Abstract
The β-lactam antibiotic temocillin (6-α-methoxy-ticarcillin) shows stability to most extended spectrum β-lactamases, but is considered inactive against Pseudomonas aeruginosa. Mutations in the MexAB-OprM efflux system, naturally occurring in cystic fibrosis (CF) isolates, have been previously shown to reverse this intrinsic resistance. In the present study, we measured temocillin activity in a large collection (n = 333) of P. aeruginosa CF isolates. 29% of the isolates had MICs ≤ 16 mg/L (proposed clinical breakpoint for temocillin). Mutations were observed in mexA or mexB in isolates for which temocillin MIC was ≤512 mg/L (nucleotide insertions or deletions, premature termination, tandem repeat, nonstop, and missense mutations). A correlation was observed between temocillin MICs and efflux rate of N-phenyl-1-naphthylamine (MexAB-OprM fluorescent substrate) and extracellular exopolysaccharide abundance (contributing to a mucoid phenotype). OpdK or OpdF anion-specific porins expression decreased temocillin MIC by ~1 two-fold dilution only. Contrarily to the common assumption that temocillin is inactive on P. aeruginosa, we show here clinically-exploitable MICs on a non-negligible proportion of CF isolates, explained by a wide diversity of mutations in mexA and/or mexB. In a broader context, this work contributes to increase our understanding of MexAB-OprM functionality and help delineating how antibiotics interact with MexA and MexB.
Similar content being viewed by others
Introduction
Pseudomonas aeruginosa is the most prevalent pathogen isolated in the respiratory tract of adult cystic fibrosis (CF) patients and a major cause of morbidity and mortality in this population1. These patients are frequently exposed to antipseudomonal antibiotics and, as a result, become colonized by multidrug-resistant strains. Since therapeutic choices narrow, clinicians are increasingly forced to look after “forgotten” antibiotics against which resistance rates could be low because they were scarcely used2.
Temocillin (6-α-methoxy-ticarcillin) is one of these old antibiotics recently revived, based on a renewed interest for its activity against many β-lactam-resistant Enterobacteriaceae3. Temocillin, indeed, shows remarkable stability to most β-lactamases, including AmpC-type cephalosporinases and extended-spectrum β-lactamases (ESBLs) such as TEM, SHV, and CTX-M enzymes4. Temocillin also obtained an orphan drug designation for the treatment of Burkholderia cepacia complex infection in CF patients5. However, as temocillin was long considered as intrinsically inactive against P. aeruginosa, it was not included in conventional susceptibility testing of this organism when isolated from CF patients. Previous work from our laboratory actually showed that the intrinsic resistance of P. aeruginosa to temocillin was due to active efflux by the constitutively-expressed MexAB-OprM efflux transporter, while the other major efflux transporters were not involved6. Moreover, some isolates from CF patients regained susceptibility to temocillin because of natural mutations in the proteins constituting the MexAB-OprM efflux pump6. MexAB-OprM belongs to the Resistance Nodulation Division superfamily of efflux transporters. It is energized by proton motive force and consists of three proteins, namely an inner membrane exporter (homotrimer of MexB), an outer membrane gated channel (homotrimer of OprM), and a periplasmic linker (6 to 13-mer of MexA), the concerted action of which allows for the extrusion of substrates from the inner membrane or the periplasmic space directly out of the bacteria (ref. 7 for review). The role of porins in the capacity of temocillin to cross the outer membrane is unknown so far. Among porins described in P. aeruginosa, the cation-selective channel OprD (OccD1) is involved in the entry of carbapenems (most notably, imipenem), while the anion-specific channels OpdK (OccK1) and OpdF (OccK2) are involved in the entry of carboxypenicillins like carbenicillin and of cefoxitin8.
In this context, the objective of this study was to better document the mechanisms of intrinsic resistance and acquired susceptibility to temocillin in P. aeruginosa isolated from CF patients. We exploited a large collection of 333 isolates collected from 4 CF centres in Northern Europe, which has been partly characterized for its resistance to commonly used anti-pseudomonas agents9. We demonstrate that a non-negligible proportion of these isolates (~15–30%) showed low and clinically-exploitable MICs to temocillin, associated with a wide variety of mutations in mexA or mexB genes. No β-lactamase hydrolysing temocillin was found in the collection. A marginal role of anion-specific porins (OpdK and OpdF) in temocillin influx was also demonstrated. Besides its immediate interest for the management of infections in CF patients, this work also brings innovative pieces of information regarding mutations affecting the transport activity of the MexAB-OprM efflux system in P. aeruginosa, which can refine our understanding of the mechanism of substrate recognition and transport by this pump and may also help in reviving other antibiotics that are substrates of the same transporter.
Results
Activity of temocillin against clinical isolates and comparison with other β-lactams
Figure 1a shows the MIC distribution of temocillin compared to ticarcillin against the whole collection, with MIC50 and MIC90 of conventional antipseudomonal β-lactams illustrated in the accompanying Table. For temocillin, 15% and 29% of isolates had an MIC ≤ 8 and 16 mg/L, respectively. These values are close to those observed with ticarcillin (13% and 18%, respectively). Of interest, a similar proportion of isolates were susceptible to first-line antipseudomonal agents such as piperacillin, piperacillin/tazobactam, or ceftazidime. Carbapenems were active against a higher proportion of isolates.
