Abstract
Stroke survivors often experience social isolation. Social interaction improves quality of life and decreases mortality after stroke. Male mice (20–25 g; C57BL/6N), all initially pair housed, were subjected to middle cerebral artery occlusion (MCAO). Mice were subsequently assigned into one of three housing conditions: (1) Isolated (SI); (2) Paired with their original cage mate who was also subjected to stroke (stroke partner (PH-SP)); or (3) Paired with their original cage mate who underwent sham surgery (healthy partner (PH-HP)). Infarct analysis was performed 72 h after stroke and chronic survival was assessed at day 30. Immediate post-stroke isolation led to a significant increase in infarct size and mortality. Interestingly, mice paired with a healthy partner had significantly lower mortality than mice paired with a stroke partner, despite equivalent infarct damage. To control for changes in infarct size induced by immediate post-stroke isolation, additional cohorts were assessed that remained pair housed for three days after stroke prior to randomization. Levels of brain-derived neurotrophic factor (BDNF) were assessed at 90 days and cell proliferation (in cohorts injected with 5-bromo-2′-deoxyuridine, BrdU) was evaluated at 8 and 90 days after stroke. All mice in the delayed housing protocol had equivalent infarct volumes (SI, PH-HP and PH-SP). Mice paired with a healthy partner showed enhanced behavioral recovery compared with either isolated mice or mice paired with a stroke partner. Behavioral improvements paralleled changes in BDNF levels and neurogenesis. These findings suggest that the social environment has an important role in recovery after ischemic brain injury.
Similar content being viewed by others
Introduction
It is increasingly accepted that social interactions are critical for maintaining physical, mental and social well-being. Social isolation has been associated with increased blood pressure, elevated cortisol levels,1 enhanced inflammatory and metabolic responses to stress,2,3 and modifications in transcriptional pathways linked with glucocorticoid and inflammatory signaling.4, 5, 6, 7 Compared with individuals with social cohesion, isolated individuals have enhanced susceptibility for the development of cardiovascular disorders,8 infectious diseases,9 cognitive decline10 and increased risk of cerebrovascular disorders including ischemic stroke.11 Stroke is now the fourth leading cause of death in the United States.12 Stroke-related mortality has dropped over the past 10 years, primarily due to improvements in medical care and acute stroke treatment, but this has not been accompanied by a decrease in the incidence of ischemic stroke. This is alarming, as stroke is the leading cause of chronic disability in adults, and increases in the number of stroke survivors will further escalate health care costs over the next several decades as our population ages.12,13 Therefore, any potential opportunity to reduce stroke-related disability and improve functional outcome would have tremendous public health impact.
Data from the US census show an increase in the number of people living alone since 1980s; a trend that is expected to continue.14 Individuals who report lack of social support or isolation have an increased incidence of recurrent stroke, poorer recovery, greater functional decline and higher mortality following a stroke compared with individuals with strong social support.15, 16, 17, 18 As not all strokes can be prevented, more attention is needed to develop strategies to improve function after a stroke has occurred.
Attesting to the importance of social factors in stroke outcome is that these same effects can be reproducibly demonstrated in animals; social interaction has been shown to reduce histological damage from experimental stroke, whereas pre-stroke social isolation (SI) enhances injury.7,19,20 To date, previous studies have focused on isolation prior to, or at the time of, induction of injury.21,22 However, as most patients are not identified as ‘isolated’ until after their stroke, the examination of the effects of post-stroke isolation is important. In addition, as isolation increases infarct size, prior studies have been confounded by the larger infarcts seen in isolated animals. We hypothesized that social interaction will enhance functional recovery independently of infarct size and that the partner’s health status might have a role. To test this hypothesis, we utilized a novel delayed post-stroke housing paradigm to normalize infarct size between groups. The effect of post-stroke environmental manipulation on histological outcomes, IL-6 levels, chronic behavioral recovery and neurogenesis was assessed.
Materials and methods
Experimental animals
Six-week-old C57Bl/6 mice weighing 20–22 g were purchased from Charles River laboratories (Wilmington, MA, USA) and allowed to acclimate in groups of four for two weeks prior to any housing manipulation. All mice were maintained in a temperature- and humidity-controlled vivarium, with ad libitum access to food and water.
Housing conditions and experimental design
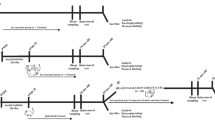
All experimental animals were screened for baseline locomotor activity in the open field. Mice were housed in standard mouse cages (11"L, 6"W, 6"H) with a 12-h light/dark schedule (lights on at 0700 h). Body weights were recorded and mice were pair housed (all mice were housed two per cage with an original cage mate) for 2 weeks before subjecting them to right middle cerebral artery occlusion (MCAO) or sham surgery. Animals were then randomly assigned to one of the three housing conditions—either housed isolated (SI), pair housed with a stroke partner (PH-SP) or pair housed with a healthy partner (PH-HP) either immediately after stroke (cohort-1) or three days after MCAO (cohort-2). In the first cohort, two endpoints were assessed; a three-day survival group was used for neurological deficit scores (NDS), infarct analysis and changes in serum interleukin-6 levels; a 30-day survival group that was used to assess mortality rates (see Figure 1a). A second cohort (cohort-2) was used to study delayed post-stroke housing effects with two different endpoints at 8 and 90 days. Animals were tested in the cylinder, corner and on the tail suspension test (TST) (see Figure 2a for timeline of behavioral testing) and sacrificed at day 90, for BDNF analysis. In the 8-day endpoint, mice were injected with BrdU from day 3–7 and analysis was performed 24 h after the last injection (day 8), an additional sub-group of mice was allowed to survive for 90 days to confirm neuronal maturation.
