Abstract
Rice bacterial blight (BB), caused by Xanthomonas oryzae pv. oryzae (Xoo), is one of the devastating diseases of rice. It is well established that the wild rice Oryza meyeriana is immune to BB. In this study, the transcriptomic analysis was carried out by RNA sequencing of O. meyeriana leaves, inoculated with Xoo to understand the transcriptional responses and interaction between the host and pathogen. Totally, 57,313 unitranscripts were de novo assembled from 58.7 Gb clean reads and 14,143 unitranscripts were identified after Xoo inoculation. The significant metabolic pathways related to the disease resistance enriched by KEGG, were revealed to plant-pathogen interaction, phytohormone signaling, ubiquitin mediated proteolysis, and phenylpropanoid biosynthesis. Further, many disease resistance genes were also identified to be differentially expressed in response to Xoo infection. Conclusively, the present study indicated that the induced innate immunity comprise the basal defence frontier of O. meyeriana against Xoo infection. And then, the resistance genes are activated. Simultaneously, the other signaling transduction pathways like phytohormones and ubiquitin mediated proteolysis may contribute to the disease defence through modulation of the disease-related genes or pathways. This could be an useful information for further investigating the molecular mechanism associated with disease resistance in O. meyeriana.
Similar content being viewed by others
Introduction
Rice (Oryza sativa L.) is one of the most important food crops in the world. However, rice production is seriously constrained by various diseases1. More than 70 diseases caused by fungi, bacteria, viruses, and nematodes have been reported on rice2. Bacterial leaf blight (BB) caused by Xanthomonas oryzae pv. oryzae (Xoo) has been become a devastating disease of rice worldwide in 1960s and 1970s when the high-yielding varieties were introduced3. In practice, development of the resistant cultivars and transgenic rice is one of the major strategies for BB management1,2. Nevertheless, the high degree of pathogenic variation in Xoo4 and rapid loss of resistance in the rice varieties against BB5, development of resistant varieties become a new challenge for breeders. Therefore, exploring new resistant genes become an urgent and arduous task both in practice and theory.
Wild rice, which is growing widely in 77 countries of the Asia, Africa, Latin America and Australia, is considered to be an important and rich gene pool6. Many species of wild rice may possess substantial elite genes that are resistant to the diseases and insects6,7. Plant breeders and geneticists of the world have made great progress to identify the resistant materials to the rice BB. Until today, more than 40 Xanthomonas resistance (Xa) genes specifically resistant to various strains of Xoo were identified or isolated from a variety of rice materials8. Some of these genes were obtained from different wild rice species. For example, Xa21 is the first resistant gene derived from the wild rice (O. longistaminata)7.
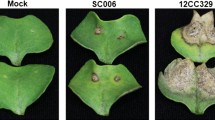
The awnless wild rice species of O. meyeriana Baill is a unique source9 (Fig. 1a), possessing GG genome that is different from the cultivated rice with AA genome10. O. meyeriana collected from different regions of South China has been demonstrated highly resistant to Xoo infection, usually accompanied by the typical hypersensitive response (HR)11. In our study, disease spot was restricted to the inoculated points on the leaves after inoculated with Xoo (Fig. 1a). In contrast, the susceptible cultivar of O. sativa showed spreading lesions after inoculation with the same bacterial pathogen (Fig. 1a). Previous studies on the physiological and anatomical changes of O. meyeriana after Xoo inoculation revealed that xylem cell wall was thickened and nitric oxide (NO) level was raised after Xoo infection12,13. Therefore, O. meyeriana is regarded as a valuable gene pool. The genetic resources of O. meyeriana have been successfully used for BB resistance through asymmetric somatic hybridization14. As a result, highly resistant lines were selected from the hybrid progeny between O. meyeriana and O. sativa subsp. japonica15. In addition, several molecular approaches were also used to identify the resistance-related genes to BB. For example, some genes involved in cell wall lignification were identified in O. meyeriana in response to the Xoo infection by the suppression subtractive hybridization16. Most recently, a transcriptomic sequencing also illustrated that many putative resistance-related genes were transcribed in O. meyeriana under the normal growing condition17. Although these studies provide valuable information with regards to the BB resistance, available information concerning molecular mechanism or genes involved in the response to Xoo infection for O. meyeriana is still limited.
Comparison of leaf symptoms of Oryza meyeriana and O. sativa inoculated with Xanthomonas oryzae pv. oryzae and (a) the expression patterns of the differentially expressed unitranscripts (DEUs) of O. meyeriana in response to X. oryzae pv. oryzae (Xoo) attack. (b) Heat-map depicting the expression changes in all the DEUs. (c) Venn diagram exhibiting the DEUs’ distribution in three samples.
