Abstract
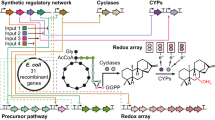
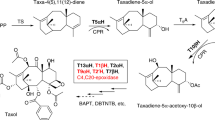
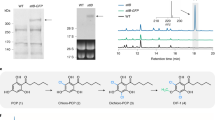
Terpenoids are a highly diverse class of natural products that have historically provided a rich source for discovery of pharmacologically active small molecules1, such as paclitaxel (Taxol) and artemisinin. Unfortunately, these secondary metabolites are typically produced in low abundance in their host organism, and their isolation consequently suffers from low yields and high consumption of natural resources. Furthermore, chemical synthesis of terpenoids can also be difficult to scale for industrial production. For these reasons, an attractive alternative strategy is to engineer metabolic pathways for production of pharmaceuticals or their precursors in a microbial host such as Escherichia coli. A key step is developing methods to carry out cytochrome P450 (P450)-based oxidation chemistry in vivo. Toward this goal, we have assembled two heterologous pathways for the biosynthesis of plant-derived terpenoid natural products, and we present the first examples of in vivo production of functionalized terpenoids in E. coli at high titer using native plant P450s.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Conolly, J.D. & Hill, R.A. Dictionary of Terpenoids (Chapman & Hall, London, 1991).
Durst, F. & Nelson, D.R. Diversity and evolution of plant P450 and P450-reductases. Drug Metabol. Drug Interact. 12, 189–206 (1995).
Schuler, M.A. & Werck-Reichhart, D. Functional genomics of P450s. Annu. Rev. Plant Biol. 54, 629–667 (2003).
Chau, M., Jennewein, S., Walker, K. & Croteau, R. Taxol biosynthesis: molecular cloning and characterization of a cytochrome P450 taxoid 7β-hydroxylase. Chem. Biol. 11, 663–672 (2004).
Ortiz de Montellano, P. Cytochrome P450: Structure, Mechanism, and Biochemistry (Springer, New York, 2004).
Sono, M., Roach, M.P., Coulter, E.D. & Dawson, J.H. Heme-containing oxygenases. Chem. Rev. 96, 2841–2887 (1996).
Wuts, P.G.M. Semisynthesis of taxol. Curr. Opin. Drug Discov. Devel. 1, 329–337 (1998).
Roth, R.J. & Acton, N. A simple conversion of artemisinic acid into artemisinin. J. Nat. Prod. 52, 1183–1185 (1989).
Wittstock, U. & Halkier, B.A. Cytochrome P450 CYP79A2 from Arabidopsis thaliana L. catalyzes the conversion of L-phenylalanine to phenylacetaldoxime in the biosynthesis of benzylglucosinolate. J. Biol. Chem. 275, 14659–14666 (2000).
Hansen, C.H. et al. Cytochrome P450 CYP79F1 from Arabidopsis catalyzes the conversion of dihomomethionine and trihomomethionine to the corresponding aldoximes in the biosynthesis of aliphatic glucosinolates. J. Biol. Chem. 276, 24790–24796 (2001).
Naur, P. et al. CYP79B1 from Sinapis alba converts tryptophan to indole-3-acetaldoxime. Arch. Biochem. Biophys. 409, 235–241 (2003).
Haudenschild, C., Schalk, M., Karp, F. & Croteau, R. Functional expression of regiospecific cytochrome P450 limonene hydroxylases from mint (Mentha spp.) in Escherichia coli and Saccharomyces cerevisiae. Arch. Biochem. Biophys. 379, 127–136 (2000).
Bertea, C.M., Schalk, M., Karp, F., Maffei, M. & Croteau, R. Demonstration that menthofuran synthase of mint (Mentha) is a cytochrome P450 monooxygenase: cloning, functional expression, and characterization of the responsible gene. Arch. Biochem. Biophys. 390, 279–286 (2001).
Schroder, G. et al. Light-induced cytochrome P450-dependent enzyme in indole alkaloid biosynthesis: tabersonine 16-hydroxylase. FEBS Lett. 458, 97–102 (1999).
Irmler, S. et al. Indole alkaloid biosynthesis in Catharanthus roseus: new enzyme activities and identification of cytochrome P450 CYP72A1 as secologanin synthase. Plant J. 24, 797–804 (2000).
Leonard, E., Yan, Y. & Mattheos, M.A.G. Functional expression of a P450 flavonoid hydroxylase for the biosynthesis of plant-specific hydroxylated flavonols in Escherichia coli. Metab. Eng. 8, 172–181 (2006).
Carter, O.A., Peters, R.J. & Croteau, R. Monoterpene biosynthesis pathway construction in Escherichia coli. Phytochemistry 64, 425–433 (2003).
Martin, V.J., Pitera, D.J., Withers, S.T., Newman, J.D. & Keasling, J.D. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nat. Biotechnol. 21, 796–802 (2003).