Effects of post-stroke social isolation on stroke outcome: (a) Schematic of experimental design. Following a 60-min MCAO, mice were either single housed or pair housed with a partner mouse also subjected to stroke (PH-SP) or who was a healthy sham (PH-HP) on day 0. (b) Seventy-two hours after stroke (d3), brains were taken for histological analysis using TTC staining. (c) Quantification of infarcts at 72 h after stroke (d3) showed that SI mice had significantly larger ischemic damage compared with PH mice (*P<0.05); however, no significant differences were seen in mice that were pair housed with either a stroke partner or a healthy partner; n=10 per group. (d) Neurological deficit scores prior to sacrifice showed a lack of recovery in SI cohort relative to intra-ischemic score, while both pair-housed groups recovered significantly (there were no significant differences between PH-SP and PH-HP groups); Friedman’s test was used for analysis, *P<0.05; n=10 per group. (e) Post-stroke, SI mice had significantly enhanced serum levels of IL-6 compared with PH-SP and PH-HP mice; n=6 per group; *P<0.05 compared with PH groups. Error bars denote s.e.m. HP, healthy partner; PH, pair housed; MCAO, middle cerebral artery occlusion; SI, social isolation; SP, stroke partner; TTC, 2,3,5-triphenyltetrazolium chloride.
Beneficial effects of delayed post-stroke pair housing on stroke outcome were independent of infarct volume. (a) Schematic of the experimental design used to assess effects of delayed housing on functional recovery. (b) Mice housed with a healthy partner (PH-HP) show enhanced recovery in the cylinder test by day 7 compared with PH-SP and SI. PH-HP cohorts almost fully recovered by day 21. Conversely, SI mice demonstrated significant forelimb impairment even at day 28 compared with shams and both pair-housed stroke groups. (c) Significant delay in recovery was also observed in SI mice in the corner test compared with PH-SP mice at day 14, whereas a significantly faster recovery was achieved in PH-HP mice by day 7 compared with PH-SP. *Significant difference (P<0.05) compared with SI group; **Significant difference in PH-HP compared with PH-SP. Error bars denote s.e.m. HP, healthy partner; PH, pair housed; SI, social isolation; SP, stroke partner.
All animals were given wet mash for the initial 72 h after stroke or sham surgery. As mortality is higher when chronic endpoints are assessed, all animals that had planned survival endpoints over 3 days were injected with 100 μl of saline subcutaneously for 5 days after surgery. This prevents weight loss and reduces mortality, both stroke and sham mice were administered injections to control for any potential stress of handling or repeated injection. Quantification of infarct volume and all behavioral tests were performed and recorded by an investigator blinded to housing conditions. All behavior tests were performed between 0800 and 1100 h, immediately at the beginning of the light cycle after acclimatizing mice to the testing room lighting conditions. Testing chambers were cleaned with 70% ethanol between mice.
Exclusion criteria
If any mouse did not gain at least one gram of weight during the 2 weeks of pre-stroke pair housing, lost weight during the two weeks of PH, or exhibited signs of fight wounds due to dominant/aggressive behavior of the partner, both mice were excluded from the study as social aversion/stress can also influence stroke outcome.4,23,24 If one mouse of the pair dies at any point during the experiment, that pair is excluded from further study.
Middle cerebral artery occlusion model
Focal transient cerebral ischemia was induced by right MCAO followed by reperfusion as described previously.7 Core body temperature was monitored with a rectal probe connected to a temperature control system (Fine science tools, North Vancouver, BC, Canada). Temperature was maintained with an automatic heating pad at ∼37 °C during surgery and ischemia. Cerebral blood flow measurements by laser Doppler flowmetry confirmed ischemia during MCAO (to <80% of baseline) and restoration of flow during reperfusion in all cohorts.
Infarct analysis
Following 72 h of reperfusion, all animals in cohort 1 were euthanized by pentobarbital overdose, brains were removed and cut into five 2-mm coronal sections and stained with 1.5% 2,3,5-triphenyltetrazolium chloride (TTC) for 8 min at 38 °C. Slices were formalin-fixed (4%) and then digitalized and infarct volumes analyzed (Sigma Scan Pro, San Jose, CA, USA) as previously described.7 The final infarct volumes are presented as a percentage (percentage of contralateral structures with correction for edema) as in Venna et al.7 For histology and immunohistochemistry, brains were collected from additional chronic survival cohorts. Animals were deeply anesthetized and perfused transcardially with ice-cold sodium phosphate-buffered saline followed by 4% paraformaldehyde. Brains were cryoprotected in 30% sucrose.
Neurological deficit scores
Neurological deficit scores were obtained in the intra-ischemic period and at 72 h post-stroke in short-term survival cohort-1. The scoring system was as follows: 0, no deficit; 1, forelimb weakness and torso turning to one side when held by tail; 2, circling to affected side; 3, unable to bear weight on affected side and circling immediately when placed on a bench; and 4, no spontaneous locomotor activity or barrel rolling as described previously as in Venna et al.7
IL-6 ELISA
Serum was collected from the mice sacrificed at 72 h post-stroke in mice subjected to the three housing conditions (SI, PH-SP or PH-HP) immediately after stroke. Interleukin-6 (IL-6) levels were examined utilizing an enzyme-linked immune absorbent (ELISA) assay (eBiosciences, San Diego, CA, USA) per manufacturer’s instructions.