Panel (a): Cumulative MIC distribution for temocillin (TMO) compared to ticarcillin (TIC), with indication of MIC50, MIC90 and percentage of susceptibility according to the interpretive criteria of EUCAST (S, susceptible; R, resistant) for ticarcillin (S ≤ 16 mg/L; R > 16 mg/L); piperacillin (PIP, S ≤ 16 mg/L; R > 16 mg/L); piperacillin-tazobactam (TZP, S ≤ 16 mg/L; R > 16 mg/L); ceftazidime (CAZ, S ≤ 8 mg/L; R > 8 mg/L); imipenem (IPM, S ≤ 4 mg/L; R > 8 mg/L); meropenem (MEM, S ≤ 2 mg/L; R > 8 mg/L). A value of 16 mg/L (dotted line in the graph; EUCAST susceptibility breakpoint of ticarcillin) has been considered as cut-off value for temocillin (TMO), for comparison purposes. Panels (b–e): cross-resistance between TMO and other β-lactams. Correlation between MICs of TMO (abscissa) and TIC, CAZ, TZP or MEM (ordinate) for each individual isolate in the collection using quantile density contour analysis (JMP® version 12.1.0). The intensity of each zone (from warm to cold colours) is indicative of the proportion of isolates (from large to small) with MICs at the corresponding coordinates. The broken lines point to the MIC value above which the isolates are considered resistant for TIC, CAZ, TZP and intermediate for MEM according to EUCAST interpretive criteria. A value of 16 mg/L has been considered for TMO. The percentage of isolates is indicated in each quadrant of the figures. MICs values are expressed as the log2 of their value.
Figure 1b–e shows cross-resistance between temocillin and the other β-lactams for individual isolates. While a high proportion of isolates were cross-resistant to temocillin and ticarcillin (64%), piperacillin/tazobactam (53%), ceftazidime (48%), or meropenem (40%), a small but meaningful proportion of isolates that were resistant to the comparator (ranging from 11% for meropenem to 18% for ticarcillin) remained susceptible to temocillin.
β-lactamases screening and identification
β-lactamase production is a main mechanism of β-lactam resistance in P. aeruginosa. We therefore screened the collection for extended spectrum β-lactamases (ESBLs) and carbapenemases. No carbapenemases were detected using both phenotypic and genotypic methods. Moreover, detection of genes encoding CTX-M, TEM, SHV, and BEL, PER, GES, VEB, or OXA β-lactamases returned negative results in all isolates that were simultaneously resistant to CAZ and MEM (MIC > 8 mg/L).
Influence of active efflux on temocillin activity
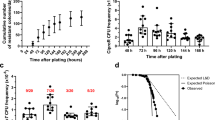
Previous studies suggested that active efflux by MexAB-OprM plays a major role in P. aeruginosa resistance to temocillin6. Using a representative subset of isolates (n = 124) selected to cover the whole range of MICs (8–14 isolates for each MIC value), we therefore examined the influence of the broad spectrum efflux pump inhibitor Phe-Arg-β-naphthylamide (PAβN10) on temocillin activity. We checked that PAβN was not toxic by itself for these isolates in the conditions of the experiment. MICs were shifted towards lower values in the presence of the inhibitor (Fig. 2a), with its effect being particularly marked for isolates showing MICs ranging between 128 and 512 mg/L in the absence of PAβN (Fig. 2b). To confirm the role of MexAB-OprM-mediated efflux in resistance to temocillin, we followed in real-time the efflux of N-phenyl-1-naphthylamine (NPN), a preferential MexAB-OprM substrate11, using temocillin as a competitor and comparing PAO1 to its MexAB-OprM deletion mutant PAO1mexAB (Fig. 2c). NPN efflux was much slower in the deletion mutant than in the parent strain. Temocillin was able to decrease the rate of efflux of NPN in PAO1 but had no effect in the MexAB-OprM deletion mutant even when using a temocillin concentration much higher than its MIC (2 mg/L). These data strongly suggest that temocillin is a preferential substrate of MexAB-OprM. The rate of efflux of NPN was therefore evaluated in 32 isolates (29 clinical isolates plus PAO1, PT629, PAO1mexAB) harbouring increasing temocillin MICs (Fig. 2d). NPN efflux was significantly slower for isolates with a temocillin MIC < 512 mg/L and increased in proportion to temocillin MIC.
(a) Cumulative MIC distribution of temocillin in a subset of the collection (n = 124) selected to cover the whole range of MICs and influence of the efflux pump inhibitor PAβN (20 mg/L). (b) Fold reduction (log2 scale) in temocillin MIC in the presence of PAβN, according to temocillin MICs for the same isolates. The graph shows the box and whiskers plot with 10-90 percentiles, with the red line connecting the medians. (c) Kinetics of NPN efflux from PAO1 or PAO1mexAB in the absence (control) or presence of temocillin (TMO) at the indicated concentrations. Vmax are expressed in reduction in the fluorescence signal per second. (d) Kinetics of efflux of NPN as a function of temocillin MIC for the 32 isolates for which mexA and mexB were sequenced (Table 1). The ordinate is expressed as the Vmax (arbitrary fluorescence units). R2 for a one-phase association: 0.9043. Node splitting value for slower efflux: MIC of 256 mg/L or lower (LogWorth statistic: 29.5157 [p < 0.001]). (e,f) MIC and rate of NPN efflux in sequenced isolates classified according to the type of mutations observed in mexA and mexB (Table 1). ‘Truncated/aberrant’ refer to deletions of more than 3 consecutive nucleotides in the sequence, nonstop mutations, insertion of minisatellites or aberrant signal peptides. ‘Missense mutations’ refer to mutations leading to the replacement of at least one amino acid in the corresponding protein. The graphs shows individual values together with means and SD. Statistical analysis (1-way ANOVA; Tuckey post-hoc test): data series with different letters are different from one another (p < 0.05).
