Abstract
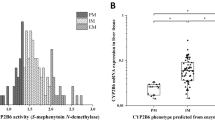
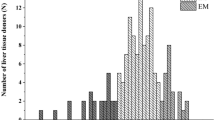
Genetic variations in cytochrome P450 2C19 (CYP2C19) contribute to interindividual variability in the metabolism of therapeutic agents such as clopidogrel. Polymorphisms in CYP2C19 are associated with large interindividual variations in the therapeutic efficacy of clopidogrel. This study evaluated the in vitro oxidation of clopidogrel by 21 CYP2C19 variants harboring amino acid substitutions. These CYP2C19 variants were heterologously expressed in COS-7 cells, and the kinetic parameters of clopidogrel 2-oxidation were estimated. Among the 21 CYP2C19 variants, 12 (that is, CYP2C19.5A, CYP2C19.5B, CYP2C19.6, CYP2C19.8, CYP2C19.9, CYP2C19.10, CYP2C19.14, CYP2C19.16, CYP2C19.19, CYP2C19.22, CYP2C19.24 and CYP2C19.25) showed no or markedly low activity compared with the wild-type protein CYP2C19.1B. This comprehensive in vitro assessment provided insights into the specific metabolic activities of CYP2C19 proteins encoded by variant alleles, and this may to be valuable when interpreting the results of in vivo studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Zhou SF, Liu JP, Chowbay B . Polymorphism of human cytochrome P450 enzymes and its clinical impact. Drug Metab Rev 2009; 41: 89–295.
Zanger UM, Schwab M . Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther 2013; 138: 103–141.
Furuta T, Ohashi K, Kamata T, Takashima M, Kosuge K, Kawasaki T et al. Effect of genetic differences in omeprazole metabolism on cure rates for Helicobacter pylori infection and peptic ulcer. Ann Intern Med 1998; 129: 1027–1030.
Furuta T, Shirai N, Watanabe F, Honda S, Takeuchi K, Iida T et al. Effect of cytochrome P4502C19 genotypic differences on cure rates for gastroesophageal reflux disease by lansoprazole. Clin Pharmacol Ther 2002; 72: 453–460.
Mega JL, Close SL, Wiviott SD, Shen L, Hockett RD, Brandt JT et al. Cytochrome p-450 polymorphisms and response to clopidogrel. N Engl J Med 2009; 360: 354–362.
Maeda A, Ando H, Asai T, Ishiguro H, Umemoto N, Ohta M et al. Differential impacts of CYP2C19 gene polymorphisms on the antiplatelet effects of clopidogrel and ticlopidine. Clin Pharmacol Ther 2011; 89: 229–233.
Mao L, Jian C, Changzhi L, Dan H, Suihua H, Wenyi T et al. Cytochrome CYP2C19 polymorphism and risk of adverse clinical events in clopidogrel-treated patients: a meta-analysis based on 23,035 subjects. Arch Cardiovasc Dis 2013; 106: 517–527.
Kim KA, Park PW, Hong SJ, Park JY . The effect of CYP2C19 polymorphism on the pharmacokinetics and pharmacodynamics of clopidogrel: a possible mechanism for clopidogrel resistance. Clin Pharmacol Ther 2008; 84: 236–242.
Hiratsuka M . In vitro assessment of the allelic variants of cytochrome p450. Drug Metab Pharmacokinet 2012; 27: 68–84.
Kazui M, Nishiya Y, Ishizuka T, Hagihara K, Farid NA, Okazaki O et al. Identification of the human cytochrome P450 enzymes involved in the two oxidative steps in the bioactivation of clopidogrel to its pharmacologically active metabolite. Drug Metab Dispos 2010; 38: 92–99.
Pan Y, Mak JW, Ong CE . Heterologous expression of human cytochrome P450 (CYP) 2C19 in Escherichia coli and establishment of RP-HPLC method to serve as activity marker. Biomed Chromatogr 2013; 27: 859–865.
Lewis DF . The CYP2 family: models, mutants and interactions. Xenobiotica 1998; 28: 617–661.
Wang H, An N, Gao Y, Liu D, Bian T, Zhu J et al. Evaluation of the effects of 20 nonsynonymous single nucleotide polymorphisms of CYP2C19 on S-mephenytoin 4'-hydroxylation and omeprazole 5'-hydroxylation. Drug Metab Dispos 2011; 39: 830–837.
Ibeanu GC, Goldstein JA, Meyer U, Benhamou S, Bouchardy C, Dayer P et al. Identification of new human CYP2C19 alleles (CYP2C19*6 and CYP2C19*2B) in a Caucasian poor metabolizer of mephenytoin. J Pharmacol Exp Ther 1998; 286: 1490–1495.
Bridges A, Gruenke L, Chang YT, Vakser IA, Loew G, Waskell L . Identification of the binding site on cytochrome P450 2B4 for cytochrome b5 and cytochrome P450 reductase. J Biol Chem 1998; 273: 17036–17049.
Straub P, Ramarao MK, Kemper B . Preference for aromatic substitutions at tryptophan-120, which is highly conserved and a potential mediator of electron transfer in cytochrome P450 2C2. Biochem Biophys Res Commun 1993; 197: 433–439.
Hichiya H, Tanaka-Kagawa T, Soyama A, Jinno H, Koyano S, Katori N et al. Functional characterization of five novel CYP2C8 variants, G171S, R186X, R186G, K247R, and K383N, found in a Japanese population. Drug Metab Dispos 2005; 33: 630–636.
Zhou Q, Yu XM, Lin HB, Wang L, Yun QZ, Hu SN et al. Genetic polymorphism, linkage disequilibrium, haplotype structure and novel allele analysis of CYP2C19 and CYP2D6 in Han Chinese. Pharmacogenomics J 2009; 9: 380–394.
Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (KAKENHI 23659070), Smoking Research Foundation, Takeda Science Foundation and Japan Research Foundation for Clinical Pharmacology. We thank the Biomedical Research Core of Tohoku University Graduate School of Medicine for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Rights and permissions
About this article
Cite this article
Takahashi, M., Saito, T., Ito, M. et al. Functional characterization of 21 CYP2C19 allelic variants for clopidogrel 2-oxidation. Pharmacogenomics J 15, 26–32 (2015). https://doi.org/10.1038/tpj.2014.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2014.30
This article is cited by
-
Allele frequencies of single nucleotide polymorphisms of clinically important drug-metabolizing enzymes CYP2C9, CYP2C19, and CYP3A4 in a Thai population
Scientific Reports (2021)
-
Next-Generation Sequencing of CYP2C19 in Stent Thrombosis: Implications for Clopidogrel Pharmacogenomics
Cardiovascular Drugs and Therapy (2021)
-
In Vitro Functional Characterisation of Cytochrome P450 (CYP) 2C19 Allelic Variants CYP2C19*23 and CYP2C19*24
Biochemical Genetics (2017)