Cylinder test
The cylinder test was used to assess forelimb use after stroke. The mouse was placed in a transparent Plexiglas cylinder 9-cm diameter and 15 cm in height. A mirror was placed behind the cylinder with an angle to enable the rater to record forelimb movements when the mouse was turned away from the rater. A total of 20 forelimb placements were counted for analysis as previously described.7,25
Corner test
Following the cylinder test animals were tested in the corner test as described in (Venna et al.26). The mouse was placed between two pieces of cardboard, each with a dimension of 30 × 20 × 1 cm. The two boards were gradually moved to enclose the mouse from both sides to encourage the mouse to enter into a corner of 30° angle. After vibrissae stimulation, the mouse rears forward and upward, then turns back to face the opening. Twenty trials were performed for each mouse and the percentage of right turns was calculated.
Open field
Mice were placed in an open field chamber (15" × 15") equipped with 16 infrared beam emitting LEDs. The total number of beam breaks were automatically registered by a computer-operated PAS Open Field system (San Diego Instruments, San Diego, CA, USA). The total number of beam breaks during a 20 min session was analyzed as a measure of spontaneous locomotor activity.
Tail suspension test
Animals were returned to their home cages after open field analysis and allowed free access to food and water at least for 30 min before testing them in TST. TST was performed as in Venna et al.26 Mice were suspended horizontally by the tail to a metal bar using adhesive scotch tape for a total duration of 6 min. The test was analyzed and the times of active escape behavior (mobility) versus passive behavior with no limb or head movements (immobility) was recorded. Latency to first immobile period was also quantified27 and is reported as latency. Behavior was scored manually by an experienced rater blinded to housing condition.
BrdU Treatment
Separate cohorts of mice were injected intraperitoneally with 75 mg kg−1per day of BrdU once daily during days 3–7 after stroke.28 BrdU (Sigma Aldrich, MO, USA) was dissolved in 0.007 N NaOH in 0.9% NaCl. Mice were either sacrificed on day 8 or on day 90 (n=4) to confirm neuronal maturation. BrdU+ cell counts were performed on stained sections from perfused brains29,30 (n=8 per group) as detailed below.
Immunofluorescence
Brains were cut in 30 micron sections and mounted on superfrost plus slides (Fisher scientific, Pittsburg, PA, USA), then subjected to microwave irradiation for 5 min in 0.1 M, pH 6 citrate buffer solution followed by incubation in blocking solution.7 Sections were incubated overnight with rat anti-BrdU (1:100, AbD Serotec, Raleigh, NC, USA) and mouse Anti-NeuN (1:200, Millipore, Billerica, MA, USA) to confirm neuronal lineage. Sections were subjected to three five-minute washes with PBS and then incubated for 60 min with fluorescein-conjugated anti-mouse and rhodamine-conjugated anti-rat secondary antibodies (Alexafluor, Grand Island, NY). Then brain sections were washed, then dipped in DAPI solution (1:1000) for five minutes and cover slipped. Sections were visualized under an inverted light Zeiss axiovert fluorescence microscope.7 Newborn cells were identified by co-labeling with BrdU+ cells and DAPI. To perform quantification of BrdU+ cells, four coronal brain slices per animal (n=8 animals per group) were stained from ~1.1 mm bregma, 0.8 mm bregma, 0.5 mm bregma and −0.1 mm bregma. Two 20 × fields per section were analyzed on the infarct side. BrdU+ cells were counted using MacBiophotonics ImageJ software (Hamilton, ON, Canada). The total numbers of cells per field of view were counted by a blinded investigator and the average number of cells per field was used for statistical analysis.29,30
Western blot
Brains were rapidly extracted; hippocampi were isolated from brains and were immediately flash frozen. The protein concentration was determined by BCA Protein Assay Kit (Thermo Fisher Scientific Inc., Rockford, lL, USA) to achieve equal loading of 10 μg per well.7 Sample proteins were resolved on SDS electrophoresis gels and transferred to a polyvinylidene diflouride membrane. All blots were blocked with 5% milk and incubated overnight with BDNF (1:200; Millipore) and actin (1:5000; Sigma-loading control) at 4 °C in TBS containing 4% bovine serum albumin and 0.1% Tween. Secondary antibodies (goat anti-rabbit IgG 1:5000 for BDNF, anti-mouse 1:1000 for actin) were diluted, and ECL detection kit (Amersham Biosciences, Buckinghamshire, UK) was used for signal detection. Densitometry was performed (Adobe). Data is presented as ratio to actin normalized to SI.
Statistics
Data were expressed as mean±s.e.m. except for NDS, which was presented as median (interquartile range). Statistics were performed using SPSS version PASW17. One-way analysis of variance (ANOVA) for analysis involving more than two independent groups, Bonferroni post hoc for multiple comparisons, repeated measures for cylinder and corner tests, a Z-test for proportions was used to analyze survival rates and the Friedman test with Bonferroni correction used for repeated measures ordinal data (i.e., NDS). A probability value, P<0.05 was considered to be statistically significant. Each dyad was treated as one individual sample for statistical analysis. Investigators performing MCAO, behavioral and infarct size analysis were blind to housing conditions.
Results
Infarct analysis
To examine whether post-stroke housing manipulation influences stroke outcome, all mice were initially pair housed for two weeks prior to a 60-min MCAO and assigned to one of the three ‘immediate post-stroke’ housing conditions: (1) Housed in a cage individually (SI), (2) paired with its original partner also subjected to stroke surgery (PH-SP) (3) paired with its original partner (not subjected to stroke) (PH-HP) (Figure 1a). Regardless of the partner’s health condition, pair-housed mice had significantly smaller infarcts 72 h after stroke compared with SI cohorts in the cortex F(2,26)=5.12, P<0.05; striatum F(2,26)=7.24, P<0.01 and in the total hemisphere F(2,26)=13.03, P<0.01. No significant differences were seen in infarct volumes between PH-HP and PH-SP cohorts; the mean difference (MD) in cortex=4.3, P=0.644; MD striatum=0.344, P=0.997 and MD total=1.56, P=0.93 (Figure 1c).
