1999).
Alon, U., Surette, M. G., Barkai, N. & Leibler, S. Robustness in bacterial chemotaxis. Nature 397, 168–171 (1999).
Meir, E., von Dassow, G., Munro, E. & Odell, G. M. Robustness, flexibility, and the role of lateral inhibition in the neurogenic network. Curr. Biol. 12, 778–786 (2002).
Little, J. W., Shepley, D. P. & Wert, D. W. Robustness of a gene regulatory circuit. EMBO J. 18, 4299–4307 (1999).
Csete, M. E. & Doyle, J. C. Reverse engineering of biological complexity. Science 295, 1664–1669 (2002).
Morohashi, M. et al. Robustness as a measure of plausibility in models of biochemical networks. J. Theor. Biol. 216, 19–30 (2002).
Hartwell, L. H., Hopfield, J. J., Leibler, S. & Murray, A. W. From molecular to modular cell biology. Nature 402, C47–C52 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, C., Wolf, D. & Arkin, A. Control, exploitation and tolerance of intracellular noise. Nature 420, 231–237 (2002). https://doi.org/10.1038/nature01258
Issue Date:
DOI: https://doi.org/10.1038/nature01258
This article is cited by
-
Patterning of the cell cortex by Rho GTPases
Nature Reviews Molecular Cell Biology (2024)
-
Flagellar dynamics reveal fluctuations and kinetic limit in the Escherichia coli chemotaxis network
Scientific Reports (2023)
-
Dynamic fluctuations in a bacterial metabolic network
Nature Communications (2023)
-
Frequency spectra and the color of cellular noise
Nature Communications (2022)
-
Dynamics and Sensitivity of Signaling Pathways
Current Pathobiology Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.