Abstract
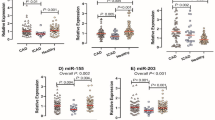
MicroRNAs (miRNAs) are a novel class of small, non-coding RNAs that play a significant role in both inflammatory and cardiovascular diseases. Immune cells, especially T helper (Th) cells, are critical in the development of atherosclerosis and the onset of acute coronary syndrome (ACS). To assess whether inflammation-related miRNAs (such as miR-155, 146a, 21, 125a-5p, 125b, 31) are involved in the imbalance of Th cell subsets in patients with ACS, we measured the expression of related miRNAs in patients with acute myocardial infarction (AMI), unstable angina (UA), stable angina (SA) and chest pain syndrome (CPS); analyzed the relationship between miRNA expression and the frequency of Th cell subsets; and observed the co-expression of miR-155 and IL-17A in peripheral blood mononuclear cells (PBMCs) of patients with ACS. The results showed that the expression of miR-155 in the PBMCs of patients with ACS was decreased by approximately 60%, while the expression of both miR-21 and miR-146a was increased by approximately twofold. The expression patterns of miRNAs in plasma correlated with those in PBMCs, except for miR-21, which was increased by approximately sixfold in the AMI group and showed no significant difference between the UA group and the CPS group. We also found that the expression of miR-155 inversely correlated with the frequency of Th17 cells (r=−0.896, P<0.01) and that miR-155 was co-expressed with IL-17A in patients with ACS. In conclusion, our study revealed the expression patterns of inflammation-related miRNAs in patients with ACS and found that miR-155 may be associated with Th17 cell differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zhang B, Pan X, Wang Q, Cobb GP, Anderson TA . Computational identification of microRNAs and their targets. Comput Biol Chem 2006; 30: 395–407.
Urbich C, Kuehbacher A, Dimmeler S . Role of microRNAs in vascular diseases, inflammation, and angiogenesis. Cardiovasc Res 2008; 79: 581–588.
Cobb BS, Nesterova TB, Thompson E, Hertweck A, O'Connor E, Godwin J et al. T cell lineage choice and differentiation in the absence of the RNase III enzyme Dicer. J Exp Med 2005; 201: 1367–1373.
O'Carroll D, Mecklenbrauker I, Das PP, Santana A, Koenig U, Enright AJ et al. A Slicer-independent role for Argonaute 2 in hematopoiesis and the microRNA pathway. Genes Dev 2007; 21: 1999–2004.
Lindsay MA . microRNAs and the immune response. Trends Immunol 2008; 29: 343–351.
Stanczyk J, Pedrioli DM, Brentano F, Sanchez-Pernaute O, Kolling C, Gay RE et al. Altered expression of MicroRNA in synovial fibroblasts and synovial tissue in rheumatoid arthritis. Arthritis Rheum 2008; 58: 1001–1009.
Bostjancic E, Glavac D . Importance of microRNAs in skin morphogenesis and diseases. Acta Dermatovenerol Alp Panonica Adriat 2008; 17: 95–102.
Laurat E, Poirier B, Tupin E, Caligiuri G, Hansson GK, Bariéty J et al. In vivo downregulation of T helper cell 1 immune responses reduces atherogenesis in apolipoprotein E-knockout mice. Circulation 2001; 104: 197–202.
Cheng X, Yu X, Ding YJ, Fu QQ, Xie JJ, Tang TT et al. The Th17/Treg imbalance in patients with acute coronary syndrome. Clin Immunol 2008; 127: 89–97.
Madhur MS, Funt SA, Li L, Vinh A, Chen W, Lob HE et al. Role of interleukin 17 in inflammation, atherosclerosis, and vascular function in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol 2011; 31: 01–09.
Taleb S, Tedgui A, Mallat Z . Adaptive T cell immune responses and atherogenesis. Curr Opin Pharmacol 2010; 10: 197–202.
Rodriguez A, Vigorito E, Clare S, Warren MV, Couttet P, Soond DR et al. Requirement of bic/microRNA-155 for normal immune function. Science 2007; 316: 608–611.
Lu LF, Boldin MP, Chaudhry A, Lin LL, Taganov KD, Hanada T et al. Function of miR-146a in controlling Treg cell-mediated regulation of Th1 responses. Cell 2010; 142: 914–929.
Taganov KD, Boldin MP, Chang KJ, Baltimore D . NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci USA 2006; 103: 12481–12486.
Sheedy FJ, Palsson-McDermott E, Hennessy EJ, Martin C, O'Leary JJ, Ruan Q et al. Negative regulation of TLR4 via targeting of the proinflammatory tumor suppressor PDCD4 by the microRNA miR-21. Nat Immunol 2010; 11: 141–147.
Cheng Y, Liu X, Zhang S, Lin Y, Yang J, Zhang C . MicroRNA-21 protects against the H2O2-induced injury on cardiac myocytes via its target gene PDCD4. J Mol Cell Cardiol 2009; 47: 5–14.
Stahl HF, Fauti T, Ullrich N, Bopp T, Kubach J, Rust W et al. miR-155 inhibition sensitizes CD4+ Th cells for TREG mediated suppression. PLoS One 2009; 4: e7158.
Banerjee A, Schambach F, DeJong CS, Hammond SM, Reiner SL . Micro-RNA-155 inhibits IFN-gamma signaling in CD4+ T cells. Eur J Immunol 2010; 40: 225–231.