Neurological deficit scores and serum IL-6 levels
The detrimental effects of SI were also reflected in the NDS, Friedman test for repeated measures revealed no recovery in SI mice χ2(1)=0.0001; P=1.0, but a significant recovery in both PH-SP χ2(1)=10, P=0.002 and in PH-HP χ2(1)=9; P=0.003 (Figure 1d). SI mice had significantly elevated serum IL-6 compared with PH cohorts after stroke F(2,15)=16.38, P<0.05. (Figure 1e), post hoc tests revealed no significant difference between PH-SP and PH-HP groups; P>0.05. Sham mice has no significant changes in serum IL-6 levels F(2,13)=0.64, P=0.54. These results revealed a similar pattern to that previously seen in mice exposed to pre-stroke SI with isolated animals sustaining larger infarcts, an enhanced inflammatory response and poorer functional recovery than animals housed together.7,19,20
Exclusion and mortality rates
A total of 4% of original mouse pairs were excluded at the beginning of the study due to partner aggression as per pre-set exclusion criteria. In cohort-1 (immediate post-stroke isolation) SI mice had significantly higher mortality (61%) compared with PH-SP (39%), P<0.05. PH-HP mice had only a 17% mortality (P<0.01) (Figure 3) at 30 days. These findings suggest that interactions with a healthy partner might offer additional benefit and improve post-stroke survival. These effects appear to be independent of the effects on acute infarct damage, which was equivalent between the two cohorts of PH mice. Survival rates in cohort-2 (delayed post-stroke isolation) were higher as post-stroke care was more aggressive, but SI mice still had significantly higher mortality (20%) compared with PH-SP (10%); (P<0.05), and in PH-HP (3%); (P<0.001).
Chronic survival rates in isolated and pair-housed mice: survival rates were recorded in mice that were placed immediately into assigned housing conditions after stroke. Mortality rates assessed through day 30 revealed a significantly higher mortality rate in SI mice compared with PH-SP, *P<0.05 and **P<0.01 in PH-HP cohorts (n=18 mice per group initially). Data presented in Kaplan–Meier curves and Z-test for proportions are used for statistical analysis. HP, healthy partner; PH, pair housed; SI, social isolation; SP, stroke partner.
Functional recovery is independent of infarct size and associated with interactions and health of the partner
One possible explanation for the improved recovery in PH mice is that infarct damage is less due to the acute neuroprotection induced by immediate post-stroke housing. To investigate the specificity of relationship between social interaction, partner’s health and post-stroke recovery, we subjected mice to a ‘delayed post-stroke’ housing paradigm (Figure 2a). As infarct damage is complete in this stroke model by 24 h,31 we allowed the infarct to mature prior to housing in an attempt to normalize infarct size between groups. All mice were kept pair housed for 72 h immediately after stroke. In this way, recovery can be separated from the degree of histological damage, which will be equivalent in all cohorts. Mice were then placed back with its previous partner who was either a ‘healthy’ or ‘stroke’ mouse or maintained in isolation (SI). Long-term recovery was assessed with both the cylinder and corner test.25
Repeated measures ANOVA revealed that there were significant asymmetries (reduced contralateral forelimb use) in the cylinder test in stroke cohorts compared with sham animals regardless of housing condition. There was an overall significant effect of surgery F(1,44)=309.09, P<0.01 (Figure 2b), an overall significant effect of housing F(2,44)=18.3, P<0.05. There was also significant surgery-by-housing interaction, F(2,44)=18.83, P<0.01 suggesting differences in recovery. Independent analyses at each time point (days 3–28) revealed that these observed effects are due to more rapid and complete forelimb motor recovery in PH-HP mice compared with PH-SP; P<0.05. SI mice had significantly reduced contralateral limb use compared with PH cohorts even at day 28 (*P<0.01) (Figure 2b). Sham operated mice had no significant differences between groups. Figure 2c illustrates the results of mean non-impaired side turn percentages using the corner test that showed a consistent pattern of recovery. A significant effect of day-by-surgery F(1,44)=53.73, P<0.01 was seen, due to the fact that there was an early reduction in turning preference in PH-HP compared with PH-SP; SI mice had a continued turning preference at day 29 compared with PH cohorts. There was also significant surgery F(1,44)=321.22, P<0.05 and a housing F(2,44)=8.87; P<0.01 for the entire testing period.
Long-term social isolation reduced mobility in tail suspension test and reduced hippocampal BDNF levels
To explore the role of social interaction on depressive phenotypes, mice were subjected to the ‘delayed post-stroke’ housing paradigm detailed above (Figure 2a). When changes in mobility using the TST were evaluated, there was a significant difference between groups F(3,32)=18.8, P<0.01; Bonferroni post hoc analysis revealed a significantly reduced mobility in SI group compared with both pair-housed conditions P<0.05, the PH-SP group had significantly less mobility compared with PH-HP (P=0.04) (Figure 4a). When analyzed for latency to the first bout of immobility, there were significant differences between groups F(3,32)=23.73, P<0.01. Bonferroni post hoc analysis revealed significant differences in latencies between SI and PH-SP (P=0.02) and also a significant difference between PH-HP compared with PH-SP groups (P=0.03). Pair housing significantly reduced the duration of immobility and prolonged the latency to the first bout of immobility compared with SI group (Figure 4b). Independent analysis showed no differences between sham groups, data are presented as pooled for sham groups (Figure 4). Spontaneous locomotor analysis in open field showed no differences between groups F(3,32)=0.98; P=0.42, suggesting that decreased mobility is not due to motor deficits (Figure 4c). Assessment of hippocampal brain-derived neurotrophic factor (BDNF) levels, an important growth factor for neuronal survival and maturation with known antidepressant effects,23,26 showed no differences in sham groups F(2,5)=0.44, P>0.05; but revealed a significant difference between stroke groups F(3,28)=20.12, P<0.05. Bonferroni post hoc tests showed that there was a significant reduction of BDNF expression in SI compared with PH-SP (P=0.008) and PH-HP (P<0.001). BDNF levels in PH-HP were higher than PH-SP (P=0.048) but not significantly different from pooled sham group, P=0.9 (Figure 4d).
