Abstract
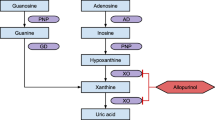
Many patients fail to achieve the recommended serum urate (SU) target (<6 mgdl−1) with allopurinol. The aim of our study was to examine the association of ABCG2 with SU target in response to standard doses of allopurinol using a cohort with confirmed adherence. Good response was defined as SU<6 mgdl−1 on allopurinol ⩽300 mgd−1 and poor response as SU⩾6 mgdl−1 despite allopurinol >300 mgd−1. Adherence was confirmed by oxypurinol concentrations. ABCG2 genotyping was performed using pre-designed single nucleotide polymorphism (SNP) TaqMan assays. Of 264 patients, 120 were good responders, 68 were poor responders and 76 were either non-adherent or could not be classified. The minor allele of ABCG2 SNP rs2231142 conferred a significantly increased risk of poor response to allopurinol (odds ratio=2.71 (1.70–4.48), P=6.0 × 10−5). This association remained significant after adjustment for age, sex, body mass index, ethnicity, estimated glomerular filtration rate, diuretic use and SU off urate-lowering therapy. ABCG2 rs2231142 predicts poor response to allopurinol, as defined by SU⩾6 mgdl−1 despite allopurinol >300 mgd−1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Khanna D, Fitzgerald J, Khanna P, Sangmee B, Singh M, Neogi T et al. 2012 American College of Rheumatology Guidelines for the Management of Gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricaemia. Arthritis Care Res 2012; 64: 1431–1446.
Stamp L, Merriman T, Barclay M, Singh J, Roberts R, Wright D et al. Inadequate response to allopurinol in the treatment of gout – defining the population and potential causes. Semin Arthritis Rheum 2014; 44: 170–174.
Graham G, Kannangara D, Stocker S, Portek I, Pile K, Indraratna P et al. Understanding the dose-response relationship of allopurinol: predicting the optimal dosage. Br J Clin Pharmacol 2013; 76: 932–938.
Roberts R, Stamp L . Pharmacogenetic considerations in the treatment of gout. Pharmacogenetics 2015; 16: 619–629.
Saito Y, Stamp L, Caudle K, Hershfield M, McDonagh M, Callaghan J et al. Clinical pharmacogenetics implementation consortium (CPIC) guidelines for human leukocyte antigen B (HLA-B) genotype and allopurinol dosing: 2015 update. Clin Pharm Ther 2016; 99: 36–37.
Wen C, Yee S, Liang X, Hoffmann T, Kvall M, Banda Y et al. Genome-wide association study identifies ABCG2 (BRCP) as an allopurinol transporter and a determinant of drug response. Clin Pharm Ther 2015; 97: 518–525.
Wallace S, Robinson H, Masi A, Decker J, McCarty D, Yu T . Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum 1977; 20: 895–900.
Stamp L, O’Donnell J, Zhang M, James J, Frampton C, Barclay M et al. Using allopurinol above the dose based on creatinine clearance is effective and safe in chronic gout, including in those with renal impairment. Arthritis Rheum 2011; 63: 412–421.
Team RC. A language and environment for statistical computing R Foundation for Statistical Computing2013. Available from http://www.R-project.org/.
Becker M, Fitz-Patrick D, Choi H, Dalbeth N, Storgarde C, Cravetsf M et al. An open-label, 6-month study of allopurinol safety in gout: The LASSO study. Semin Arthritis Rheum 2015; 45: 174–183.
Acknowledgements
We thank the patients for generously giving their time to participate in this study. Financial support for this study was provided by project and programme grants from the Health Research Council of New Zealand.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dalbeth, Stamp, Merriman and Roberts report grants from the Health Research Council of New Zealand, during the conduct of the study and from Ardea Biosciences for related work that is not part of this manuscript. Dalbeth reports personal fees from Takeda, Teijin, Menarini, Pfizer and Cymabay, grants and personal fees from AstraZeneca, Ardea Biosciences and Fonterra, outside the submitted work. In addition, Dalbeth has a patent Milk Products for Gout pending. Stamp reports consulting fees from Astra Zeneca unrelated to the submitted work.
Rights and permissions
About this article
Cite this article
Roberts, R., Wallace, M., Phipps-Green, A. et al. ABCG2 loss-of-function polymorphism predicts poor response to allopurinol in patients with gout. Pharmacogenomics J 17, 201–203 (2017). https://doi.org/10.1038/tpj.2015.101
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2015.101
This article is cited by
-
Emerging Urate-Lowering Drugs and Pharmacologic Treatment Strategies for Gout: A Narrative Review
Drugs (2023)
-
Comparison Between Early-Onset and Common Gout: A Systematic Literature Review
Rheumatology and Therapy (2023)
-
Genetic polymorphisms and decreased protein expression of ABCG2 urate transporters are associated with susceptibility to gout, disease severity and renal-overload hyperuricemia
Clinical and Experimental Medicine (2022)
-
Multidrug efflux transporter ABCG2: expression and regulation
Cellular and Molecular Life Sciences (2021)
-
Pleiotropic effect of the ABCG2 gene in gout: involvement in serum urate levels and progression from hyperuricemia to gout
Arthritis Research & Therapy (2020)