Mutations in mexA and mexB in relation with temocillin MIC and efflux pump activity
MexA and MexB, but not OprM, determine the β-lactam specificity of the MexAB-OprM efflux system12. We therefore sequenced mexA and mexB genes in the 32 isolates for which NPN efflux was evaluated. Figure 2e,f illustrates the relationship between the type of mutations evidenced and the MIC of temocillin or the rate of efflux of NPN. Isolates harboring deletions in mexA or mexB or aberrant sequences (deletion of ≥3 consecutive nucleotides, nonsense mutation, nonstop mutation13, aberrant signal peptide, minisatellite repeat) had the slowest rate of efflux of NPN and temocillin MICs ranging between 2 and 128 mg/L. Isolates with missense mutations globally showed a slower efflux than PAO1 and temocillin MICs of 128 or 256 mg/L. Synonymous mutations did not impact NPN efflux or temocillin MICs. In addition, most of the isolates showed a high frequency of synonymous mutations and codon degeneracy especially at the third position. Most of them were conserved between different isolates originating from different countries.
Table 1 summarizes these results and shows in parallel for each isolate the Vmax for NPN efflux, together with (i) the MICs of temocillin in control conditions or in the presence of the efflux pump inhibitor PAβN or of the OpdK porin substrate vanillate, and (ii) the relative amount of exopolysaccharides. The constructed molecular graphics of the corresponding proteins are illustrated in Table S2 (see also Fig. S1 for a view of the wild-type proteins, with specification of their subdomains).
Tables S2b–e specifically focus on sequenced isogenic isolates collected either from the same patient or from different patients originating either from the same or from different countries. Table S2b illustrates isolates collected in the UK and belonging to the Liverpool epidemic strain (LES)9. Isolates from five different patients with an MIC of 16 mg/L shared the same synonymous mutations and also the same nonsense mutation in mexA leading to the production of a 119 amino acid protein. Of note, two other LES isolates with an MIC of 32 mg/L shared the same synonymous mutations in mexA and mexB genes as those described for the first 5 LES isolates, but harboured different deletions in their mexB gene, leading to proteins of 30 and 672 amino acids, respectively. Table S2c illustrates 2 pairs of isolates harbouring the same mutations although collected from different patients in the same country. Thus, in the isogenic isolates 191-4 and 2079 with a temocillin MIC of 64 mg/L, MexA was truncated to 27 amino acids. Likewise, in isogenic isolates W024 and W049 with a MIC of 4 mg/L, MexA was truncated to 69 residues due to the loss of 1 nucleotide leading to a premature stop codon. Interestingly also, isogenic isolates can be found in different countries (Table S2d). These three isolates belonging to the same multidrug-resistant clonal complex9 and originating from the UK (AG3), Belgium (128) and Germany (129-6) shared the same synonymous mutations. Only AG3 had a low MIC to temocillin (8 mg/L), associated with a MexA protein truncated in its MP domain (372 amino acids left). The two other isolates had MICs of 1024 mg/L, with one conservative missense mutation (L376V) found in the transmembrane domain TM3 of the MexB protein of isolate 128. Finally, Table S2e illustrates three other clonal isolates differing in their susceptibility to temocillin. They come from two different patients in Germany and share (i) a radical missense mutation (G616T) in the mexA part encoding the third domain of MexA interacting with the distal domain of MexB, (ii) a conservative missense mutation (A556G), and (iii) a deletion of nucleotides 1947 and 1948 in mexB. In addition, isolate 135-1, with a temocillin MIC of 8 mg/L, showed an insertion in mexB (G1261_C1262insG) leading to the production of an aberrant and truncated MexB protein. In contrast, isolates 208-3 and 208-2, with temocilllin MIC of 256 and 512 mg/L, differed only by the position of an additional 1 nucleotide deletion (in position 1854 or 1889, respectively) in the mexB gene part encoding the pore (PC1) subdomain, leading to the replacement of 30 by 29 amino acids, and of 19 by 18 amino acids, respectively. This difference could explain the 1 two-fold dilution difference in temocillin MIC between these two isolates.
Despite the evidenced truncations or mutations in MexA and MexB, most of the clinical isolates showed nevertheless higher temocillin MICs than the reference strain PAO1mexAB lacking MexA and MexB proteins (see Fig. 2e). This strongly suggests that other mechanisms of resistance independent of the functionality of MexAB-OprM are present.
Culture content in extracellular polymeric saccharides with β-1, 4 and β-1, 3 linkages
P. aeruginosa isolated from CF patients often harbour a mucoid phenotype, related to the overproduction of extracellular polysaccharides, among which alginate contributes to the chronicity of infections. Alginate production has been shown to impede the activity of β-lactams, including ticarcillin14,15.
We therefore compared exopolysaccharide abundance in cultures of 73 clinical isolates (included those which had been sequenced) vs. PAO1 by measuring calcofluor white binding to polysaccharides (Fig. 3). A high variability in fluorescence values was observed among isolates, but a global statistical analysis showed that the increase in fluorescence signal was significant for isolates with a temocillin MIC ≥ 4 mg/L. Notably, among sequenced isolates with truncated or aberrant MexA or MexB, the fluorescence signal was at least 2.6-fold higher than in PAO1 for those for which temocillin MICs are between 32 to 128 mg/L.
Calcofluor white (CFW) fluorescence was measured for 73 clinical isolates, including those for which mexA and mexB were sequenced (Table 1). Data expressed as a ratio to the value measured for the non-mucoid reference strain PAO1 (highlighted by the dotted line), the MIC of which is 512 mg/L). The red horizontal lines show the mean of values for 7–8 isolates for each MIC (3 only for 32 mg/L [no more isolates with this MIC value in the whole collection]). Statistical analysis: figures above each bar represents the p value (multiple t-test) versus PAO1. Node splitting value for significant increase in CFW fluorescence signal vs. PAO1: MIC = 4 mg/L or higher LogWorth statistic: 1.6343 [p 0.02].
Influence of porins on temocillin activity
β-lactams are known to cross the outer membrane of Gram-negative bacteria via porins.
