Abstract
Background
Non-accidental head injury (NAI) is an inflicted injury usually on a child, often resulting in long-term neurological impairment and occasionally death. This study aimed to investigate the predictive values of acute findings, especially ocular, for long-term neurological outcomes.
Methods
Medical records including retinal images of all children who attended the local Children’s hospital with a diagnosis of NAI from over a period of 5 years were reviewed and data collected via the electronic patient record system. Patient demographics, injuries sustained, wide-field digital retinal images, visual acuity and sequalae, neurological function, and global function was noted. IBM SPSS software program was used for statistical analysis.
Results
Of the 38 patients (24 males, 14 females), 12 children died acutely from the head injury with the remaining 26 children available for long-term follow-up. A younger age of injury (P=0.004) was the only statistically significant predictor of good neurological outcome as compared with absence of macular retinoschisis, unilateral retinal haemorrhage, and unilateral subdural haemorrhage. Of the 38 children, 17 children had retinoschisis; 9 children with macular retinoschisis died acutely while 4 suffered a degree of developmental delay and only 4 were developmentally normal at the last follow-up. Long-term visual acuity data was available for 18 of the 26 survivors (range: NPL to Snellen 6/5). A statistical significance was noted between retinoschisis and worsened visual acuity (P<0.05).
Conclusions
Bilateral macular retinoschisis on acute presentation of NAI is associated with a seven-fold and unilateral with a four-fold increase in the development of a poor neurological outcome and eventual death. Conflicting to other studies, younger children presented better neurological outcomes.
Similar content being viewed by others
Introduction
Non-accidental head injury (NAI) is an inflicted traumatic brain injury upon a child, often resulting in debilitating long-term neurological deficits and at times death.1 NAI is otherwise also referred as shaken baby syndrome, whiplash-shaking injury, inflicted head trauma, and abusive head trauma.2 The neurological deficits, including learning disabilities, motor disabilities, cortical blindness, and epilepsy, occur in almost 60% of surviving children.1 The majority of these injuries are inflicted in the form of physical abuse on young defenseless children under the age of 2 years.3 Not surprisingly, these injuries are fatal in about 20% of these children.4
The incidence of NAI in the United Kingdom was 14.2/100 000 in the year 1998–1999, whereas a retrospective study published in 2005 reported an increased figure of 24.6/100 000.5, 6 In New Zealand, the incidence of NAI in children under the age of 2 years is estimated between 14.7 and 19.6/100 000 population.7 However, this incidence is 32.5–38.5/100 000 among the indigenous New Zealand Māori population.7 Interestingly, the incidence of mild cases of NAI is as high as 750 cases per 100 000 people per year in New Zealand.8
Ophthalmologists are often consulted in an acute setting of every case of suspected NAI in order to ascertain the presence of retinal haemorrhages. Retinal haemorrhages have a strong predictive value of NAI with odd ratios of 14.7, as computed by multilevel logistic regression analysis in a systematic review of six relevant studies.9 The characteristic findings are that retinal haemorrhages occur simultaneously in different layers of the retina and extend to the far peripheral retina. These retinal haemorrhages are often extensive and too numerous to count.10, 11 Reportedly, the presence of a macular retinoschisis (a dome-shaped haemorrhage within the retina at the macula) is highly indicative of NAI.12, 13, 14
Although the severity of retinal haemorrhages and the presence of retinoschisis is associated with the severity of acute neurological injury and death following NAI,15, 16 these studies have not yet examined the long-term prognostic value in survivors.
The current retrospective study aimed to investigate the correlation between the acute findings in NAI and long-term neurological outcome. This information might prove beneficial in forecasting victims’ prognoses for caregivers, society, and health-care providers.
Materials and methods
The location of the study was Blind and Low Vision Education Network New Zealand, Homai Campus, Auckland, New Zealand. The study was approved by our hospital Research Ethics Committee and followed the tenets of the Declaration of Helsinki.
The medical records of all patients diagnosed with NAI and retinal haemorrhages over a 5-year period (between May 2005 and May 2010) were retrospectively reviewed. Information collected from each medical record included date of birth, gender, ethnicity, injuries sustained, wide-field digital retinal images (RetCam-II; Clarity Medical Systems, Pleasanton, CA, USA), visual sequalae, neurological function, and global function. Visual sequalae included non-standardised data collection, that is, reduced visual fields, cortically blind, normal. Visual acuity data were available for only 18 children. Visual acuity was assessed in a variety of age and intellectual appropriate methods, including forced preferential looking, Lea symbols, and Sheridan–Gardiner and Snellen visual acuity. However, these were converted to Snellen equivalents in their medical notes with notation of the method of visual acuity testing to maintain consistency.
Self-described ethnicity was recorded according to the 2006 New Zealand Census data description with six main categories: European, Māori, Pacific peoples, Asian, Middle Eastern/Latin American/African, and others.