It is well documented that plants have evolved complex innate immune systems in response to the biotic and abiotic stresses. The plant immune system is composed of surveillance systems to perceive the microbe elicitors, such as the well-characterized flagellin and peptidoglycan, which in turn allow plants to switch from the normal growth and development to a defence mode18,19. This process is so called as pathogen-associated molecular patterns (PAMP)-triggered immunity (PTI), which is considered as an important initial level in pathogen response. However, the bacterial pathogens may inhibit PTI via secreting virulence factors (effectors) into the plant cells19. To overcome these virulence factors, plants have evolved disease resistance (R) genes, typically encoding the nucleotide-binding site and leucine-rich repeat (NBS-LRR) domains, to recognize these effectors and initiate a second layer of response called the effector-triggered immunity (ETI). ETI often leads to HR mediated by the R proteins in plant and is an accelerated and amplified PTI response, resulting in disease resistance to protect the plant itself19. However, non-host resistance to pathogens is often associated with the HR, in which the molecular recognition events and activation of plant innate immunity are involved20. Besides, the other signaling pathways like phytohormone signaling, ubiquitin mediated proteolysis, etc, are involved in the defence response to the pathogens in plants, too21. Overall, plant defence response is a complex process, involving multiple signaling pathways and their cross-talks.
Transcriptomic sequencing based on the next-generation sequencing technologies, provides a powerful and effective strategy to produce genome-scale sequence data, which allows the whole transcriptome to be investigated in a high-throughput and quantitative manner, especially for non-model organisms with genomic sequences that are yet to be measured22. Therefore, RNA-seq has been successfully used to investigate the differential gene expression in many pathosystems, like Xoo in cultivar rice23, X. axonopodis pv. glycines in soybean24, or X. arboricola pv. pruni in peach25. To get insights on the global response of O. meyeriana to Xoo infection, deep RNA sequencing was performed in this work. Through identifying differentially expressed genes (DEGs) of O. meyeriana after Xoo infection, the major metabolic pathways related to disease resistance including the plant innate immunity, phytohormone signaling, ubiquitin mediated proteolysis, and phenylpropanoid biosynthesis were enriched and the candidate genes targeting to the disease resistance in the plant were also inferred.
Results
RNA sequencing and de novo assembly
To understand the molecular basis of the disease resistance of O. meyeriana against Xoo, total RNAs from the leaves at 0 h, 1 h, 2 h, 4 h, 24 h, and 48 h after Xoo inoculation were isolated and used for high-throughput sequencing. The RNAs were pooled into three groups: CO acts as the control; TE represents the RNAs from 1 h, 2 h and 4 h after inoculation as the early response; TO signifies the late response derived from the 24 h and 48 h RNA samples. Each group was further divided 3 sub-samples. Altogether, 9 sub-samples were applied to Illumina HiSeq2000 platform for sequencing. The sequencing data are summarized (Table 1) and total 58.7 Gb 100-base clean reads were obtained.
The short reads pooled from the three libraries were primarily assembled into 210,601 contigs using the Trinity package. Prediction of ORFs was carried out to obtain 97,596 contigs encoding ORFs with 4 types (no-start-codon, no-stop-codon, no-start-stop-codon and intact ORFs). All the contigs encoding ORFs were further assembled using the software CAP3, resulting in 57,313 unitranscripts with a mean length of 2,637 bp (Table 1). All the 57,313 unitranscripts were annotated by sequence similarity using BLASTx against the NCBI non-redundant (NR) protein database. In a total of 55,671 (97.14%) unitranscripts have at least one significant alignment to existing gene models in the NR database at an E-value cut-off of 1e-5. The distribution and abundance of the transcripts in species had the top three matches of O. sativa, O. brachyantha, and Brachypodium distachyon.
Identification of differentially expressed unitranscripts (DEUs)
The relative expression level of each transcript in terms of fragment per kilobase per million (PKFM) was estimated by mapping all the clean reads from each library back to the 57,313 transcripts. Consequently, 14,143 DEUs (24.7%) were identified between the three libraries of CO, TE and TO with P values ≤0.05 (Table S1). The analysis of DEU profiles across the three libraries indicated that the DEUs were roughly clustered into three main groups (Fig. 1b). Cluster A comprised the transcripts mostly up-regulated at an early stage; cluster B contained the transcripts mostly up-regulated at a late stage, and cluster C mostly down-regulated after Xoo infection.