While OprD (OccD1) has been well characterized as facilitating the entry of carbapenems (mainly imipenem16) into P. aeruginosa, other members of the OprD family like OpdK (OccK1, archetype of the OccK subfamily17) and OpdF (OccK2) are involved in the transport of the carboxypenicillin carbenicillin and of the anionic cephalosporin cefoxitin8. As temocillin is also a carboxypenicillin, we examined whether these anion-selective porins were involved in temocillin transport into P. aeruginosa. OprD was studied in parallel. Other members of the OprD family of porins were not investigated in the present work.
MICs of temocillin, cefoxitin, imipenem, and meropenem were measured in transformants of a porin-deficient E. coli strain expressing OpdK, OpdF, or OprD (Table 2; carbenicillin could not be tested due to the presence of a β-lactamase-encoding gene on the transforming plasmid). Consistent with previous studies8, OprD expression increased the activity of carbapenems but not that of the other drugs. Conversely, the expression of OpdK, and to a slightly lesser extent, of OpdF, increased the activity of temocillin and cefoxitin; this effect was best seen when the antibiotics were combined with PAβN.
Subsequently, MICs were measured in P. aeruginosa mutants of each of these three porins (Table 3). MICs for carbapenems were markedly (3–4 log2 dilutions) increased by the loss of OprD while those of temocillin and carbenicillin were not affected. Conversely, deletion of OpdK or OpdF modestly (<1 log2 dilution) increased the MIC of carboxypenicillins. This effect was amplified in the presence of vanillate (substrate for OpdK17,18) in the OpdF mutant for carboxypenicillins, suggesting that these two porins cooperate for the import of these antibiotics. Likewise, L-Arginine (substrate for OprD19) reduced carbapenem activity in the OpdK or OpdF mutants. The effect of vanillate on temocillin activity against clinical isolates is illustrated in Fig. 4a (MIC distribution) and in Table 1 (individual isolates). This effect was modest, but best seen in isolates with temocillin MICs ≤ 64 mg/L, in which efflux was deficient (Fig. 4b).
(a) Cumulative MIC distribution of temocillin in a subset of the collection (n = 31) selected to cover the whole range of MICs and influence of the OpdK competitor vanillate (VNL 2 mM). (b) Fold reduction (log2 scale) in temocillin MIC in the presence of VNL, according to temocillin MICs for the same isolates. The graph shows the box and whiskers plot with 10–90 percentiles, with the red line connecting the medians.
Discussion
This study is the first to describe the activity of temocillin in a large collection of P. aeruginosa collected from CF patients, and to determine the mechanisms that can lead to a phenotype of ‘acquired susceptibility’ to this antibiotic, which is uncommon in this pathogen3. Our results showed that a low but significant proportion of the isolates have MICs lower than the current temocillin BSAC (British Society for Antimicrobial Chemotherapy) susceptibility breakpoint for systemic infections, and to the current EUCAST breakpoint for the parent molecule ticarcillin, as well as to the breakpoint proposed for high dosing regimens of temocillin based on pharmacokinetic/pharmaocodynamic considerations20. Even if far from being the majority, these susceptible isolates cannot be ignored, as 30 to 60% of them are resistant to conventional antipseudomonal β-lactams, including carbapenems, making temocillin one of the last viable therapeutic option.
We confirm that altered active efflux mediated by MexAB-OprM is the main driver of restored susceptibility to temocillin. Non-functional efflux, as evidenced using NPN as a tracer, was observed in isolates with temocillin MICs < 512 mg/L, and these also harbour mutations in mexA and/or mexB leading to protein alterations. Among the sequenced isolates, we could evidence a whole panel of mutations that may lead to loss of functionality, many of which had not been reported in our earlier study of temocillin-susceptible CF isolates6, neither in other studies investigating the effect of mexA/B mutations on the pump activity21,22,23.
The most susceptible isolates show major deletions in mexA or mexB leading to the production of truncated or aberrant proteins. Over the 8–512 mg/L range of MICs, mexA mutations are found in the regions encoding the α-helical hairpin and/or of the second, third, and membrane-proximal (MP) domains, which interact with MexB and/or OprM proteins24, possibly affecting the linkage with MexB or OprM, and therefore the correct assembly of the tripartite efflux system22. mexB mutations are located either in the regions building the pore domain (PC1, PC2, PN1, and PN2) implicated in drug recognition and extrusion25, or in those encoding the periplasmic tip of the docking subdomains (DN and DC) involved in the interaction with OprM25. Only one isolate (3319) showed an additional amino acid substitution outside these domains, namely in the cytoplasmic C-terminal domain of MexB25. Deletions in transmembrane helices TM8-12 were also identified in a few isolates. TM10 has been shown to mediate proton translocation (with the TM4) in the inner membrane domains26. Of interest, we noticed different mutations and hence, different susceptibility to temocillin, among clonal isolates, suggesting a high adaptability of each of them to its own environment.
Of note, most of the isolates also showed a high frequency of synonymous mutations and codon degeneracy especially at the third position. Most of these mutations were conserved among several isolates from different countries. Synonymous substitutions are often considered as silent mutations, but they can impact gene transcription and mRNA transport or translation, which could alter protein folding and function27. Codon degeneracy is described as highly frequent in P. aeruginosa28.
