Abstract
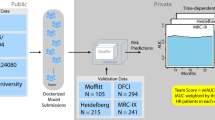
There is a strong need to better predict the survival of patients with newly diagnosed multiple myeloma (MM). As gene expression profiles (GEPs) reflect the biology of MM in individual patients, we built a prognostic signature based on GEPs. GEPs obtained from newly diagnosed MM patients included in the HOVON65/GMMG-HD4 trial (n=290) were used as training data. Using this set, a prognostic signature of 92 genes (EMC-92-gene signature) was generated by supervised principal component analysis combined with simulated annealing. Performance of the EMC-92-gene signature was confirmed in independent validation sets of newly diagnosed (total therapy (TT)2, n=351; TT3, n=142; MRC-IX, n=247) and relapsed patients (APEX, n=264). In all the sets, patients defined as high-risk by the EMC-92-gene signature show a clearly reduced overall survival (OS) with a hazard ratio (HR) of 3.40 (95% confidence interval (CI): 2.19–5.29) for the TT2 study, 5.23 (95% CI: 2.46–11.13) for the TT3 study, 2.38 (95% CI: 1.65–3.43) for the MRC-IX study and 3.01 (95% CI: 2.06–4.39) for the APEX study (P<0.0001 in all studies). In multivariate analyses this signature was proven to be independent of the currently used prognostic factors. The EMC-92-gene signature is better or comparable to previously published signatures. This signature contributes to risk assessment in clinical trials and could provide a tool for treatment choices in high-risk MM patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Avet-Loiseau H . Ultra high-risk myeloma. Am Soc Hematol Educ Program 2010, 489–493.
Palumbo A, Anderson K . Multiple myeloma. N Engl J Med 2011; 17: 1046–1060.
Munshi NC, Anderson KC, Bergsagel PL, Shaughnessy J, Palumbo A, Durie B et al. Consensus recommendations for risk stratification in multiple myeloma: report of the International Myeloma Workshop Consensus Panel 2. Blood 2011; 117: 4696–4700.
Neben K, Jauch A, Bertsch U, Heiss C, Hielscher T, Seckinger A et al. Combining information regarding chromosomal aberrations t(4;14) and del(17p13) with the International Staging System classification allows stratification of myeloma patients undergoing autologous stem cell transplantation. Haematologica 2010; 95: 1150–1157.
Bergsagel PL, Kuehl WM . Molecular pathogenesis and a consequent classification of multiple myeloma. J Clin Oncol 2005; 23: 6333–6338.
Zhan F, Huang Y, Colla S, Stewart JP, Hanamura I, Gupta S et al. The molecular classification of multiple myeloma. Blood 2006; 108: 2020–2028.
Broyl A, Hose D, Lokhorst H, de Knegt Y, Peeters J, Jauch A et al. Gene expression profiling for molecular classification of multiple myeloma in newly diagnosed patients. Blood 2010; 116: 2543–2553.
Chng WJ, Kuehl WM, Bergsagel PL, Fonseca R . Translocation t(4;14) retains prognostic significance even in the setting of high-risk molecular signature. Leukemia 2008; 22: 459–461.
Shaughnessy JD, Zhan F, Burington BE, Huang Y, Colla S, Hanamura I et al. A validated gene expression model of high-risk multiple myeloma is defined by deregulated expression of genes mapping to chromosome 1. Blood 2007; 109: 2276–2284.
Shaughnessy JD, Qu P, Usmani S, Heuck CJ, Zhang Q, Zhou Y et al. Pharmacogenomics of bortezomib test-dosing identifies hyperexpression of proteasome genes, especially PSMD4, as novel high-risk feature in myeloma treated with total therapy 3. Blood 2011; 118: 3512–3524.
Mulligan G, Mitsiades C, Bryant B, Zhan F, Chng WJ, Roels S et al. Gene expression profiling and correlation with outcome in clinical trials of the proteasome inhibitor bortezomib. Blood 2007; 109: 3177–3188.