To find out the intrinsic differences in the transcriptome of the three libraries, DEUs in the libraries of TE and TO were compared with the control (CO) (Fig. 1c). Totally, 9,561 (5,298 up-regulated and 4,263 down-regulated) and 6,507 (3,428 up-regulated and 3,079 down-regulated) DEUs were identified between TE/CO and TO/CO, respectively. Meanwhile, 8,634 (4,150 up-regulated and 4,484 down-regulated) DEUs were also found between TE/TO. However, 92 DEGs were common between TE/CO and TE/TO. It is noteworthy that 3,633 DEUs were discovered up-regulated solely in TE compared with CO.
Furthermore, the functional classification of the DEUs in O. meyeriana after Xoo infection was analyzed using GO (Gene Ontology). A total of 8,134 (57.6%) transcripts were assigned to at least one GO term. Among the other biological processes, where the term occurrences took place, metabolic process (25.6%) and cellular process (23.4%) were highly represented (Fig. S1). In addition, transcripts participation or involvement in biological process like response to stimulus, immune system, cell death, were recognized through GO annotation.
To explore the biochemical pathways in which DEUs were involved, the metabolic pathway analysis was performed for all the DEUs using the KEGG Automatic Annotation Server (KAAS). The results revealed that 5,137 DEUs was assigned into 313 Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways (Table S2). Most of the DEUs were involved in the primary metabolisms, notably biosynthesis of amino acids, purine metabolism, and carbon metabolism, etc. Most importantly, several pathways associated with disease defence were also enriched by KEGG which would be stressed on in more detail below.
Differentially expressed genes (DEGs) involved in the plant-pathogen interaction pathway
In the plant-pathogen interaction pathway (Ko04626), 19 DEGs containing 133 DEUs were identified as up-regulated at the early stage in response to Xoo inoculation (Fig. 2 and Table S3). Except WRKY1 (WRKY transcriptional factor 1), PTI1 (pto-interacting protein 1) and CREK1 (chitin elicitor receptor kinase 1), 16 DEGs were enriched in two immune signaling pathways: the PTI and ETI. However, 6 DEGs involved in PTI were up-regulated at the early stage (Fig. 2). In plants, FLS2 (flagellin-sensing 2) perceives the bacterial flagellin, and then activates a signaling tandem of the mitogen-activated protein kinase (MAPK) cascades, like MEKK1 (mitogen-activated protein kinase kinase kinase 1), MKK1/2 (mitogen-activated protein kinase kinase 1), and MKK4/5 (mitogen-activated protein kinase kinase 4/5). Finally, the transcriptional factors of WRKY22/29 (WRKY transcription factor 22) and WRKY25/33 (WRKY transcription factor 25) were activated, which in turn induce expression of the defence-related genes18,19.
PAMP usually triggers a rapid increase in the cytoplasmic Ca2+ concentration and activates the calcium-dependent protein kinase (CDPK) in plant cells26. In the present study, in O. meyeriana, 4 genes involved in Ca2+ signaling were up-regulated at an early stage of Xoo infection (Fig. 2 and Table S3). These DEGs are the cyclic nucleotide gated channels (CNGCs), CDPK, calmodulin (CaM), and calmodulin-like proteins (CML). Additionally, 11 transcripts encoding nitric oxide synthase (NOS) were enriched but only the minority showed up-regulated expression in the early and late stage after Xoo inoculated (Table S3). The calcium signaling has long been demonstrated to be related to response to the pathogen attack, which is usually accompanied by an increase of both ROS and NO26.
The secondary immune response known as ETI is thought to mount a second layer of defence18,19. The second signaling pathway is composed of five genes encoding the receptor proteins to perceive the bacterial effector proteins. Here, three DEGs encoding receptor proteins (RPM1, the disease resistance protein RPM1; RIN4, PRM1-interacting protein 4; PBS1, the disease resistance protein PBS1) showed up-reregulated gene expression pattern at the early stage of Xoo infection (Fig. 2). In addition, the downstream HSP90 (heat shock protein 90) was found down-regulated at the early stage (TE/CO) followed by up-regulated at the late stage (TO/CO). HSP90 has been reported to form a complex with RAR1 (disease resistance protein RAR1) and SGT1 (suppressor of G2 allele of SKP1), which may interact with the R proteins27, perhaps to make a contribution to the observed HR in O. meyeriana12,13.
DEGs in the phytohormone and ubiquitin-mediated pathways
Previous studies have demonstrated that various disease resistance gene are involved in phytohormone signal transduction and ubiquitin-mediated proteolysis28,29. With a view to indentifying genes involved in phytohormone signal transduction and ubiquitin-mediated proteolysis, gene expression was calculated. As expected, 44 DEGs encoding the proteins involved in these signaling pathways were detected (Table S4).