Injuries sustained by patients were compiled from medical records, discharge summaries, computed tomography scans, and radiology reports stored on the local hospital’s patient software, Concerto and Web1000 computer network system.
Wide-field digital retinal images of all patients were reviewed by an experienced paediatric ophthalmologist (SD) to determine the presence and extent of retinal hemorrhage and macular retinoschisis. Neurological function was measured with the Glasgow Outcome Scale—Extended Pediatric version (GOS-E Ped)17 instead of the gold standard Glasgow Outcome Scale—Extended due to its lack of validation in children. The GOS-E Ped was scored (by CFC) according to eight categories: 1=upper good recovery to 8=death (Table 1). Furthermore, neurological outcomes were defined as the child’s functional neurological status at their last follow-up appointment with their Paediatrician.
Statistical analysis was performed using the IBM SPSS for Windows (Version 21.0; IBM Corp., Armonk, NY, USA). Ordinal logistic regression was used to investigate correlation between variables and neurological outcomes. The explanatory variables used were age of injury, subdural haemorrhage (SDH)—unilateral or bilateral, retinal haemorrhage involving the fovea—unilateral or bilateral and the presence or absence of retinoschisis, and types of retinal haemorrhages. The ordinal outcome of GOS-E Ped score included reduced to normal (GOS-E Ped=1), some neurological impediment (GOS-E Ped=2–7), or dead (GOS-E Ped=8).
Results
Thirty-eight children with a clinical diagnosis of NAI were identified during the study period between May 2005 and May 2010. Twelve children died acutely from the injury (died on admission); the remaining 26 children were included in this cross-sectional study. The average age of injury presentation was 9 months (age range 1–37 months) with 24 male and 14 female children (Table 2) The average duration of follow-up in this group was 26 months (range: 11–62 months).
Children in this study were from five of the six ethnic groups in New Zealand. The highest representation was Māori (69%) followed by Caucasian (17%), Tongan (10%), Indian (2%), and African (2%). All 38 children had SDH and retinal hemorrhage at the time of initial assessment. Twenty-two (58%) children had bilateral SDH while bilateral retinal haemorrhage was noted in 29 (74%) children. Thirteen children presented with some degree of clinical neurological impediment while retinoschisis was noted in 17 children. Nine (53%) children with retinoschisis died acutely compared with 3 (14%) without retinoschisis.
Of the 26 surviving children, visual acuity data were available for 18 children. Eleven children had visual acuity better than Snellen visual acuity 6/12, nine without retinoschisis, and two with retinoschisis. The visual acuities were between Snellen 6/12 and 6/60 in three children, and of these three, two had retinoschisis. Four children had visual acuity worse than Snellen 6/60 in the better eye; however, retinoschisis was noted in three of them. Children with poor visual acuity were noted to have more diffuse retinal haemorrhage, retinal haemorrahge involving the fovea, and the presence of retinoschisis. Retinoschisis was significantly associated with worsened visual outcome at the final visit (P<0.01; Table 3).
Using ordinal logistic regression models, there was evidence that age of injury was related to outcome (P=0.004), with older children less likely to have good neurological outcome (Table 4). The estimation for the odds ratio of good neurological outcome was observed three times better in unilateral SDH and unilateral retinal haemorrhage compared with bilateral involvement (P=0.18 and P=0.26, respectively), though the results were statistically insignificant.
Unilateral retinoschisis was associated with a four-time increased chance of poor neurological outcome (GOS-E Ped=8) compared with a child without retinoschisis. Bilateral retinoschisis was noted to be a predictor of worse neurological outcome with possibly up to seven times the odds of poor long-term neurological outcome, in comparison with no retinoschisis (95% confidene interval 0.02–1.2). Although statistical analysis was inconclusive due to the large confidence interval, the estimated odds of poorer neurological outcome was greater in bilateral compared with unilateral retinoschisis and for bilateral compared with no retinoschisis.
Discussion
The current study investigated the age of injury, laterality of SDH, laterality of retinal hemorrhage, presence of preretinal haemorhages, and presence of macular retinoschisis for prognostic determinant of long-term neurological outcome and visual outcome. To our knowledge, this is the first study to investigate association between retinal haemorrhages and retinoschisis in NAI to predict ‘cause and effect’ of these injuries to neurological outcome and visual outcome.
