Abstract
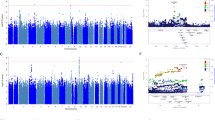
A genome-wide association (GWA) study of treatment outcomes (response and remission) of selective serotonin reuptake inhibitors (SSRIs) was conducted using 529 subjects with major depressive disorder. While no SNP associations reached the genome-wide level of significance, 14 SNPs of interest were identified for functional analysis. The rs11144870 SNP in the riboflavin kinase (RFK) gene on chromosome 9 was associated with 8-week treatment response (odds ratio (OR)=0.42, P=1.04 × 10−6). The rs915120 SNP in the G protein-coupled receptor kinase 5 (GRK5) gene on chromosome 10 was associated with 8-week remission (OR=0.50, P=1.15 × 10−5). Both SNPs were shown to influence transcription by a reporter gene assay and to alter nuclear protein binding using an electrophoretic mobility shift assay. This report represents an example of joining functional genomics with traditional GWA study results derived from a GWA analysis of SSRI treatment outcomes. The goal of this analytical strategy is to provide insights into the potential relevance of biologically plausible observed associations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kessler RC, Berglund P, Demler O, Jin R, Koretz D, Merikangas KR et al. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA 2003; 289: 3095–3105.
Thase ME, Haight BR, Richard N, Rockett CB, Mitton M, Modell JG et al. Remission rates following antidepressant therapy with bupropion or selective serotonin reuptake inhibitors: a meta-analysis of original data from 7 randomized controlled trials. J Clin Psychiatry 2005; 66: 974–981.
Kato M, Serretti A . Review and meta-analysis of antidepressant pharmacogenetic findings in major depressive disorder. Mol Psychiatry 2010; 15: 473–500.
Mrazek DA . Psychiatric Pharmacogenomics. Oxford University Press: New York, NY, 2010.
Serretti A, Artioli P . The pharmacogenomics of selective serotonin reuptake inhibitors. Pharmacogenomics J 2004; 4: 233–244.
Motsinger-Reif AA, Jorgenson E, Relling MV, Kroetz DL, Weinshilboum R, Cox NJ et al. Genome-wide association studies in pharmacogenomics: successes and lessons. Pharmacogenet Genomics 2010.
Garriock HA, Kraft JB, Shyn SI, Peters EJ, Yokoyama JS, Jenkins GD et al. A genomewide association study of citalopram response in major depressive disorder. Biol Psychiatry 2010; 67: 133–138.
Ising M, Lucae S, Binder EB, Bettecken T, Uhr M, Ripke S et al. A genomewide association study points to multiple loci that predict antidepressant drug treatment outcome in depression. Arch Gen Psychiatry 2009; 66: 966–975.
Uher R, Perroud N, Ng MY, Hauser J, Henigsberg N, Maier W et al. Genome-wide pharmacogenetics of antidepressant response in the GENDEP project. Am J Psychiatry 2010; 167: 555–564.
Laje G, McMahon FJ . Genome-wide association studies of antidepressant outcome: a brief review. Prog Neuropsychopharmacol Biol Psychiatry 2011; 35: 1553–1557.
Malhotra AK . The pharmacogenetics of depression: enter the GWAS. Am J Psychiatry 2010; 167: 493–495.
Ji Y, Hebbring S, Zhu H, Jenkins GD, Biernacka J, Snyder K et al. Glycine and a glycine dehydrogenase (GLDC) SNP as citalopram/escitalopram response biomarkers in depression: pharmacometabolomics-informed pharmacogenomics. Clin Pharmacol Ther 2010; 89: 97–104.
Ji Y, Biernacka J, Snyder K, Drews M, Pelleymounter LL, Colby C et al. Catechol O-methyltransferase pharmacogenomics and selective serotonin reuptake inhibitor response. Pharmacogenomics J 2012; 12: 78–85.
Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry 2006; 163: 28–40.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81: 559–575.
Pritchard JK, Stephens M, Donnelly P . Inference of population structure using multilocus genotype data. Genetics 2000; 155: 945–959.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D . Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 2006; 38: 904–909.
Niu N, Qin Y, Fridley BL, Hou J, Kalari KR, Zhu M et al. Radiation pharmacogenomics: a genome-wide association approach to identify radiation response biomarkers using human lymphoblastoid cell lines. Genome Res 2010; 20: 1482–1492.
Hustad S, Midttun O, Schneede J, Vollset SE, Grotmol T, Ueland PM . The methylenetetrahydrofolate reductase 677C—>T polymorphism as a modulator of a B vitamin network with major effects on homocysteine metabolism. Am J Hum Genet 2007; 80: 846–855.