Phytohormones, such as salicylic acid (SA), jasmonic acid (JA), gibberellic acids (GAs), ethylene (ET), and brassinosteroids (BRs), act as signaling molecules to trigger or mediate a diverse array of plant immune responses28. In this study, 8 DEGs related to SA, JA, and ET signaling pathways were enriched in O. meyeriana in response to Xoo infection (Fig. 3). In JA signaling, two DEGs (encoding the coronatine insensitive protein 1, COI1; jasmonate ZIM-domain containing protein, JAZ) were enriched to be up-regulated. The other genes encoding JAR1 (jasmonic acid-amino synthetase) and MYC2 (transcription factor MYC2) involved in JA signaling did not respond to Xoo infection. It is well-known that SA is also an important phytohormone that regulates plant defence processes. NPR1 (regulatory protein NPR1) is an essential regulator in SA-signaling was up-regulated at the early stage in O. meyeriana. The downstream gene encoding the transcription factor TGA was showed significantly different expression at different stages, which was down-regulated between TE/CO, while up-regulated between TO/CO (Fig. 3 and Table S4).
As one of the backbone of the induced defence signaling pathway, ET signal transduction has been well documented30. In our study, the genes encoded with serine/threonine-protein kinase CTR1, mitogen-activated protein kinase 6 (MAPK6), and ethylene-insensitive protein 3 (EIN3) were shown up-regulated between TE/CO. In contrast, Xoo infection did not induce the expression of genes encoding ethylene receptor ETR and ethylene-insensitive protein 2 (EIN2).
Recent studies indicated that the auxin and cytokinin (CK) signaling pathways may be involved in defence response31. Here, several DEGs involved in auxin and CK signaling pathways were also identified (Fig. 3).
Ubiquitination exists extensively in plants and may be involved in the regulation of plant disease resistance32. In this study, 4 DEGs encoding E2 were identified between TE/CO after Xoo infection, among them, three showed up-regulated and another one showed down-regulated (Table 2). Eighteen DEGs encoding E3 ubiquitin ligase belonging to five types were identified (Table 2), 9 of which were up-regulated between TE/CO. Especially, 7 DEGs out of the 18 belong to the type of the multi subunit RING-finger, which were shown to mediate plant disease resistance33.
DEGs in the phenylpropanoid biosynthesis pathway
The phenylpropanoid biosynthesis pathway is one of the important secondary metabolic pathways in the plant, which plays important roles in plant defence34. Phenylpropanoid biosynthesis pathway of O. meyeriana in response to Xoo infection was annotated by KEGG analysis (Fig. 4). At most 128 unitranscripts encoding 12 DEGs were shown to be up-regulated especially at the early stage after Xoo infection (Table S5). In phenylpropanoid pathway, the first key enzyme is the phenylalanine ammonia-lyase (PAL), which was well known to be induced in many plants by pathogen attack. Here, all the 14 transcripts encoding PAL were up-regulated between the TE/CO. Further, there are a large number of enzymes involved in lignin biosynthesis34. Although it is not clear which transcript encodes this specific enzyme, the majority of the transcripts encoding 4-coumarate-CoA ligase (4CL), cinnamoyl-CoA reductase (CCR), cinnamyl-alcohol dehydrogenase (CAD), and peroxidase (PO) were shown to be up-regulated after Xoo infection (Fig. 4 and Table S5).
Enriched R genes in response to Xoo infection
The R genes, which encode disease resistance proteins especially containing the LRR/NBS-LRR domains, are widely recognized to defeat pathogens in many plants35. In rice, most of the R genes especial to BB are unique and recessively regulated36. Previously, more than 40 resistance genes (Xa) have been identified, conferring resistance to Xoo in rice7. In O. meyeriana, two Xa genes (xa13 and Xa21) were identified as DEGs after Xoo infection (Table S6). There is only one transcript annotated as the disease resistant allele xa13, which exhibited high expression at the early stage and continue to rise at the late stage after Xoo infection.
Xa21, which encodes a receptor-like kinase, is a broad-spectrum resistance gene of rice against BB37. Here, 14 and 3 transcripts were identified and annotated to encode Xa21 and Xa21-binding proteins, respectively (Table S6). Among the 17 transcripts encoding Xa21-related proteins, 8 were up-regulated between TE/CO, while 6 were continuously up-regulated between TO/TE. It is suggested that most of the Xa21 transcripts were stimulated by the Xoo infection.