Housing with a healthy partner significantly improved long-term behavior and enhanced BDNF levels compared with isolated mice or mice housed with a stroke partner. (a) SI mice showed a depression-like phenotype, expressing less mobility when tested at day 90 using the tail suspension test compared with PH-HP and shams. (b) SI mice also expressed a reduced latency to the first bout of immobility compared with PH-HP and shams. (c) All stroke mice expressed similar spontaneous locomotor activity when tested prior to TST. (d) BDNF levels were significantly reduced at day 90 in whole brain homogenates of SI mice. β-actin was used as loading control and densitometry data are presented as normalized ratio. *P<0.05 compared with SI and **P<0.05 compared with PH-SP. Error bars denote s.e.m. HP, healthy partner; PH, pair housed; SI, social isolation; SP, stroke partner.
Functional recovery paralleled enhanced neurogenesis
Brains were analyzed for histological assessment at 8 days post stroke, no significant differences in infarct size between groups was seen (Figure 5a). Equivalent behavioral deficits were seen at day 3 post stroke prior to housing randomization. Groups (SI vs PH) significantly diverged in their recovery by post-stroke day 7. As ischemia is known to induce neurogenesis, which may be an important factor in functional recovery,32,33 we assessed stroke-induced cell proliferation on day 8 after stroke in the subventricular zone in additional cohorts of animals injected with BrdU at days 3–7 after stroke. SI mice had significantly fewer BrdU+ cells compared with either PH cohorts F(2,96)=60.98, P<0.001 (Figure 5c). There was an additional effect in mice housed with a healthy partner (PH-HP) compared with mice that PH with a stroke partner (PH-SP) at day 8; MD 16.6, P<0.01 (Figure 5c). Quantification revealed that SI mice had significantly fewer BrdU+ cells compared with either PH cohorts F(2,96)=66.07, P<0.001 in the infarct area. There was an additional effect in mice housed with a healthy partner (PH-HP) compared with mice that PH with a stroke partner (PH-SP) at day 8; MD 22.8, P<0.01 (Figure 5d), these changes are consistent with the enhanced recovery seen in that cohort. When assessed for neuronal maturation on confocal brain sections obtained at day 90, we found that a number of BrdU+ cells exhibited markers of mature neurons (NeuN) (Figure 5f).
Functional recovery in pair-housed mice paralleled the increase in BrDU+ cell proliferation. (a) Similar infarct volumes were observed between groups with delayed housing when analyzed at day 8 (n=8 per group). (b) Representative illustration showing how the brain sections were obtained for cell counts; a total of 32 images per group were counted, 4 images per brain and 8 brains per group; images from red-boxed area were used to count BrDU+ cells in SVZ and images from white-boxed area were used to obtain average BrDU+ cell counts within the infarct. (c) Images showing differences in cell proliferation rates seen in PH mice compared with isolated mice at day 8 suggest a potential implication of this mechanism in recovery; bar=100 μm. (d) Data represent BrDU+ cells count from 32 images collected from SVZ (n=8 mice per group). (e) Quantification of BrDU+ cells in infarct area shows significant differences in SI vs PH-SP and in PH-SP vs PH-HP groups. (f) Image showing co-labeling confirms the maturation of newborn cells to neurons, marked with BrDU+NeuN. *P<0.05 compared with SI and **P<0.05 compared with PH-SP. Error bars denote s.e.m. HP, healthy partner; PH, pair housed; SI, social isolation; SP, stroke partner; SVZ, subventricular zone.
Discussion
This study has established that early post-stroke interactions can reduce histological damage after ischemic challenge. These findings are consistent with the beneficial effects of pair housing observed in pre-stroke isolation models, and suggest that this may have important translational relevance for treatment of stroke patients.21 More importantly, this work shows that pair housing, even if delayed for days after stroke, leads to enhanced functional recovery after injury, even in the absence of a significant reduction of histological damage. This novel finding suggests that there is considerable brain plasticity and capacity for repair, even days after injury, enhancing the translational significance of this work. Our results also demonstrate that housing stroke mice with a healthy partner significantly hastens functional recovery and reduces mortality compared with mice housed with a stroke partner, whereras isolated mice demonstrated the poorest recovery. These effects parallel increased cell proliferation and higher hippocampal BDNF levels.
In previous work, we have demonstrated that pre-stroke social isolation is detrimental.7 However, as stroke is an unpredictable event, manipulating social environments prior to an event in patients at risk is likely to be ineffective from a therapeutic standpoint. Therefore we initially wanted to determine if these same manipulations performed after injury would have similar effects at a time when patients would be presenting to medical care. We found that immediate post-stroke pair housing significantly decreased infarct volume, and this was seen in animals paired with either a healthy partner or a partner that also had been subjected to stroke. We further hypothesized that housing with a sick partner would reduce the benefit of PH while a healthy partner may further ameliorate beneficial effects of PH. When tested, no significant differences were observed between PH-HP and PH-SP in either the amount of histological injury or serum IL-6 levels. We also found a similar improvement in functional outcome, as measured by the NDS, regardless of the health of the cage partner. This suggests that the detrimental effects of acute isolation might also be due to pair dissociation/separation effects.34 However, an unexpected and interesting pattern emerged when these animals were followed for 30 days, which was the pre-specified behavioral endpoint. Despite having similar infarct sizes, mice housed with a healthy partner (PH-HP) had significantly higher survival rates than mice housed with a partner that had also been subjected to stroke (PH-SP), although both PH cohorts did better than the isolated cohort. This suggested that there were unknown factors that enhanced survival in mice paired with a healthy partner.