Smythies JR, Alarcon RD, Morere D, Monti JA, Steele M, Tolbert LC et al. Abnormalities of one-carbon metabolism in psychiatric disorders: study of methionine adenosyltransferase kinetics and lipid composition of erythrocyte membranes. Biol Psychiatry 1986; 21: 1391–1398.
Alarcon RD, Tolbert LC, Monti JA, Morere DA, Walter-Ryan WG, Kemp B et al. One-carbon metabolism disturbances in affective disorders. A preliminary report. J Affect Disord 1985; 9: 297–301.
Reynolds EH, Carney MW, Toone BK . Methylation and mood. Lancet 1984; 2: 196–198.
Stanger O, Fowler B, Piertzik K, Huemer M, Haschke-Becher E, Semmler A et al. Homocysteine, folate and vitamin B12 in neuropsychiatric diseases: review and treatment recommendations. Expert Rev Neurother 2009; 9: 1393–1412.
Lewis SJ, Lawlor DA, Davey Smith G, Araya R, Timpson N, Day IN et al. The thermolabile variant of MTHFR is associated with depression in the British Women's Heart and Health Study and a meta-analysis. Mol Psychiatry 2006; 11: 352–360.
Murakami K, Miyake Y, Sasaki S, Tanaka K, Arakawa M . Dietary folate, riboflavin, vitamin B-6, and vitamin B-12 and depressive symptoms in early adolescence: the Ryukyus Child Health Study. Psychosom Med 2010; 72: 763–768.
Chumakov I, Blumenfeld M, Guerassimenko O, Cavarec L, Palicio M, Abderrahim H et al. Genetic and physiological data implicating the new human gene G72 and the gene for D-amino acid oxidase in schizophrenia. Proc Natl Acad Sci USA 2002; 99: 13675–13680.
Wang X, He G, Gu N, Yang J, Tang J, Chen Q et al. Association of G72/G30 with schizophrenia in the Chinese population. Biochem Biophys Res Commun 2004; 319: 1281–1286.
Schumacher J, Jamra RA, Freudenberg J, Becker T, Ohlraun S, Otte AC et al. Examination of G72 and D-amino-acid oxidase as genetic risk factors for schizophrenia and bipolar affective disorder. Mol Psychiatry 2004; 9: 203–207.
Nixon DC, Prust MJ, Sambataro F, Tan HY, Mattay VS, Weinberger DR et al. Interactive effects of DAOA (G72) and catechol-O-methyltransferase on neurophysiology in prefrontal cortex. Biol Psychiatry 2011; 69: 1006–1008.
Gaysina D, Cohen-Woods S, Chow PC, Martucci L, Schosser A, Ball HA et al. Association analysis of DAOA and DAO in bipolar disorder: results from two independent case-control studies. Bipolar Disord 2010; 12: 579–581.
Addington AM, Gornick M, Sporn AL, Gogtay N, Greenstein D, Lenane M et al. Polymorphisms in the 13q33.2 gene G72/G30 are associated with childhood-onset schizophrenia and psychosis not otherwise specified. Biol Psychiatry 2004; 55: 976–980.
Villafuerte SM, Vallabhaneni K, Sliwerska E, McMahon FJ, Young EA, Burmeister M . SSRI response in depression may be influenced by SNPs in HTR1B and HTR1A. Psychiatr Genet 2009; 19: 281–291.
Mekli K, Payton A, Miyajima F, Platt H, Thomas E, Downey D et al. The HTR1A and HTR1B receptor genes influence stress-related information processing. Eur Neuropsychopharmacol 2011; 21: 129–139.
Dickel DE, Veenstra-VanderWeele J, Bivens NC, Wu X, Fischer DJ, Van Etten-Lee M et al. Association studies of serotonin system candidate genes in early-onset obsessive-compulsive disorder. Biol Psychiatry 2007; 61: 322–329.
Cao JX, Hu J, Ye XM, Xia Y, Haile CA, Kosten TR et al. Association between the 5-HTR1B gene polymorphisms and alcohol dependence in a Han Chinese population. Brain Res 2011; 1376: 1–9.
Garcia-Sevilla JA, Alvaro-Bartolome M, Diez-Alarcia R, Ramos-Miguel A, Puigdemont D, Perez V et al. Reduced platelet G protein-coupled receptor kinase 2 in major depressive disorder: antidepressant treatment-induced upregulation of GRK2 protein discriminates between responder and non-responder patients. Eur Neuropsychopharmacol 2010; 20: 721–730.
Hu LA, Chen W, Premont RT, Cong M, Lefkowitz RJ . G protein-coupled receptor kinase 5 regulates beta 1-adrenergic receptor association with PSD-95. J Biol Chem 2002; 277: 1607–1613.