The R genes occur ubiquitously in the plant kingdom and comprise the second mechanism of disease resistance by recognizing the pathogen Avr gene directly or indirectly38. Until now, five different types of R genes have been identified up to date39. A large number of R genes were identified as the family that encoded NBS-LRR containing domain protein38. In this study, 103 transcripts encoding various R genes were enriched in O. meyeriana after Xoo infection (Table S6). Among them, 42 DEUs belonging to the NBS-encoding R genes, were detected in the transcriptomic profile (Table S6). Some of the NBS-encoding R genes were also detected to encode other domains, such as TIR and CC domain attached to the N-terminal. However, different expression patterns were observed for these transcripts encoding NBS-containing domain, some of them were down-regulated and the rests were up-regulated in response to the Xoo infection (Fig. S6).
In addition, many DEUs associated with the disease resistance genes annotated to the NBS-encoding transcripts. At least, five groups could be detected in terms of the annotation information (Table S6). The first group is consisted of the 6 transcripts encoding disease resistance gene analog (RGA) proteins40, of which two transcripts were up-regulated and 3 down-regulated at the early stage. The second group (24 transcripts) was annotated to encode the rust-related R proteins, such as resistance kinase and stripe rust resistance protein. Of these, 17 transcript were up-regulated and 5 showed lower expression in TE or TO. The third group is those to encode nb-arc domain. Only one transcript was up-regulated in TE compared with CO while the other three transcripts were down-regulated between TE/CO. The fourth group was annotated to encoding negative regulator of systemic acquired resistance, all of the 3 transcripts showed down-regulated at the early stage. The final group was consisted of 24 transcripts encoding proteins relative to various resistance, of which some showed up-regulated and the others down-regulated.
Validation of the selected DEUs by RT-qPCR
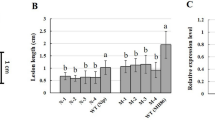
Twenty-five DEUs involved in the plant innate immunity, phytohormone signaling, phenylpropanoid biosynthesis pathway, and the R genes were selected for RT-qPCR using the specific primers to confirm the reliability and availability of DEUs obtained from the RNA sequencing (Table S7). The relative expression levels of the selected genes were determined at 0, 1, 2, 4, 24, and 48 h after Xoo inoculation. The RT-PCR results (Fig. 5) showed that 9 genes involved in the plant innate immunity, were up-regulated at an early stage (1–2 h), and thereafter showed decreased expression at the later stage (24–24 h) of Xoo infection. Only the transcript encoding RBOH continued to rise until 24 h post-inoculation and the HSP90 was down regulated at the early stage of infection. These results further indicated that the innate immune response arose at early stage of Xoo infection in O. meyeriana. The similar pattern was observed for these transcripts involved in JA and SA-mediated signaling pathways. Various E3 genes showed different expressing responses.
Validations of the differentially expressed transcripts by RT-qPCR.
Samples were collected from the leave at 0, 1, 2, 4, 24, and 48 h after Xoo infection. All data were normalized to the expression level of β-actin. The data represent the change fold at each time point in the infected samples vs. the control sample (Time 0). Bars show the standard deviations from three replicates.
Discussion
In fact, immune machinery may widely exist in the plant world. More importantly, the expression of the resistance is mostly dependent upon how the common signaling machinery is used in a given plant-pathogen interaction41. Therefore, RNA-seq was used to get clear understanding of how the wild rice O. meyeriana responded to the Xoo infection. In this study, 327 Mb of transcripts in length was obtained by assembling 58.7 Gb clean reads. Finally, 57,313 unitranscripts encoding ORF were de novo assembled.
By bioinformatic analysis of the transcriptomic data, we provide novel insights into the defence responses of O. meyeriana to Xoo infection. Moreover, several metabolic pathways related to disease resistance were significantly enriched. In plants, the innate immunity is triggered through PTI, and then ETI, which provides the first line of disease resistance18,19. In the case of O. meyeriana, the expression level of all these genes involved in PTI was up-regulated at the early stage after Xoo inoculation (Fig. 2 and Table S3). These DEGs may comprise an FLS2-mediated plant immune pathway to confer O. meyeriana against Xoo infection. In addition, we have manually detected 4 transcripts encoding the brassinosteroid insensitive 1-associated receptor kinase (BAK1) being up-regulated after Xoo infection (Table S3). BAK1 was previously reported to form a flg22-induced complex with FLS2, which was required for all known downstream flg22-signaling responses in Arabidopsis thaliana42. However, two members of MPK3/6 and MPK4 involved in the MAPK cascades were not identified in this wild rice. In fact, MAPK cascades have been reported to play important roles, especially in dicot plant responses to pathogen infection. Further, a large number of MAPK genes were also demonstrated to be differentially expressed in response to Xoo infection in both of the resistant and susceptible rice lines43. In terms of these missed components of MAPK cascades, the innate immunity of O. meyeriana may be different from that of A. thaliana and rice.