Intriguingly, restoration of temocillin activity was only partial in some isolates that showed major deletions in mexA or mexB. Since we did not investigate the impact of each evidenced mutation on temocillin MIC in a PAO1 background, we cannot exclude that this is due to the expression of other, still undescribed mechanisms of resistance, or to an overexpression of other efflux systems that may compensate for the loss of MexAB-OprM activity, as previously described in CF isolates29. However, we previously showed that these systems only play a marginal role in temocillin extrusion6. Therefore, we suggest a possible phenotypic resistance related to an overproduction of extracellular polymeric substances, including alginate, in isolates with temocillin MICs ≥ 4 mg/L. Alginate has been shown to impair the diffusion, and therefore the activity, of ticarcillin, the parent compound of temocillin14,15. We show here that alginate production is systematically low in isolates with a temocillin MIC of 2 mg/L, i.e. a value corresponding to that measured for the mexAB deletion mutant of the non-mucoid reference PAO1, while it is high in clinical isolates with inactive efflux but temocillin MICs of 32–128 mg/L. Of note, we were able to exclude or minimize the role of two known mechanisms of resistance to β-lactams, namely the expression of β-lactamase(s) and reduced expression of porins. Thus, no β-lactamase among the few ones capable of hydrolysing temocillin (VIM, NDM, IMP, OXA-4830) was genotypically detected in our isolates. For porins, while OprD alterations have been previously evidenced in this collection31, we show here that, in contrast to carbapenems, temocillin does not require this porin to enter bacteria, but rather uses the anion-specific porins, OpdK and OpdF, the role of which seems redundant. Temocillin indeed shares a methoxy moiety with cefoxitine and vanillate, two well-established substrates of these porins. This moiety has been described for vanillate as interacting with the L3-L7 loops forming the internal constriction of OpdK18. Yet, the importance of these porins in temocillin uptake seems rather marginal since their inhibition increases temocillin MIC of 1 two-fold dilution only. For this reason, we did not specifically look for mutations or reduced expression of these porins in the collection. We cannot exclude that other porins expressed by P. aeruginosa32 that were not investigated here also play a role in temocillin uptake. We acknowledge this is a limitation of this work.
Considering all data, and despite remaining uncertainty regarding the reason for variable susceptibility to temocillin in mutated isolates, our work brings three major pieces of scientific progress. Firstly, from a clinical perspective, we demonstrate a potential use for temocillin in the management of P. aeruginosa infections in CF patients. Although limited to a small proportion of isolates, this is important as temocillin susceptibility was observed for some isolates that were resistant to other β-lactams, either by production of β-lactamases or by alteration of the OprD porin, as well as in isolates belonging to clones described as multidrug-resistant. Thus our data call for introducing temocillin in routine susceptibility testing for P. aeruginosa collected from CF patients. Moreover, temocillin may offer the advantage of acting at the same time on Burkholderia spp5 that can co-infect CF patients.
Second, in a context of drug design, we observed a large diversity of mutations in mexA and mexB, opening perspectives for studying their role in substrate recognition and/or transport activity. The mutants described here could also be used to study the efflux of other antibiotics and to help delineating how these interact with specific domains of MexA and MexB. Third, from an ecological perspective, our data reinforce the concept that a functional MexAB-OprM pump is not needed for P. aeruginosa survival in the CF lung29 and illustrates the high adaptability of this bacteria to its environment.
Materials and Methods
P. aeruginosa isolates
A total of 333 isolates were randomly collected from 155 patients in four CF centres from different countries (Hôpital des enfants malades Reine Fabiola/Erasme; Belgium, n = 88; Hôpital Jean Minjoz, Besançon, France; n = 80; University Hospital of Münster, n = 66, Germany; Queen’s University of Belfast, UK, n = 99) during routine periodic visits (Table S1). PAO1, mutants thereof overexpressing or deleted for MexAB-OprM (Table 1), and PA14 were used as control strains. The molecular typing of part of this collection has been previously performed by pulse field gel electrophoresis, focusing on isolates collected simultaneously from the same patients but differing in their susceptibility profile9. It was completed here for selected sequenced isolates following the same procedure.
Antibiotics
The following antibiotics were obtained as microbiological standard from Sigma-Aldrich, St Louis, MO (sodium salts, potency in brackets): ticarcillin (TIC; 85.25%), piperacillin (PIP; 94.20%), carbenicillin (CAR; 89.16%), and cefoxitin (FOX; potency, 95.11%). The remaining antibiotics were obtained as powder for parenteral use from their corresponding manufacturers (potency in brackets): temocillin (TMO, 78.12%) as Negaban® from Eumedica (Manage, Belgium), piperacillin-tazobactam (TZP; 97.00%) as Tazocin® from Wyeth (Louvain-La-Neuve, Belgium), ceftazidime (CAZ, 88.20%) as Glazidim® from Glaxo-SmithKline (Genval, Belgium), imipenem (IPM, 45.60% [due to the presence of cilastatin in the powder]) as Tienam® from MSD (Brussels, Belgium), and meropenem (MEM, 74.00%) as Meronem® from AstraZeneca (Brussels, Belgium).
Susceptibility testing
MICs were determined by broth microdilution following the guidelines from the Clinical and Laboratory Standards Institute and using P. aeruginosa ATCC27853 as a quality control. Susceptibility was assessed according to the interpretive criteria of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) or using a cut-off value of 16 mg/L for temocillin (no EUCAST breakpoint) by analogy with the current susceptibility breakpoint of its parent compound ticarcillin. For specific isolates, MICs were also measured in the presence of 20 mg/L Phe-Arg-β-naphthylamide dihydrochloride (PAβN, broad-spectrum efflux pump inhibitor10; Sigma-Aldrich) and of 1 mM MgSO4 (to prevent the outer membrane permeabilization caused by PAβN33) or in the presence of substrates of P. aeruginosa porins, namely vanillate (VNL, OpdK substrate18), or L-arginine (L-Arg, OprD substrate19), both obtained from Sigma-Aldrich.