Tiberi M, Nash SR, Bertrand L, Lefkowitz RJ, Caron MG . Differential regulation of dopamine D1A receptor responsiveness by various G protein-coupled receptor kinases. J Biol Chem 1996; 271: 3771–3778.
Rockman HA, Choi DJ, Rahman NU, Akhter SA, Lefkowitz RJ, Koch WJ . Receptor-specific in vivo desensitization by the G protein-coupled receptor kinase-5 in transgenic mice. Proc Natl Acad Sci USA 1996; 93: 9954–9959.
Gainetdinov RR, Bohn LM, Walker JK, Laporte SA, Macrae AD, Caron MG et al. Muscarinic supersensitivity and impaired receptor desensitization in G protein-coupled receptor kinase 5-deficient mice. Neuron 1999; 24: 1029–1036.
Liggett SB, Cresci S, Kelly RJ, Syed FM, Matkovich SJ, Hahn HS et al. A GRK5 polymorphism that inhibits beta-adrenergic receptor signaling is protective in heart failure. Nat Med 2008; 14: 510–517.
Lobmeyer MT, Wang L, Zineh I, Turner ST, Gums JG, Chapman AB et al. Polymorphisms in genes coding for GRK2 and GRK5 and response differences in antihypertensive-treated patients. Pharmacogenet Genomics 2011; 21: 42–49.
Marchetto MC, Brennand KJ, Boyer LF, Gage FH . Induced pluripotent stem cells (iPSCs) and neurological disease modeling: progress and promises. Hum Mol Genet 2011; 20: R109–R115.
Takeuchi F, McGinnis R, Bourgeois S, Barnes C, Eriksson N, Soranzo N et al. A genome-wide association study confirms VKORC1, CYP2C9, and CYP4F2 as principal genetic determinants of warfarin dose. PLoS genetics 2009; 5: e1000433.
Shuldiner AR, O’Connell JR, Bliden KP, Gandhi A, Ryan K, Horenstein RB et al. Association of cytochrome P450 2C19 genotype with the antiplatelet effect and clinical efficacy of clopidogrel therapy. Jama 2009; 302: 849–857.
Ingle JN, Schaid DJ, Goss PE, Liu M, Mushiroda T, Chapman JA et al. Genome-wide associations and functional genomic studies of musculoskeletal adverse events in women receiving aromatase inhibitors. J Clin Oncol 28: 4674–4682.
Acknowledgements
This research was supported, in part, by NIH grants RO1 GM28157, U19 GM61388 (The Pharmacogenomics Research Network), P20 1P20AA017830-01 (The Mayo Clinic Center for Individualized Treatment of Alcohol Dependence), and a PhRMA Foundation Center of Excellence in Clinical Pharmacology Award. Dr Yuan Ji’s work was supported by a KL2 Mentored Career Development Award (NIH/NCRR/NCATS KL2 TR000136) and a Gerstner Family Mayo Career Development Award in Individualized Medicine. This publication was supported by NIH/NCRR/NCATS CTSA Grant Number UL1 RR024150. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. We thank the staff of the Mayo Clinic Rochester Department of Psychiatry and Psychology for their effort in recruiting the patients to the Mayo PGRN-AMPS, Dr Ryan Abo for his assistance in gene annotation, and Lori Solmonson for her assistance with the preparation of the paper. The authors also wish to thank the STAR*D investigators for both their effort and generosity in sharing the clinical data and DNA samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Mrazek has developed intellectual property that has been licensed by AssureRx Health, which has been subsequently incorporated into a physician decision support software product. He has also received research funding from AssureRx Health to create and maintain a bibliographic system designed to monitor the scientific literature related to studies addressing psychiatric pharmacogenomic relationships. No other author has any potential conflict of interest to report.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Ji, Y., Biernacka, J., Hebbring, S. et al. Pharmacogenomics of selective serotonin reuptake inhibitor treatment for major depressive disorder: genome-wide associations and functional genomics. Pharmacogenomics J 13, 456–463 (2013). https://doi.org/10.1038/tpj.2012.32
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2012.32
Keywords
This article is cited by
-
Investigating genetic variants for treatment response to selective serotonin reuptake inhibitors in syndromal factors and side effects among patients with depression in Taiwanese Han population
The Pharmacogenomics Journal (2023)
-
Optimizing prediction of response to antidepressant medications using machine learning and integrated genetic, clinical, and demographic data
Translational Psychiatry (2021)
-
Whole-genome sequencing reveals KRTAP1-1 as a novel genetic variant associated with antidepressant treatment outcomes
Scientific Reports (2021)
-
Influence of GRK5 gene polymorphisms on ritodrine efficacy and adverse drug events in preterm labor treatment
Scientific Reports (2020)
-
Genome-wide association studies of antidepressant class response and treatment-resistant depression
Translational Psychiatry (2020)