In compatible plant-pathogen interaction, the bacterial pathogen may secret effectors into the plant cells to suppress the PTI44. However, plants adopt the second pathogen-sensing mechanism called ETI to defeat the effectors. During ETI, the effector proteins are recognized by a class of plant receptor proteins that usually contain NB-LRR domains18,19. Up to now, several receptor proteins such as RIN4, RPM1, EDS1, have been identified in various plants19. In O. meyeriana, at least three genes encoding RIN4, RPM1, PBS1 were enriched as up-regulated at the early stage in response to Xoo infection (Figs 2 and 5). Conclusively, our data suggest that the innate immunity is an early responding event and may comprise a basal resistant mechanism to defeat Xoo attack in the incompatible O. meyeriana-Xoo interaction.
In addition, the receptor-kinase of flg22 also triggers Ca2+ signaling by FLS226. Almost the whole components (CNGC, CDPK, CaM/CML, Rboh, and NOS at least for partial transcripts) involved in the Ca2+ signaling were shown to be up-regulated in O. meyeriana in response to Xoo infection (Fig. 2 and Table S4). Previous studies showed that the crucial role for the induction of Ca2+ signaling in response to abiotic and biotic stresses26. In rice, Ca2+ signaling was also identified as up-regulated in response to the infection of several pathogens by the microarray-based transcriptomic studies45. One of the outcome of Ca2+ signaling is HR, which is one of the hallmarks of the plant innate immunity, especially in the incompatible systems46. HR was observed on the Xoo-inoculated leaves of O. meyeriana, which suggested the presence of immunity in O. meyeriana11. HR is usually associated with persistent production of ROS, NO, SA and Ca2+ fluxes26. The increase of NO especially in the xylem cell walls in O. meyeriana was indeed observed after Xoo inoculation by the immunohistochemistry assay13. Therefore, we believe that the observed increase of HR and NO in the O. meyeriana leaves inoculated with Xoo can be ascribed to the inducible expression of these genes involved the innate immune and Ca2+ signaling pathways.
Phytohormones, such as JA, SA, ET, etc., play key roles in the complex signaling cascades in growth, development, as well as defence responses. Several DEGs involving in these phytohormone signaling pathways were enriched (Fig. 2), suggesting that JA, SA and ET signaling pathways may be included in the response of O. meyeriana to Xoo. SA is usually effective against biotrophic pathogens; whereas JA and ET mainly involve in defence of necrotrophic pathogens. And SA-JA antagonism was observed in O. sativa and A. thaliana47. For example, overexpression of OsNPR1 led to activation of SA-response genes and the concomitant suppression of JA maker genes in rice48. However, both SA and JA pathways were up-regulated in O. meyeriana after Xoo infection. Similar results were also reported in a resistant rice line carrying Xa39, but not in its susceptible parental lines23. However, only JA-signaling genes were up-regulated after inoculation of pathogen in soybean-X. axonopodis pv. glycines pathosystem24. Even no genes involved in both SA or JA-signaling was enriched in peach leaves inoculated with the invasive X. arboricola pv. pruni25. Therefore, response of SA- and JA-signaling to microbial pathogens seems different and may be dependent upon various pathosystems.
The ubiquitin-proteasome system (UPS) play important roles in various cellular processes, which contains four basal components of E1, E2, E3 and ubiquitin49. Plant genomes usually encode large numbers of E3 ubiquitin ligase. In fact, UPS-mediated protein degradation has been demonstrated in regulation of almost all of hormone signaling pathways, such as the auxin, CK, ET, JA and SA50. For example, the E3 ubiquitin-ligase SCFCOI1 complex may activate with the downstream response of the JA signaling through degradation of the JAZ repressor51. Furthermore, many E3 ligases were also identified to participate in the regulation of plant immunity52. In our study, up to 19 unitranscripts encoding various types of E3 ubiquitin ligase were revealed to be differentially expressed in response to Xoo infection (Table 2). We believe these E3 ubiquitin ligases may play an important role in the defence response after Xoo infection in O. meyeriana, perhaps through cross-talking with the other disease resistance pathways, like phytohormone and plant immune signaling pathways.