Decaux O, Lode L, Magrangeas F, Charbonnel C, Gouraud W, Jezequel P et al. Prediction of survival in multiple myeloma based on gene expression profiles reveals cell cycle and chromosomal instability signatures in high-risk patients and hyperdiploid signatures in low-risk patients: a study of the Intergroupe Francophone du Myelome. J Clin Oncol 2008; 26: 4798–4805.
Dickens NJ, Walker BA, Leone PE, Johnson DC, Brito JL, Zeisig A et al. Homozygous deletion mapping in myeloma samples identifies genes and an expression signature relevant to pathogenesis and outcome. Clin Cancer Res 2010; 16: 1856–1864.
Hose D, Reme T, Hielscher T, Moreaux J, Messner T, Seckinger A et al. Proliferation is a central independent prognostic factor and target for personalized and risk-adapted treatment in multiple myeloma. Haematologica 2011; 96: 87–95.
Dispenzieri A, Rajkumar SV, Gertz MA, Fonseca R, Lacy MQ, Bergsagel PL et al. Treatment of newly diagnosed multiple myeloma based on Mayo Stratification of Myeloma and Risk-adapted Therapy (mSMART): consensus statement. Mayo Clin Proc 2007; 82: 323–341.
Sonneveld P, Schmidt-Wolf I, van der Holt B, Jarari Le, Bertsch U, Salwender H et al. HOVON-65/GMMG-HD4 randomized phase III trial comparing bortezomib, doxorubicin, dexamethasone (PAD) vs VAD followed by high-dose melphalan (HDM) and maintenance with bortezomib or thalidomide in patients with newly diagnosed multiple myeloma (MM). Blood 2010; 116: 40.
Barlogie B, Pineda-Roman M, van Rhee F, Haessler J, Anaissie E, Hollmig K et al. Thalidomide arm of Total Therapy 2 improves complete remission duration and survival in myeloma patients with metaphase cytogenetic abnormalities. Blood 2008; 112: 3115–3121.
Pineda-Roman M, Zangari M, Haessler J, Anaissie E, Tricot G, van Rhee F et al. Sustained complete remissions in multiple myeloma linked to bortezomib in total therapy 3: comparison with total therapy 2. Br J Haematol 2008; 140: 625–634.
Morgan GJ, Davies FE, Gregory WM, Bell SE, Szubert AJ, Navarro-Coy N et al. Thalidomide maintenance significantly improves progression-free survival (PFS) and overall survival (OS) of myeloma patients when effective relapse treatments are used: MRC myeloma IX results. Blood 2010; 116: 623–623.
Morgan GJ, Davies FE, Owen RG, Rawstron AC, Bell S, Cocks K et al. Thalidomide Combinations improve response rates; results from the MRC IX study. Blood 2007; 110: 3593–3593.
Jagannath S, Barlogie B, Berenson J, Siegel D, Irwin D, Richardson PG et al. A phase 2 study of two doses of bortezomib in relapsed or refractory myeloma. Br J Haematol 2004; 127: 165–172.
Richardson PG, Sonneveld P, Schuster MW, Irwin D, Stadtmauer EA, Facon T et al. Bortezomib or high-dose dexamethasone for relapsed multiple myeloma. N Engl J Med 2005; 352: 2487–2498.
Richardson PG, Barlogie B, Berenson J, Singhal S, Jagannath S, Irwin D et al. A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med 2003; 348: 2609–2617.
Gentleman R, Carey V, Bates D . Bioconductor: Open software development for computational biology and bioinformatics. Genome Biol 2004; 5: R80.
Johnson WE, Li C, Rabinovic A . Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 2007; 8: 118–127.
Therneau T, Lumley T . Survival: survival analysis, including penalised likelihood. R package version 236-2. 2010.
Bair E, Hastie T, Paul D, Tibshirani R . Prediction by supervised principal components. J Amer Statistical Assoc 2006; 101: 119–137.
Benjamini Y, Hochberg Y . Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc B Met 1995; 57: 289–300.
Arulampalam V, Kolosenko I, Hjortsberg L, Bjorklund AC, Grander D, Tamm KP . Activation of STAT1 is required for interferon-alpha-mediated cell death. Exp Cell Res 2011; 317: 9–19.
