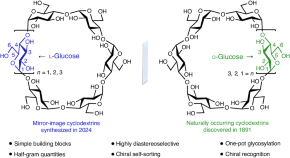
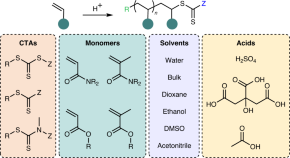
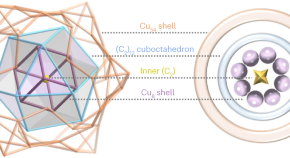
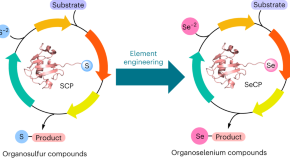
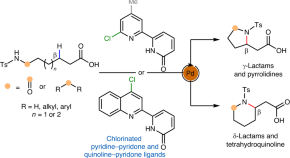
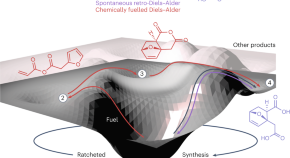
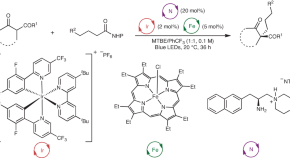
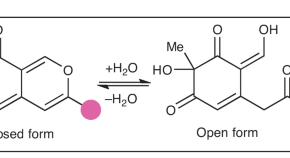
Featured

Advertisement
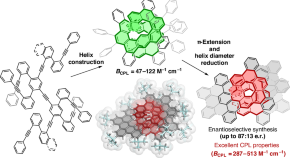
-
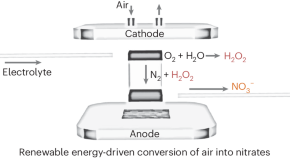
-
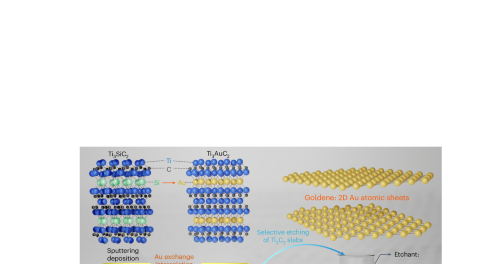
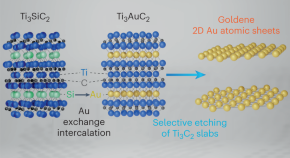
Synthesis of single-atom-thick gold layers
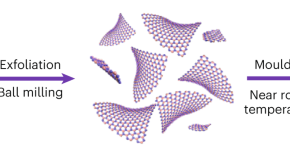
Monolayer gold could exhibit properties of benefit to various applications, but has been challenging to synthesize. Now, the exfoliation of two-dimensional single-atom-thick gold layers — termed goldene — is achieved through wet-chemically etching away Ti3C2 from Ti3AuC2, a nanolaminated MAX-phase. Goldene shows lattice contraction and an increase in the gold 4f binding energy compared with the bulk.
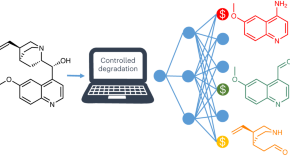
-
-