The secondary phenylpropanoid metabolic pathway was revealed to be overall up-regulated. The similar results have been reported in many plant-pathogen interactions53. This pathway can synthesize many small molecules and strengthen the cell wall by depositing callose and lignin, which may inhibit the pathogenic bacteria growth and spread in plants34. In O. meyeriana, thickening of cell wall especially in the leaf xylem tissue was reported as a defence mechanism against Xoo12. Therefore, induction of phenylpropanoid pathway by Xoo infection may contribute to the cellular wall thickening of the leaf xylem tissue in O. meyeriana.
The R genes are regarded as the second mechanism of disease resistance and have been recognized in various plants. To date, about 40 R genes specific to BB have been identified in rice, and nine (Xa1, Xa3/Xa26, xa5, xa13, Xa10, Xa21, Xa23, xa25 and Xa27) have been isolated by map-based cloning7. Two known genes of Xa21 and xa13 were identified as DEGs in this work, but not in previous studies17. In addition, our data revealed more than 100 unitranscripts encoding various R genes that were differentially expressed in response to Xoo infection (Table S6). Among them, the majority of these DEUs encode the typical R proteins containing NBR domain.
In conclusion, the transcriptomic analysis provide us with a glimpse of the transcriptional response of O. meyeriana to Xoo attack, indicating that several resistant-related signaling pathways were up-regulated and consisted of a complex defence network. The plant innate immune (MTI and ETI) may comprise the basal defence frontier, and then activates the R gene expression, which in turn induces HR and thickening of xylem cell wall to restrict spreading of the bacterial cells in the leaf xylem tissue of O. meyeriana.
Methods
Plant materials
O. meyeriana was collected from the native niches at Yuanjiang county, Yunnan province of China, and grown in a greenhouse. For one individual plant, three leaves were inoculated with X. oryzae pv. oryzae at an inoculum concentration of 3 × 108 cells per milliliter by the leaf clipping method54. After inoculation, the leaves were collected separately at, 1 h, 2 h, 4 h, 24 h and 48 h, immediately frozen in liquid nitrogen, and then stored at −80 °C for isolation of the total RNAs. The leaves sampled immediately after inoculation with the same Xoo cell suspension, were served as the control (0 h). Three replicates of leaf samples (3 × 3 leaves) were collected for each time points.
RNA extraction and library construction
Total RNAs were extracted from all the leaf samples using Trizol reagent (Roche, Switzerland) according to manufacturer’s instructions. The genomic DNAs were removed by DNase I (Fermentas, USA). The RNA concentration and quality were measured with the Qubit fluorometer (Invitrogen, USA) and the Agilent 2100 Bioanalyzer, respectively. Only the RNA samples with a RIN (RNA integrity number) >8.0, OD260/280 ratio ranged from 1.9 to 2.1, and OD260/230 ratio of 2.0 to 2.5, were used for the next processing.
For RNA sequencing, the RNA samples were combined into three groups: CO, TE and TO, which were designed for the control leaves (0 h); early stage (1 h, 2 h and 4 h); and the late stage (24 h and 48 h), respectively. Each group was equally divided into 3 sub-samples. For each sub-sample, the mRNAs were further purified from the total RNAs (20 μg) using Dynaloligo (dT) 25 magnetic beads and fragmented into smaller pieces. The fragmented mRNAs were used for the first-strand cDNA synthesis with the random primer by reverse transcriptase. Second strand cDNAs were synthesized using RNase H and DNA polymerase I. The resulting cDNA fragments were further repaired at 3′-end by adding a single base (A), and then ligated with the Illumina adapters. Subsequently, the cDNA fragments were purified and enriched by PCR. The quality of cDNA libraries was measured by Agilent 2100 Bioanalyzer.
Illumina sequencing and De novo assembly
The cDNA libraries were sequenced using the Illumina HiSeq2000 platform. The fluorescent image processing, base-calling and calculation of quality value were performed by the Illumina data processing pipeline 1.4. Meanwhile, sequencing quality was assessed by fastQC (http://ww.bioinformatics.bbsrc.ac.uk/projects/fastqc/). Finally, 100 bp paired-end (PE) clean reads were obtained.
The sequence assembly was performed on a server with 24 cores and 256 GB random access memory. All the PE reads derived from the above 9 libraries were de novo assembled using the Trinity_release_2013111055 under default parameters. In order to realize better results in the following analysis, the best candidate coding sequence (CDS) analysis was performed for each contig using the perl script in the Trinity package. All the contigs with potential CDS were further reassembled using software CAP356 to generate a non-redundant set of unitranscripts. The parameters used for the reassembly were >95% identical with a minimum of 40 bases with maximum of 20 bases of unmatched overhangs at the sequence end.