Of the four factors studied in this study, only age of injury at presentation corresponded with positive long-term neurological outcome; that is, younger age at the time of acute admission is expected to achieve better neurological outcome. Several studies have reported contradictory findings with younger age leading to higher rates of mortality, morbidity, or cognitive impairment.18, 19 Notably, a report from the New Zealand Children’s Commissioner stated that injury at a later age corresponds with good neurological outcome or poorer outcome at younger age.20 Interestingly, some studies have reported no correlation between age of injury and global outcome.21, 22 Clearly, there is no consensus among clinicians or clinical researchers with regards to the prediction of global outcomes and NAI presentation age. The studies that report on these types of injuries have an age range of about 1–36 months at presentation similar to the current study with an age range of 3–37 months.21, 22 Arguably, better repair mechanism and good brain plasticity in younger age group potentially leads to positive neurological outcomes. Furthermore, severity of injury at the age of admission possibly has an important role in determining long-term neurological outcomes. The younger children in the current study potentially suffered less severe injuries. Further research on NAI and its comparative outcomes in infants, older children, and adolescents might help in explaining the importance of age at the time of initial injury presentation.22
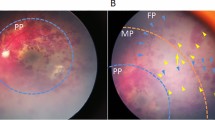
The current study utilised the GOS-E Ped to assess neurological function.17 GOS-E Ped provides sensitive results across the injury spectrum along with its ability to detect subtle changes in functional status as compared with standard GOS that can significantly underestimate the impact of brain injury in young children.17, 22 According to GOS-E Ped, the presence of macular retinoschisis was associated with an approximate seven-fold increase in poor long-term neurological outcome and associated with higher rate of mortality in the present study. Arguably, macular retinoschisis is one of the specific ocular signs of NAI and potentially be a crucial diagnostic feature.23 The ocular autopsy and histopathology of 118 cases noted the presence of macular retinoschisis in about 23% of deceased children.24 Furthermore, a smaller published case series of 10 children has reported significant association of macular retinoschisis with a fatal outcome15 Figure 1.
SDH often co-exists with retinal haemorrhages and the two are key features of NAI.25 Although, the severity of injury can be associated with findings of bilateral SDH and retinal haemorrhage, the current study did not show statistical significance. It is generally accepted that the more severe the intracranial bleed and the more extensive the retinal haemorrhages, the more severe the mechanism of injury.26 However, current study only accounted for the presence of SDH, retinal haemorrhages, and retinoschisis, rather than the extent of injury that could have resulted in statistically insignificant results. The presence of bilateral SDH and retinal haemorrhage corresponds to a three-fold increase of poor long-term neurological outcome as compared with unilateral findings. NAI children with SDH carry a poor prognosis, with three-quarters die or live with profound disability.3
Reportedly, the severity of retinal haemorrhage is significantly associated with acute neurological injury.27 A systematic review of about thousand cases that included meta-analysis of five studies reported a strong correlation of retinal haemorrhages and NAI (or abusive head injury) with an odds ratio of 14.7 and 91% probability.26 Similar to the current study, bilateral involvement also had significant association.26 Typically, macular retinoschisis, retinal haemorrhages, and acute encephalopathy are noted in NAI.13, 14
An incidental finding of this study was a two-fold increase in prevalence of NAI in Māori children compared with the other ethnic groups in New Zealand. This unfortunate outcome is supported by other reports of higher incidence of childhood visual impairment and non-accidental injuries in Māori children.28
This study has limitations due to the nature of being a retrospective study. Although the limited sample size is a limitation of this study where results have failed to reach statistical significance, a trend is noted with worse long-term neurological function when SDHs and retinal haemorrhages were bilateral and if retinoschisis was present. Collectively, these signs offer prognostic factors of long-term neurological outcome.
Conclusion
In conclusion, this study reports on the presence of retinoschisis in the event of NAI to have an approximate seven-fold increase in the risk of poor long-term neurological outcome, including death. The findings of a younger age of injury corresponding with better neurological outcome in this cohort differs from other published reports. Good neuroplasticity in children allows better neurological outcome at a later stage is a possible explanation for this unexpected result.

References
Barlow K, Thompson E, Johnson D, Minns RA . The neurological outcome of non-accidental head injury. Dev Neurorehabil 2004; 7 (3): 195–203.
David TJ . Shaken baby (shaken impact) syndrome: non-accidental head injury in infancy. J R Soc Med 1999; 92 (11): 556.
Jayawant S, Rawlinson A, Gibbon F, Price J, Schulte J, Sharples P et al. Subdural haemorrhages in infants: population based study. BMJ 1998; 317 (7172): 1558–1561.
Minns RA, Jones PA, Mok JYQ . Incidence and demography of non-accidental head injury in southeast Scotland from a national database. Am J Prev Med 2008; 34 (4): S126–S133.
Barlow KM, Minns RA . Annual incidence of shaken impact syndrome in young children. Lancet 2000; 356 (9241): 1271–1272.
Hobbs C, Childs AM, Wynne J, Livingston J, Seal A . Subdural haematoma and effusion in infancy: an epidemiological study. Arch Dis Child 2005; 90 (9): 952–955.
Kelly P, Farrant B . Shaken baby syndrome in New Zealand, 2000-2002. J Paediatr Child Health 2008; 44 (3): 99–107.