One limitation of the previously published reports examining social factors and stroke is that all have utilized pre-stroke or immediate post-stroke isolation which invariably leads to an increased infarct size.7,19,20,22 Therefore any increase in inflammation, apoptosis or behavioral deficits in isolated cohorts may simply reflect the amount of ischemic injury. To address this and examine the potential therapeutic window for environmental manipulations, we waited 3 days to place mice in the specified housing environment, as infarct is complete in this model by 72 h after injury.31 This allowed us to control the size of the infarct between the three groups, and may be a more realistic ‘therapeutic time window’ in which to treat an acute stroke patient with environmental manipulations. Despite equivalent histological damage in all groups, isolated mice continued to do poorly, with delayed functional recovery and higher mortality. Mice housed with a healthy partner had significantly better behavioral recovery than those housed with a stroke partner. This suggests that there are factors that contribute to the detrimental effects of isolation7,19,20 such as IL-6, NF-kappaB as well as additional factors that underlie the beneficial effects of affiliative interactions such as grooming, whisker stimulation,35,36 which can be further enhanced based on partner’s health. Although all mice were screened and monitored for aggressive behavior towards the partner and we excluded those dyads from the study, we cannot completely rule out the possibility that stroke induced aggressive behavioral changes by the healthy partner. We feel this is less likely as stroke animals housed with a healthy partner actually did much better both in terms of behavior and mortality than mice housed with a stroke partner. In turn, stroke mice housed with another stroke mouse had better outcomes than stroke mice that were isolated. This suggests that housing was beneficial compared with isolation as the isolated mice are clearly not exposed to any partner, aggressive or otherwise.
We hypothesized that the enhanced behavioral recovery seen with PH was secondary to an enhancement in neurogenesis, as has been described with environmental enrichment.37,38 Neurogenesis occurs throughout life in both rodents and humans.39,40 Cerebral ischemia increases cell proliferation in both the subventricular zone and the hippocampus, and these newborn cells can migrate into the infarcted area.30 Enhancements in neurogenesis have been linked to improved functional recovery in many studies,41 but how these cells integrate into the damaged brain and the ultimate potential to enhance repair after stroke is unknown. Both PH-HP and PH-SP mice demonstrated increased cell proliferation rates, which paralleled changes in functional recovery. BDNF is an important mediator of neuronal activity, neurogenesis, brain repair, cell survival and synaptic structure formation.38,41,42 Activation of BDNF and its receptor tropomyosin-related kinase B (TrkB) enhanced neuronal plasticity and neurogenesis after ischemic injury in mice.43 Recently it has been shown that transplantation of mesenchymal stem cells reduced social deficits in social preference tests mainly by enhancing BDNF levels.44 As we also found a reduction in depression-like behavior in PH mice compared with isolated cohorts, and BDNF signaling has also been implicated in the pathophysiology of depression,45 we examined BDNF levels, which were increased in PH animals compared with SI and further enhanced in mice with a healthy partner.
Over ~40% of stroke survivors suffer from depression, and this may be exacerbated by isolation and feelings of loneliness.16,46 Many epidemiological studies have shown that depression is a risk factor for stroke, and is associated with poorer recovery once a stroke has occurred.47,48 The downstream mechanism by which PH prevented depression-like behavior is not known, but likely involves numerous pathways. Nibuya et al.49 showed that antidepressant drugs increase the synthesis of BDNF.49 Traditional antidepressants may act by enhancing BDNF signaling50,51 as BDNF expression increased in depressed patients after treatment.50,52 BDNF activation is essential for manifestation of experience-dependent social aversion and mediating changes in long-term neural and behavioral plasticity in response to social aggression.23 Our work suggests that BDNF also has an important role in the beneficial effects of social interaction after injury. Whether these same effects can be recapitulated with pharmacological treatments is not yet known.
In summary, this study provides the first direct experimental evidence to support a role of post-stroke social interaction in stroke recovery independent of infarct damage. Our findings emphasize that although pair housing is beneficial to recovery, the health of the partner is also an important contributing factor to functional recovery. Given that these effects parallel the enhanced cell proliferation and increased hippocampal BDNF levels, more research is needed to understand how the beneficial effects of social interaction could be translated to clinical practice. Recent results from trials designed to provide reciprocal peer support for heart failure patients found that more than half of the participants had no or minimal engagement with the program, and no improvements in outcomes were seen.53 It is possible that social support may be more effective when provided by a healthy individual rather than a disease-affected peer. As a rehabilitative strategy, efforts to encourage interactions among stroke survivors and healthy society members could offer an important opportunity to reduce the substantial burden of stroke in our communities.
References
Grant N, Hamer M, Steptoe A . Social isolation and stress-related cardiovascular, lipid, and cortisol responses. Ann Behav Med 2009; 37: 29–37.
Loucks EB, Berkman LF, Gruenewald TL, Seeman TE . Relation of social integration to inflammatory marker concentrations in men and women 70–79 years. Am J Cardiol 2006; 97: 1010–1016.
Powell ND, Sloan EK, Bailey MT, Arevalo JM, Miller GE, Chen E et al. Social stress up-regulates inflammatory gene expression in the leukocyte transcriptome via beta-adrenergic induction of myelopoiesis. Proc Natl Acad Sci USA 2013; 110: 16574–16579.