Lu LF, Thai TH, Calado DP, Chaudhry A, Kubo M, Tanaka K et al. Foxp3-dependent microRNA155 confers competitive fitness to regulatory T cells by targeting SOCS1 protein. Immunity 2009; 30: 80–91.
Huang RS, Hu GQ, Lin B, Lin ZY, Sun CC . MicroRNA-155 silencing enhances inflammatory response and lipid uptake in oxidized low-density lipoprotein-stimulated human THP-1 macrophages. J Investig Med 2010; 58: 961–967.
Fichtlscherer S, de Rosa S, Fox H, Schwietz T, Fischer A, Liebetrau C et al. Circulating MicroRNAs in patients with coronary artery disease. Circ Res 2010; 107: 677–684.
Tili E, Croce CM, Michaille JJ . miR-155: on the crosstalk between inflammation and cancer. Int Rev Immunol 2009; 28: 264–284.
McCoy CE, Sheedy FJ, Qualls JE, Doyle SL, Quinn SR, Murray PJ et al. IL-10 inhibits miR-155 induction by Toll-like receptors. J Biol Chem 2010; 285: 20492–20498.
Methe H, Brunner S, Wiegand D, Nabauer M, Koglin J, Edelman ER . Enhanced T-helper-1 lymphocyte activation patterns in acute coronary syndromes. J Am Coll Cardiol 2005; 45: 1939–1945.
Mor A, Luboshits G, Planer D, Keren G, George J . Altered status of CD4+CD25+ regulatory T cells in patients with acute coronary syndromes. Eur Heart J 2006; 27: 2530–2537.
Libby P, Sukhova G, Lee RT, Galis ZS . Cytokines regulate vascular functions related to stability of the atherosclerotic plaque. J Cardiovasc Pharmacol 1995; 25: S9–S12.
Rabbani R, Topol EJ . Strategies to achieve coronary arterial plaque stabilization. Cardiovasc Res 1999; 41: 402–417.
Smith E, Prasad KM, Butcher M, Dobrian A, Kolls JK, Ley K et al. Blockade of interleukin-17A results in reduced atherosclerosis in apolipoprotein E-deficient mice. Circulation 2010; 121: 1746–1755.
Eid RE, Rao DA, Zhou J, Lo SF, Ranjbaran H, Gallo A et al. Interleukin-17 and interferon-gamma are produced concomitantly by human coronary artery-infiltrating T cells and act synergistically on vascular smooth muscle cells. Circulation 2009; 119: 1424–1432.
Xie JJ, Wang J, Tang TT, Chen J, Gao XL, Yuan J et al. The Th17/Treg functional imbalance during atherogenesis in ApoE−/− mice. Cytokine 2010; 49: 185–193.
Thai TH, Calado DP, Casola S, Ansel KM, Xiao C, Xue Y et al. Regulation of the germinal center response by MicroRNA-155. Science 2007; 316: 604–608.
O'Connell RM, Kahn D, Gibson WS, Round JL, Scholz RL, Chaudhuri AA et al. MicroRNA-155 promotes autoimmune inflammation by enhancing inflammatory T cell development. Immunity 2010; 33: 607–619.
Kohlhaas S, Garden OA, Scudamore C, Turner M, Okkenhaug K, Vigorito E . Cutting edge: the Foxp3 target miR-155 contributes to the development of regulatory T cells. J Immunol 2009; 182: 2578–2582.
Louafi F, Martinez-Nunez RT, Sanchez-Elsner T . Microrna-155 (miR-155) targets SMAD2 and modulates the response of macrophages to transforming growth factor-β (TGF-β). J Biol Chem 2010; 285: 41328–41336.
Jiang S, Zhang HW, Lu MH, He XH, Li Y, Gu H et al. MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Cancer Res 2010; 70: 3119–3127.
Martinez GJ, Zhang Z, Reynolds JM, Tanaka S, Chung Y, Liu T et al. Smad2 positively regulates the generation of Th17 cells. J Biol Chem 2010; 285: 29039–29043.
Yoshikawa H, Matsubara K, Qian GS, Jackson P, Groopman JD, Manning JE et al. SOCS-1, a negative regulator of the JAK/STAT pathway, is silenced by methylation in human hepatocellular carcinoma and shows growth-suppression activity. Nat Genet 2001; 28: 29–35.
Acknowledgements
The work described in this article was supported by the National Natural Science Foundation of China (no. 81000085) and the Doctor and Novo-teacher Foundation of Chinese National Ministry of Education (no. 20090142120049).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yao, R., Ma, Y., Du, Y. et al. The altered expression of inflammation-related microRNAs with microRNA-155 expression correlates with Th17 differentiation in patients with acute coronary syndrome. Cell Mol Immunol 8, 486–495 (2011). https://doi.org/10.1038/cmi.2011.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2011.22
Keywords
This article is cited by
-
Platelet miRNAs: differential expression in coronary artery disease and associations with course of left ventricular systolic function
BMC Cardiovascular Disorders (2023)
-
A miRNA-Based Blood and Mucosal Approach for Detecting and Monitoring Celiac Disease
Digestive Diseases and Sciences (2020)
-
Platelet Function in Cardiovascular Disease: Activation of Molecules and Activation by Molecules
Cardiovascular Toxicology (2020)
-
Altered microRNA Expression in Peripheral Blood Mononuclear Cells from Young Patients with Schizophrenia
Journal of Molecular Neuroscience (2015)
-
Decidual stromal cells recruit Th17 cells into decidua to promote proliferation and invasion of human trophoblast cells by secreting IL-17
Cellular & Molecular Immunology (2014)