Feigin VL, Theadom A, Barker-Collo S, Starkey NJ, McPherson K, Kahan M et al. Incidence of traumatic brain injury in New Zealand: a population-based study. Lancet Neurol 2013; 12 (1): 53–64.
Maguire SA, Watts PO, Shaw AD, Holden S, Taylor RH, Watkins WJ et al. Retinal haemorrhages and related findings in abusive and non-abusive head trauma: a systematic review. Eye (Lond) 2013; 27 (1): 28–36.
Vincent AL, Kelly P . Retinal haemorrhages in inflicted traumatic brain injury: the ophthalmologist in court. Clin Exp Ophthalmol 2010; 38 (5): 521–532.
Bechtel KSK, Leventhal JM, Ogle E, Teague B, Lavietes S, Banyas B et al. Characteristics that distinguish accidental from abusive injury in hospitalized young children with head trauma. Pediatrics 2004; 114 (1): 165–168.
Lantz PE, Sinal SH, Stanton CA, Weaver RG Jr . Perimacular retinal folds from childhood head trauma. BMJ 2004; 328 (7442): 754–756.
Morad Y, Wygnansky-Jaffe T, Levin AV . Retinal haemorrhage in abusive head trauma. Clin Exp Ophthalmol 2010; 38 (5): 514–520.
Watts P, Obi E . Retinal folds and retinoschisis in accidental and non-accidental head injury. Eye 2008; 22 (12): 1514–1516.
Mills M . Funduscopic lesions associated with mortality in Shaken baby syndrome. J AAPOS 1998; 2: 67–71.
Wilkinson WS, Han Dennis P, Rappley Marsha D, Owings Clyde L . Retinal haemorrhage predicts neurologic injury in shaken baby syndrome. Arch Ophthalmol 1989; 107: 1472–1474.
Beers SR, Wisniewski SR, Garcia-Filion P, Tian Y, Hahner T, Berger RP et al. Validity of a pediatric version of the Glasgow Outcome Scale–Extended. J Neurotrauma 2012; 29 (6): 1126–1139.
Anderson V, Catroppa C, Morse S, Haritou F, Rosenfeld J . Recovery of intellectual ability following traumatic brain injury in childhood: impact of injury severity and age at injury. Pediatr Neurosurg 2000; 32 (6): 282–290.
Filley CM, Cranberg LD, Alexander MP, Hart EJ . Neurobehavioral outcome after closed head injury in childhood and adolescence. Arch Neurol 1987; 44 (2): 194–198.
Duncanson M, Smith DA, Davies E Death and Serious Injury from Assault of Children Under 5 years in Aotearoa New Zealand: A Review of International Literature and Recent Findings [Internet]. Office of the Chilren’s Commisioner: Wellington, New Zealand 2009.
Barlow KM, Thomson E, Johnson D, Minns RA . Late neurologic and cognitive sequelae of inflicted traumatic brain injury in infancy. Pediatrics 2005; 116 (2): 174–185.
Prasad MR, Ewing-Cobbs L, Swank PR, Kramer L . Predictors of outcome following traumatic brain injury in young children. Pediatr Neurosurg 2002; 36 (2): 64–74.
Massicotte SJ, Folberg R, Torczynski E, Gilliland MGF, Luckenbach MW . Vitreoretinal traction and perimacular retinal folds in the eyes of deliberately traumatized children. Ophthalmology 1991; 98 (7): 1124–1127.
Emerson MV, Jakobs E, Green WR . Ocular autopsy and histopathologic features of child abuse. Ophthalmology 2007; 114 (7): 1384–1394.
Mian M, Shah J, Dalpiaz A, Schwamb R, Miao Y, Warren K et al. Shaken baby syndrome: a review. Fetal Pediatr Pathol 2015; 34 (3): 169–175.
Maguire SA, Watts PO, Shaw AD, Holden S, Taylor RH, Watkins WJ et al. Retinal haemorrhages and related findings in abusive and non-abusive head trauma: a systematic review. Eye 2013; 27 (1): 28–36.
Wilkinson WS, Han DP, Rappley MD, Owings CL . Retinal hemorrhage predicts neurologic injury in the shaken baby syndrome. Arch Ophthalmol 1989; 107 (10): 1472–1474.
Chong CF DS . Cross-sectional study on prevalence, causes and avoidable causes of visual impairment in Māori children. New Zeal Med J 2013; 126: 1379.
Acknowledgements
We thank The University of Auckland and Blind and Low Vision Education Network New Zealand, Homai Campus, Auckland, New Zealand.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chong, C., Misra, S., Escardo-Paton, J. et al. Predictors of long-term neurological outcomes in non-accidental head injury. Eye 32, 608–614 (2018). https://doi.org/10.1038/eye.2017.266
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2017.266