Sugo N, Hurn PD, Morahan MB, Hattori K, Traystman RJ, DeVries AC . Social stress exacerbates focal cerebral ischemia in mice. Stroke 2002; 33: 1660–1664.
Ploughman M, Windle V, MacLellan CL, White N, Dore JJ, Corbett D . Brain-derived neurotrophic factor contributes to recovery of skilled reaching after focal ischemia in rats. Stroke 2009; 40: 1490–1495.
Yang YC, McClintock MK, Kozloski M, Li T . Social isolation and adult mortality: the role of chronic inflammation and sex differences. J Health Soc Behav 2013; 54: 183–203.
Venna VR, Weston G, Benashski SE, Tarabishy S, Liu F, Li J et al. Nf-kappab contributes to the detrimental effects of social isolation after experimental stroke. Acta Neuropathol 2012; 124: 425–438.
Barth J, Schneider S, von Kanel R . Lack of social support in the etiology and the prognosis of coronary heart disease: a systematic review and meta-analysis. Psychosom Med 2010; 72: 229–238.
Cohen S, Doyle WJ, Skoner DP, Rabin BS, Gwaltney JM Jr . Social ties and susceptibility to the common cold. Jama 1997; 277: 1940–1944.
Cacioppo JT, Hawkley LC . Perceived social isolation and cognition. Trends Cogn Sci 2009; 13: 447–454.
Avendano M, Kawachi I, Van Lenthe F, Boshuizen HC, Mackenbach JP, Van den Bos GA et al. Socioeconomic status and stroke incidence in the us elderly: the role of risk factors in the epese study. Stroke 2006; 37: 1368–1373.
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB et al. Executive summary: heart disease and stroke statistics—2013 update: a report from the American heart association. Circulation 2013; 127: 143–152.
Vaartjes I, O'Flaherty M, Capewell S, Kappelle J, Bots M . Remarkable decline in ischemic stroke mortality is not matched by changes in incidence. Stroke 2013; 44: 591–597.
US_Census_Bureau http://www.Census.Gov/hhes/families/data/cps2012.html. 2012.
Hawkley LC, Cacioppo JT . Loneliness and pathways to disease. Brain Behav Immun 2003; 17 ((Suppl 1)): S98–105.
Boden-Albala B, Litwak E, Elkind MS, Rundek T, Sacco RL . Social isolation and outcomes post stroke. Neurology 2005; 64: 1888–1892.
Cacioppo JT, Hughes ME, Waite LJ, Hawkley LC, Thisted RA . Loneliness as a specific risk factor for depressive symptoms: cross-sectional and longitudinal analyses. Psychol Aging 2006; 21: 140–151.
Holt-Lunstad J, Smith TB, Layton JB . Social relationships and mortality risk: a meta-analytic review. PLoS Med 2010; 7: e1000316.
Craft TK, Glasper ER, McCullough L, Zhang N, Sugo N, Otsuka T et al. Social interaction improves experimental stroke outcome. Stroke 2005; 36: 2006–2011.
Karelina K, Norman GJ, Zhang N, Morris JS, Peng H, DeVries AC . Social isolation alters neuroinflammatory response to stroke. Proc Natl Acad Sci USA 2009; 106: 5895–5900.
Venna VR, McCullough LD . ‘won't you be my neighbor?’: Deciphering the mechanisms of neuroprotection induced by social interaction. Stroke 2011; 42: 3329–3330.
O’Keefe LM Doran SJ Mwilambwe-Tshilobo L Conti LH Venna VR McCullough LD . Social isolation after stroke leads to depressive-like behavior and decreased bdnf levels in mice. Behav Brain Res 2014; 260: 162–170.
Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ et al. Essential role of bdnf in the mesolimbic dopamine pathway in social defeat stress. Science 2006; 311: 864–868.
Barik J, Marti F, Morel C, Fernandez SP, Lanteri C, Godeheu G et al. Chronic stress triggers social aversion via glucocorticoid receptor in dopaminoceptive neurons. Science 2013; 339: 332–335.
Li X, Blizzard KK, Zeng Z, DeVries AC, Hurn PD, McCullough LD . Chronic behavioral testing after focal ischemia in the mouse: Functional recovery and the effects of gender. Exp Neurol 2004; 187: 94–104.
Venna VR, Deplanque D, Allet C, Belarbi K, Hamdane M, Bordet R . Pufa induce antidepressant-like effects in parallel to structural and molecular changes in the hippocampus. Psychoneuroendocrinology 2009; 34: 199–211.
Castagné V, Moser P, Roux S, Porsolt RD . Rodent models of depression: forced swim and tail suspension behavioral despair tests in rats and mice. Curr Protoc Neurosci 2011; Chapter 8: Unit 8.10A.
Liu F, You Y, Li X, Ma T, Nie Y, Wei B et al. Brain injury does not alter the intrinsic differentiation potential of adult neuroblasts. J Neurosci 2009; 29: 5075–5087.
Li Y, Yu SP, Mohamad O, Genetta T, Wei L . Sublethal transient global ischemia stimulates migration of neuroblasts and neurogenesis in mice. Transl Stroke Res 2010; 1: 184–196.
Jin K, Minami M, Lan JQ, Mao XO, Batteur S, Simon RP et al. Neurogenesis in dentate subgranular zone and rostral subventricular zone after focal cerebral ischemia in the rat. Proc Natl Acad Sci USA 2001; 98: 4710–4715.
Liu F, Schafer DP, McCullough LD . TTC, fluoro-jade b and neun staining confirm evolving phases of infarction induced by middle cerebral artery occlusion. J Neurosci Methods 2009; 179: 1–8.