β-lactamases screening and identification
Isolates showing MICs > 8 mg/L for both ceftazidime and meropenem (n = 53) were screened for the presence of genes encoding acquired metallo-β-lactamases (VIM, IMP, NDM), carbapenemases (OXA-48, KPC), or widespread extended-spectrum β-lactamases (ESBLs) by PCR, as previously described9. The expression of carbapenemase(s) was also screened using a phenotypic method described as the Carbapenemase Nordmann-Poirel (Carba NP) test34.
Sequencing of mexA and mexB
mexA and mexB genes were amplified by PCR using the following primers: mexA-F (5′-GCGAGGCTTTCGGACGTTTA-3′); mexA-R (5′-GGCAGACTGAGGATCGACA-3′), mexB1-F (5′-CAAGGGGATTCGTAATGTC-3′); mexB1-R (5′-GTGAACATCCAGATCATCC-3′), mexB2-F (5′-CGGATGTTCCTTTCCACCAC-3′); mexB2-R (5′-GACAGAACGACAGCGGCTA-3′). Annealing temperatures for each pair of primers and amplicon sizes were as follows: mexA, 60 °C, 1375 bp; mexB1, 61 °C, 1684 bp; mexB2, 67 °C, 1638 bp. Sanger sequencing was performed in forward and reverse directions at GATC Biotech (Konstanz, Germany) using the same primers. PAO1 was used as a reference. Molecular graphics for MexA and MexB proteins were rendered using Visual Molecular Dynamics program (VMD35), based on Protein Data Bank (PDB) files 2V4D (MexA24), and 2V50 (MexB25).
Direct measurement of efflux activity
Efflux pump functionality was assayed by following the kinetics of efflux of N-phenyl-1-naphthylamine (NPN; Sigma-Aldrich), a MexAB-OprM preferential substrate11, which is fluorescent when incorporated in bacterial membranes. As previously described36, bacteria were treated with carbonyl cyanide m-chlorophenylhydrazone (CCCP, 10 μM; Sigma-Aldrich) and then loaded with 20 μM NPN. Efflux was initiated by energizing bacteria with 50 mM D-glucose (Sigma-Aldrich). Decay in fluorescence signal was followed over time using a Spectramax® multiplate reader (Molecular devices, Sunnyvale, CA). Maximal velocity (Vmax) was calculated by the SoftMax® Pro Microplate Data Acquisition and Analysis Software (version 6.2). PAO1mexAB (ΔmexAB-mutant) was used as negative control.
Cloning and overexpression of porins
Genes encoding different porins i.e. OprD (OccD1), OpdK (OccK1), and OpdF (OccK2) were cloned in a heterologous system of Escherichia coli, which was lacking the main native porins OmpC and OmpF (E.coli K-12 W3110:ΔompFΔompC)37. The genes opdK and opdF were PCR amplified from PAO1 (with HindIII and NsiI restriction site overhangs) and further cloned into pGOmpF (a vector with the signal sequence of phoE [phosphoporin protein E] attached to the ompF gene38) via HindIII/PstI, thereby replacing only ompF. This resulting vectors (pG-OpdK or pG-OpdF) had thus opdK and opdF attached to the phoE signal sequence and was further under the control of the lac promoter. The expression of opdK or opdF was inducible by 0.5 mM IPTG. The pB22-OprD construct was a kind gift from Prof. Bert van den Berg, Newcastle, UK. In this E. coli expression vector, the oprD gene with the signal sequence of the E. coli outer membrane protein YtfM is under the control of the PBAD promoter inducible by 0.5% (w/v) arabinose. MICs were determined in the presence of inducer.
Single-porin P. aeruginosa mutants
Insertion mutants for the same 3 porins were obtained from the PA14 transposon insertion mutant library from the Harvard Medical School, Boston, MA39. This transposon library has been previously characterized and sequenced in order to identify insertion sites40; all transformants were resistant to gentamicin, confirming the expression of the gentamicin-resistance gene present on the insertion cassette.
Quantification of extracellular polymeric saccharides (EPS)
EPS were quantified using calcofluor white (BD™ Diagnostics, Sparks Glencoe, MD), a fluorophore which binds to β-(1, 4) and β-(1, 3) polysaccharides41, including alginate (co-polymer of O-acetylated β-(1, 4) D-mannuronic acid and L-guluronic acid), a major constituent of slime produced by mucoid P. aeruginosa. Bacteria were cultured in 96-well plates as for MIC determinations (starting inoculum, 106 CFU/mL; 24 h incubation), after which bacterial density was evaluated by measuring OD620nm. The content of the wells was transferred to Eppendorf tubes and calcofluor white at a final concentration of 30 mg/L was added. The tubes were incubated for 30 minutes at room temperature and subsequently centrifuged at 20,800 g. Pellets were washed with phosphate buffered saline (PBS pH 7.4), resuspended in the same buffer, and transferred in black 96-well plates to measure fluorescence in a Spectramax® multiplate reader (λexc 370 nm; λem 440 nm). Fluorescence values were normalized vs. the OD620nm of the cultures, with the value measured for PAO1 set to 1 as a reference.
Statistical analyses
Statistical analysis was performed using JMP® version 12.1.0, SAS Institute Inc., Cary, NC, USA or GraphPad Prism version 7.01 (GraphPad software Inc., San Diego, CA).
Ethics
The protocol for this study has been examined by the ethical committee of the Université catholique de Louvain, who determined that it did not fall under the scope of the law on human experimentation as (i) all isolates were collected during routine sampling and assembled retrospectively, and (ii) all patients’ data were anonymized.
Additional Information
How to cite this article: Chalhoub, H. et al. Mechanisms of intrinsic resistance and acquired susceptibility of Pseudomonas aeruginosa isolated from cystic fibrosis patients to temocillin, a revived antibiotic. Sci. Rep. 7, 40208; doi: 10.1038/srep40208 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Elborn, J. S. Cystic fibrosis. Lancet 388, 2519–2531 (2016).