Chesi M, Nardini E, Brents LA, Schrock E, Ried T, Kuehl WM et al. Frequent translocation t(4;14)(p16.3;q32.3) in multiple myeloma is associated with increased expression and activating mutations of fibroblast growth factor receptor 3. Nat Genet 1997; 16: 260–264.
Hideshima T, Catley L, Raje N, Chauhan D, Podar K, Mitsiades C et al. Inhibition of Akt induces significant downregulation of survivin and cytotoxicity in human multiple myeloma cells. Br J Haematol 2007; 138: 783–791.
Mah N, Thelin A, Lu T, Nikolaus S, Kuhbacher T, Gurbuz Y et al. A comparison of oligonucleotide and cDNA-based microarray systems. Physiol Genomics 2004; 16: 361–370.
Trudel S, Ely S, Farooqi Y, Affer M, Robbiani DF, Chesi M et al. Inhibition of fibroblast growth factor receptor 3 induces differentiation and apoptosis in t(4;14) myeloma. Blood 2004; 103: 3521–3528.
Inoue J, Otsuki T, Hirasawa A, Imoto I, Matsuo Y, Shimizu S et al. Overexpression of PDZK1 within the 1q12-q22 amplicon is likely to be associated with drug-resistance phenotype in multiple myeloma. The Am J Pathol 2004; 165: 71–81.
Kumar SK, Mikhael JR, Buadi FK, Dingli D, Dispenzieri A, Fonseca R et al. Management of newly diagnosed symptomatic multiple myeloma: updated Mayo Stratification of Myeloma and Risk-Adapted Therapy (mSMART) consensus guidelines. Mayo Clin Proc 2009; 84: 1095–1110.
Acknowledgements
We would like to thank John Shaughnessy for kindly providing the data. Peter Valk and Mathijs Sanders are acknowledged for insightful discussions. Financial support: This work was supported by the Center for Translational Molecular Medicine (BioCHIP), a clinical research grant from the European Hematology Association and an EMCR Translational Research Grant from Skyline Diagnostics, Janssen and MSCNET.
Author contributions
RK, AB, MVD and PS designed the research. AB, YDK performed the research and collected the data. RK developed the classifier, AB, RK, MVD analyzed and interpreted the data and wrote the manuscript. MVV, EVB analyzed and interpreted the data and critically reviewed the paper. BVDH and LEJ performed data management and statistical analyses. GM provided the CEL files from the APEX data set and critically reviewed the paper. GM and WG provided CEL files and clinical data from the MRC-IX data set and critically reviewed the paper. HG is principal investigator of the performed research in the German part of the H65/GMMG-HD4 and critically reviewed the paper. HL organized the trial and critically reviewed the paper. PS organized the trial, is principal investigator of the performed research in the Dutch part of the H65/GMMG-HD4 and critically reviewed the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
GM has declared a financial interest in Millennium Pharmaceuticals Inc., whose product was an object of study in the HOVON 65/ GMMG-HD4. GM is currently employed by Millennium Pharmaceuticals Inc. HG has served on the advisory boards of Johnson & Johnson. HL is on the advisory Boards of Celgene and Genmab. EVB and MVV are employees of Skyline Diagnostics. PS is on the advisory boards of Skyline Diagnostics, Janssen and Celgene. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Kuiper, R., Broyl, A., de Knegt, Y. et al. A gene expression signature for high-risk multiple myeloma. Leukemia 26, 2406–2413 (2012). https://doi.org/10.1038/leu.2012.127
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2012.127
Keywords
This article is cited by
-
Redefining high risk multiple myeloma with an APOBEC/Inflammation-based classifier
Leukemia (2024)
-
Enhancing prognostic power in multiple myeloma using a plasma cell signature derived from single-cell RNA sequencing
Blood Cancer Journal (2024)
-
Molecular characterization stratifies VQ myeloma cells into two clusters with distinct risk signatures and drug responses
Oncogene (2023)
-
Alternative splicing in multiple myeloma is associated with the non-homologous end joining pathway
Blood Cancer Journal (2023)
-
Plasma cell neoplasms and related entities—evolution in diagnosis and classification
Virchows Archiv (2023)