Newman, J.D. et al. High-level production of amorpha-4,11-diene in a two-phase partitioning bioreactor of metabolically engineered Escherichia coli. Biotechnol. Bioeng. 95, 684–691 (2006).
Luo, P., Wang, Y.-H., Wang, G.-D., Essenberg, M. & Chen, X.-Y. Molecular cloning and functional identification of (+)-δ-cadinene-8-hydroxylase, a cytochrome P450 monooxygenase (CYP706B1) of cotton sesquiterpene biosynthesis. Plant J. 28, 95–104 (2001).
Ro, D.K. et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940–943 (2006).
Sutter, T.R., Sanglard, D. & Loper, J.C. Isolation and characterization of the alkane-inducible NADPH-cytochrome P-450 oxidoreductase gene from Candida tropicalis. Identification of invariant residues within similar amino acid sequences of divergent flavoproteins. J. Biol. Chem. 265, 16428–16436 (1990).
Chen, X.Y., Chen, Y., Heinstein, P. & Davisson, V.J. Cloning, expression, and characterization of (+)-delta-cadinene synthase: a catalyst for cotton phytoalexin biosynthesis. Arch. Biochem. Biophys. 324, 255–266 (1995).
Craft, D.L., Madduri, K.M., Eshoo, M. & Wilson, C.R. Identification and characterization of the CYP52 family of Candida tropicalis ATCC 20336, important for the conversion of fatty acids and alkanes to α,ω-dicarboxylic acids. Appl. Environ. Microbiol. 69, 5983–5991 (2003).
Barnes, H.J., Arlotto, M.P. & Waterman, M.R. Expression and enzymatic activity of recombinant cytochrome P450 17-α-hydroxylase in Escherichia coli. Proc. Natl. Acad. Sci. USA 88, 5597–5601 (1991).
Schafmeister, C.E., Miercke, L.J.W. & Stroud, R.M. Structure at 2.5 Å of a designed peptide that maintains solubility of membrane proteins. Science 262, 734–738 (1993).
Sueyoshi, T., Park, L.J., Moore, R., Juvonen, R.O. & Negishi, M. Molecular engineering of microsomal P450 2α-4 to a stable, water-soluble enzyme. Arch. Biochem. Biophys. 322, 265–271 (1995).
Schoch, G.A., Attias, R., Belghazi, M., Dansette, P.M. & Werck-Reichhart, D. Engineering of a water-soluble plant cytochrome P450, CYP73A1, and NMR-based orientation of natural and alternate substrates in the active site. Plant Physiol. 133, 1198–1208 (2003).
Roosild, T.P. et al. NMR structure of Mistic, a membrane-integrating protein for membrane protein expression. Science 307, 1317–1321 (2005).
Acknowledgements
We thank X.-Y. Chen (Shanghai Institutes for Biological Sciences) for the native CAS and CAH genes, L. Anthony (Amyris Biotechnologies) for the pAM92 plasmid, K. Fisher (Amyris Biotechnologies) for authentic standards, P. Ortiz de Montellano (University of California, San Francisco) for the pCWori plasmid and J. Minshull (DNA 2.0), Y. Yoshikuni, D. Pitera, S. Withers and E. Paradise for helpful discussions. M.C.Y. Chang acknowledges a postdoctoral fellowship from the Jane Coffin Childs Memorial Fund. Funding was provided by the Institute for OneWorld Health through generous support by the Bill and Melinda Gates Foundation.
Author information
Authors and Affiliations
Contributions
M.C.Y.C. carried out experiments with assistance from R.A.E. and W.T. M.C.Y.C. designed the experiments with input from D.-K.R. M.C.Y.C. and J.D.K. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.D.K. owns shares of Amyris Biotechnologies, a company that is currently using the technology described here to produce the antimalarial drug artemisinin. Neither Amyris nor the University of California will make any profit (or royalties) from the sale of artemisinin in the developing world.
Supplementary information
Rights and permissions
About this article
Cite this article
Chang, M., Eachus, R., Trieu, W. et al. Engineering Escherichia coli for production of functionalized terpenoids using plant P450s. Nat Chem Biol 3, 274–277 (2007). https://doi.org/10.1038/nchembio875
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio875
This article is cited by
-
Hydroxylases involved in terpenoid biosynthesis: a review
Bioresources and Bioprocessing (2023)
-
Design of a redox-proficient Escherichia coli for screening terpenoids and modifying cytochrome P450s
Nature Catalysis (2023)
-
Construction of an Escherichia coli cell factory to synthesize taxadien-5α-ol, the key precursor of anti-cancer drug paclitaxel
Bioresources and Bioprocessing (2022)
-
Engineering medicinal plant-derived CYPs: a promising strategy for production of high-valued secondary metabolites
Planta (2022)
-
Amorpha-4,11-diene synthase: a key enzyme in artemisinin biosynthesis and engineering
aBIOTECH (2021)