Theuretzbacher, U. et al. Reviving old antibiotics. J. Antimicrob. Chemother. 70, 2177–2181 (2015).
Livermore, D. M. & Tulkens, P. M. Temocillin revived. J. Antimicrob. Chemother. 63, 243–245 (2009).
Glupczynski, Y. et al. In vitro activity of temocillin against prevalent extended-spectrum beta-lactamases producing Enterobacteriaceae from Belgian intensive care units. Eur. J. Clin. Microbiol. Infect. Dis. 26, 777–783 (2007).
Kent, L. et al. Temocillin in cystic fibrosis: a retrospective pilot study. J. Cyst. Fibros. 7, 551–554 (2008).
Buyck, J. M., Guenard, S., Plesiat, P., Tulkens, P. M. & Van Bambeke, F. Role of MexAB-OprM in intrinsic resistance of Pseudomonas aeruginosa to temocillin and impact on the susceptibility of strains isolated from patients suffering from cystic fibrosis. J. Antimicrob. Chemother. 67, 771–775 (2012).
Dreier, J. & Ruggerone, P. Interaction of antibacterial compounds with RND efflux pumps in Pseudomonas aeruginosa. Front Microbiol. 6, 660 (2015).
Eren, E. et al. Substrate specificity within a family of outer membrane carboxylate channels. PLoS. Biol. 10, e1001242 (2012).
Mustafa, M.-H. et al. Antimicrobial susceptibility of Pseudomonas aeruginosa isolated from Cystic Fibrosis patients through Northern Europe. Antimicrob. Agents Chemother. 60, 6735–6741 (2016).
Lomovskaya, O. et al. Identification and characterization of inhibitors of multidrug resistance efflux pumps in Pseudomonas aeruginosa: novel agents for combination therapy. Antimicrob. Agents Chemother. 45, 105–116 (2001).
Ocaktan, A., Yoneyama, H. & Nakae, T. Use of fluorescence probes to monitor function of the subunit proteins of the MexA-MexB-oprM drug extrusion machinery in Pseudomonas aeruginosa. J. Biol. Chem. 272, 21964–21969 (1997).
Srikumar, R., Li, X. Z. & Poole, K. Inner membrane efflux components are responsible for beta-lactam specificity of multidrug efflux pumps in Pseudomonas aeruginosa. J. Bacteriol. 179, 7875–7881 (1997).
Klauer, A. A. & van Hoof, A. Degradation of mRNAs that lack a stop codon: a decade of nonstop progress. Wiley. Interdiscip. Rev RNA. 3, 649–660 (2012).
Bolister, N., Basker, M., Hodges, N. & Marriott, C. Reduced susceptibility of a mucoid strain of Pseudomonas aeruginosa to lysis by ticarcillin and piperacillin. J. Antimicrob. Chemother. 24, 619–621 (1989).
Bolister, N., Basker, M., Hodges, N. A. & Marriott, C. The diffusion of beta-lactam antibiotics through mixed gels of cystic fibrosis-derived mucin and Pseudomonas aeruginosa alginate. J. Antimicrob. Chemother. 27, 285–293 (1991).
Quinn, J. P., Darzins, A., Miyashiro, D., Ripp, S. & Miller, R. V. Imipenem resistance in pseudomonas aeruginosa PAO: mapping of the OprD2 gene. Antimicrob. Agents Chemother. 35, 753–755 (1991).
Liu, J. et al. OccK channels from Pseudomonas aeruginosa exhibit diverse single-channel electrical signatures but conserved anion selectivity. Biochemistry 51, 2319–2330 (2012).
Wang, Y., Zhao, X., Sun, B., Yu, H. & Huang, X. Molecular dynamics simulation study of the vanillate transport channel of Opdk. Arch. Biochem. Biophys. 524, 132–139 (2012).
Tamber, S. & Hancock, R. E. W. Involvement of two related porins, OprD and OpdP, in the uptake of arginine by Pseudomonas aeruginosa. FEMS Microbiol. Lett. 260, 23–29 (2006).
Laterre, P. F. et al. Temocillin (6 g daily) in critically ill patients: continuous infusion versus three times daily administration. J. Antimicrob. Chemother. 70, 891–898 (2015).
Middlemiss, J. K. & Poole, K. Differential impact of MexB mutations on substrate selectivity of the MexAB-OprM multidrug efflux pump of Pseudomonas aeruginosa. J. Bacteriol. 186, 1258–1269 (2004).
Nehme, D., Li, X. Z., Elliot, R. & Poole, K. Assembly of the MexAB-OprM multidrug efflux system of Pseudomonas aeruginosa: identification and characterization of mutations in mexA compromising MexA multimerization and interaction with MexB. J. Bacteriol. 186, 2973–2983 (2004).
Ohene-Agyei, T., Lea, J. D. & Venter, H. Mutations in MexB that affect the efflux of antibiotics with cytoplasmic targets. FEMS Microbiol. Lett. 333, 20–27 (2012).
Symmons, M. F., Bokma, E., Koronakis, E., Hughes, C. & Koronakis, V. The assembled structure of a complete tripartite bacterial multidrug efflux pump. Proc. Natl. Acad. Sci. USA 106, 7173–7178 (2009).
Sennhauser, G., Bukowska, M. A., Briand, C. & Grutter, M. G. Crystal structure of the multidrug exporter MexB from Pseudomonas aeruginosa. J. Mol. Biol. 389, 134–145 (2009).
Guan, L. & Nakae, T. Identification of essential charged residues in transmembrane segments of the multidrug transporter MexB of Pseudomonas aeruginosa. J. Bacteriol. 183, 1734–1739 (2001).
Goymer, P. Synonymous mutations break their silence. Nat Rev Genet 8, 92 (2007).