Chopp M, Zhang ZG, Jiang Q . Neurogenesis, angiogenesis, and mri indices of functional recovery from stroke. Stroke 2007; 38: 827–831.
Li WL, Yu SP, Ogle ME, Ding XS, Wei L . Enhanced neurogenesis and cell migration following focal ischemia and peripheral stimulation in mice. Dev Neurobiol 2008; 68: 1474–1486.
Feltenstein MW, Lambdin LC, Webb HE, Warnick JE, Khan SI, Khan IA et al. Corticosterone response in the chick separation-stress paradigm. Physiol Behav 2003; 78: 489–493.
Wilby MJ, Hutchinson PJ . The pharmacology of chlormethiazole: a potential neuroprotective agent? CNS Drug Rev 2004; 10: 281–294.
Lay CC, Davis MF, Chen-Bee CH, Frostig RD . Mild sensory stimulation reestablishes cortical function during the acute phase of ischemia. J Neurosci 2011; 31: 11495–11504.
Nilsson M, Perfilieva E, Johansson U, Orwar O, Eriksson PS . Enriched environment increases neurogenesis in the adult rat dentate gyrus and improves spatial memory. J Neurobiol 1999; 39: 569–578.
Rossi C, Angelucci A, Costantin L, Braschi C, Mazzantini M, Babbini F et al. Brain-derived neurotrophic factor (bdnf) is required for the enhancement of hippocampal neurogenesis following environmental enrichment. Eur J Neurosci 2006; 24: 1850–1856.
Eriksson PS, Perfilieva E, Bjork-Eriksson T, Alborn AM, Nordborg C, Peterson DA et al. Neurogenesis in the adult human hippocampus. Nat Med 1998; 4: 1313–1317.
van Praag H, Kempermann G, Gage FH . Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci 1999; 2: 266–270.
Hermann DM, Chopp M . Promoting brain remodelling and plasticity for stroke recovery: therapeutic promise and potential pitfalls of clinical translation. Lancet Neurol 2012; 11: 369–380.
Murphy TH, Corbett D . Plasticity during stroke recovery: from synapse to behaviour. Nat Rev Neurosci 2009; 10: 861–872.
Han X, Wang W, Xue X, Shao F, Li N . Brief social isolation in early adolescence affects reversal learning and forebrain bdnf expression in adult rats. Brain Res Bull 2011; 86: 173–178.
Barzilay R, Ben-Zur T, Sadan O, Bren Z, Taler M, Lev N et al. Intracerebral adult stem cells transplantation increases brain-derived neurotrophic factor levels and protects against phencyclidine-induced social deficit in mice. Transl Psychiatry 2011; 1: e61.
Groves JO . Is it time to reassess the bdnf hypothesis of depression? Mol Psychiatry 2007; 12: 1079–1088.
Hafner S, Emeny RT, Lacruz ME, Baumert J, Herder C, Koenig W et al. Association between social isolation and inflammatory markers in depressed and non-depressed individuals: results from the monica/kora study. Brain Behav Immun 2011; 25: 1701–1707.
Gainotti G, Antonucci G, Marra C, Paolucci S . Relation between depression after stroke, antidepressant therapy, and functional recovery. J Neurol Neurosurg Psychiatry 2001; 71: 258–261.
Jonas BS, Mussolino ME . Symptoms of depression as a prospective risk factor for stroke. Psychosom Med 2000; 62: 463–471.
Nibuya M, Morinobu S, Duman RS . Regulation of bdnf and trkb mrna in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 1995; 15: 7539–7547.
Shimizu E, Hashimoto K, Okamura N, Koike K, Komatsu N, Kumakiri C et al. Alterations of serum levels of brain-derived neurotrophic factor (bdnf) in depressed patients with or without antidepressants. Biol Psychiatry 2003; 54: 70–75.
Mattson MP, Maudsley S, Martin B . Bdnf and 5-ht: A dynamic duo in age-related neuronal plasticity and neurodegenerative disorders. Trends Neurosci 2004; 27: 589–594.
Baj G, D’Alessandro V, Musazzi L, Mallei A, Sartori CR, Sciancalepore M et al. Physical exercise and antidepressants enhance bdnf targeting in hippocampal ca3 dendrites: further evidence of a spatial code for bdnf splice variants. Neuropsychopharmacology 2012; 37: 1600–1611.
Heisler M, Halasyamani L, Cowen ME, Davis MD, Resnicow K, Strawderman RL et al. Randomized controlled effectiveness trial of reciprocal peer support in heart failure. Circ Heart Fail 2013; 6: 246–253.
Acknowledgements
We thank Dr Lauren Sansing for her help with the statistical analysis. This work was supported by the NIH (NINDS RO1 NSO77769 to LDM) and the AHA (1POST7430045 to VRV).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Venna, V., Xu, Y., Doran, S. et al. Social interaction plays a critical role in neurogenesis and recovery after stroke. Transl Psychiatry 4, e351 (2014). https://doi.org/10.1038/tp.2013.128
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tp.2013.128
Keywords
This article is cited by
-
The effect of group size, age and handling frequency on inter-male aggression in CD 1 mice
Scientific Reports (2020)
-
The “Use It or Lose It” Dogma in the Retina: Visual Stimulation Promotes Protection Against Retinal Ischemia
Molecular Neurobiology (2020)
-
Social environment mediates cancer progression in Drosophila
Nature Communications (2018)
-
Cell Therapy in Stroke—Cautious Steps Towards a Clinical Treatment
Translational Stroke Research (2018)
-
Aging alters the immunological response to ischemic stroke
Acta Neuropathologica (2018)