Kiewitz, C. & Tummler, B. Sequence diversity of Pseudomonas aeruginosa: impact on population structure and genome evolution. J. Bacteriol. 182, 3125–3135 (2000).
Vettoretti, L. et al. Efflux unbalance in Pseudomonas aeruginosa isolates from cystic fibrosis patients. Antimicrob. Agents Chemother. 53, 1987–1997 (2009).
Livermore, D. M. et al. What remains against carbapenem-resistant Enterobacteriaceae? Evaluation of chloramphenicol, ciprofloxacin, colistin, fosfomycin, minocycline, nitrofurantoin, temocillin and tigecycline. Int J. Antimicrob. Agents 37, 415–419 (2011).
Chalhoub, H. et al. High-level resistance to meropenem in clinical isolates of Pseudomonas aeruginosa in the absence of carbapenemases: role of active efflux and porin alterations. Int J. Antimicrob. Agents in the press (2016).
Hancock, R. E. W. & Brinkman, F. S. L. Function of pseudomonas porins in uptake and efflux. Annu. Rev Microbiol. 56, 17–38 (2002).
Lamers, R. P., Cavallari, J. F. & Burrows, L. L. The efflux inhibitor phenylalanine-arginine beta-naphthylamide (PAbetaN) permeabilizes the outer membrane of gram-negative bacteria. PLoS. One. 8, e60666 (2013).
Poirel, L. & Nordmann, P. Rapidec Carba NP Test for Rapid Detection of Carbapenemase Producers. J. Clin. Microbiol. 53, 3003–3008 (2015).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Iyer, R. & Erwin, A. L. Direct measurement of efflux in Pseudomonas aeruginosa using an environment-sensitive fluorescent dye. Res. Microbiol. 166, 516–524 (2015).
Bachmann B. J. Derivations and genotypes of some mutant derivatives of Escherichia coli K-12, in Escherichia coli and Salmonella typhimurium. In Escherichia coli and Salmonella typhimurium cellular and molecular biology, 2nd ( Neidhardt, F. C., Curtis III, R., Ingraham, J. L., Lin, E. C. C., Low, K. B. J., Magasani, B., Reznikoff, W. S., Riley, M., Schaechter, M., Umbarger, H. E. Eds) pp. 2460–88 ASM Press, Washington, DC (1996).
Prilipov, A., Phale, P. S., Van Gelder, P., Rosenbusch, J. P. & Koebnik, R. Coupling site-directed mutagenesis with high-level expression: large scale production of mutant porins from E. coli. FEMS Microbiol. Lett. 163, 65–72 (1998).
Libarti, N. T. et al. PA14 transposon insertion mutant library http://ausubellab.mgh.harvard.edu/cgi-bin/pa14/home.cgi (2015).
Liberati, N. T. et al. An ordered, nonredundant library of Pseudomonas aeruginosa strain PA14 transposon insertion mutants. Proc. Natl. Acad. Sci. USA 103, 2833–2838 (2006).
Chen, M. Y., Lee, D. J., Tay, J. H. & Show, K. Y. Staining of extracellular polymeric substances and cells in bioaggregates. Appl. Microbiol. Biotechnol. 75, 467–474 (2007).
Vergnaud, G. & Denoeud, F. Minisatellites: mutability and genome architecture. Genome Res. 10, 899–907 (2000).
Moyano, A. J., Feliziani, S., Di Rienzo, J. A. & Smania, A. M. Simple sequence repeats together with mismatch repair deficiency can bias mutagenic pathways in Pseudomonas aeruginosa during chronic lung infection. PLoS. One. 8, e80514 (2013).
Acknowledgements
This work was supported in part by the Belgian Région Wallonne (program TEMOEXPAND). The authors thank V. Yfantis for expert technical assistance and Prof. B. van den Berg (Newcastle, UK) for the kind gift of the pB22-OprD construct. H.C. was Boursier of the Belgian Fonds pour la Recherche dans l’Industrie et l’Agriculture (FRIA) and F. V. B. is Maître de Recherches of the Belgian Fonds de la Recherche Scientifique (F.R.S.-FNRS).
Author information
Authors and Affiliations
Contributions
H.C., D.P., Y.B. and H.R.V. performed the experiments. H.C., P.M.T. and F.V.B. designed the studies. P.M.T., M.W. and F.V.B. supervised the work. D.P., Y.B., H.W., M.M.T., J.S.E., P.P., B.C.K. and O.D., collected or provided biological material and gave useful suggestions. H.C. and F.V.B. wrote the manuscript and all the authors revised the final text and agreed with its submission.
Corresponding author
Ethics declarations
Competing interests
P.M.T. is an unpaid advisor to Eumedica. F.V.B. has obtained a private public partnership financial support (TEMOEXPAND program) from the Region Wallonne for the performance of the present work, with Eumedica as industrial partner.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chalhoub, H., Pletzer, D., Weingart, H. et al. Mechanisms of intrinsic resistance and acquired susceptibility of Pseudomonas aeruginosa isolated from cystic fibrosis patients to temocillin, a revived antibiotic. Sci Rep 7, 40208 (2017). https://doi.org/10.1038/srep40208
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep40208
This article is cited by
-
Adaptation to the cervical environment is associated with increased antibiotic susceptibility in Neisseria gonorrhoeae
Nature Communications (2020)
-
Identification and characterization of phage protein and its activity against two strains of multidrug-resistant Pseudomonas aeruginosa
Scientific Reports (2019)
-
Pharmacokinetics and Pharmacodynamics of Temocillin
Clinical Pharmacokinetics (2018)
-
Mitigation of a nitrate reducing Pseudomonas aeruginosa biofilm and anaerobic biocorrosion using ciprofloxacin enhanced by D-tyrosine
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.