To assay the assembly quality, all the PE reads were aligned back to these contigs by using Bowtie2 program (v2.0.0-beta5)57, and the aligned rate was calculated. The common perl scripts were used to assess the length distribution of the transcripts, N50 number, average length, max length, and the numbers of contigs in different length intervals.
Functional annotation
All the reassembled contigs with predicted ORFs generated by CAP3 were used for similarity search against the NR database downloaded from GenBank (http://www.ncbi.nlm.nih.gov/) by using local blastx program with the expect E-value cut-off <10−5.
The blast results were imported into the Blast2GO58 and performed functional annotation. Gene ontology (GO) enrich was achieved on WEGO (http://wego.genomics.org.cn/cgi-bin/wego/index.pl). Meanwhile, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways were assigned to each contig on KEGG Automatic Annotation Server (KAAS) (http://www.genome.jp/kegg/). Thus, multiple contigs could be clustered to the same GO terms and the same KEGG pathway.
Abundance estimation and identification of differential expressed genes
In order to calculate the relative express level of each contig in different samples, all the PE reads pooled from the three sub-samples in each sample group were aligned back to the final annotated contigs by using perl scripts in the Trinity package under default parameters option55. The digital expression levels from the alignment for each contig were normalized using RESM-based algorithm by using perl scripts in Trinity package. So values of the fragments per kilobase per million (FPKM) for each contig were obtained59. And then, the edgeR package (the Empirical analysis of Digital Gene Expression in R)60 was used to extract those contigs which were regarded as differentially expressed genes with the P-value ≤ 0.05 and the log2fold-change (log2FC) >1.
Validation of the selected genes by qPCR
Twenty five DEUs were selected to verify the reliability of the RNA sequencing data by quantitative real-time PCR (RT-qPCR). The designed primers for amplification of the selected transcripts were presented in Table S7. Total RNAs were isolated from the O. meyeriana leaves at 0 h, 1 h, 2 h, 4 h, 24 h and 48 h post inoculation with Xoo as described above. After treated with DNase I, first strand cDNAs were synthesized using oligo (dT) and random hexamers as primers by M-MLV reverse transcriptase (Tiangen Co., Beijing, China) according to the manufacturer’s instructions. Real-time PCR were performed in a 25 μl volume using SYBR premix Ex TaqTM II (Takara Co., Dalian, China) on the CFX Connect Real-time PCR detection system (Bio-Rad, Hercules, USA). The quantitative PCR was followed by the program: 5 min at 95 °C; followed by 45 cycles of amplification with denaturation for 5 s at 95 °C, annealing for 30 s at 55 °C, and extension for 20 s at 72 °C. Triplicate under identical conditions were synchronously performed for all selected genes. The relative expression values of each transcript were then calculated by the delta-delta Ct (2−ΔΔCt) method using the CFX Manager 3.0 of the amplifier.
Data availability
Raw sequencing data are available through the NCBI Sequence Read Archive (BioSample Submission Potal, accession number: SRP071037). All samples were sequenced as 100 bp paired-end reads on an Illumina HiSeq2000 sequencer.
Additional Information
How to cite this article: Cheng, X.-J. et al. Transcriptome analysis confers a complex disease resistance network in wild rice Oryza meyeriana against Xanthomonas oryzae pv. oryzae. Sci. Rep. 6, 38215; doi: 10.1038/srep38215 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Gnanamanickam, S., Priyadarisini, V. B., Narayanan, N., Vasudevan, P. & Kavitha, S. An overview of bacterial blight disease of rice and strategies for its management. Curr. Sci. 77, 1435–44 (1999).
Zhang, H., Li, G., Li, W. & Song, F. Transgenic strategies for improving rice disease resistance. Afr. J. Biotechnol. 8, 1750–1757 (2009).
Mizukami, T. & Wakimoto, S. Epidemiology and control of bacterial leaf blight of rice. Annu. Rev. Phytopathol. 7, 51–72 (1969).
Jeung, J. U., Heu, S. G., Shin, M. S., Vera Cruz, C. M. & Jena, K. K. Dynamics of Xanthomonas oryzae pv. oryzae populations in Korea and their relationship to known bacterial blight resistance genes. Phytopathol. 96, 867–875 (2006).
Vera Cruz, C. M. et al. Predicting durability of a disease resistance gene based on an assessment of the fitness loss and epidemiological consequences of avirulence gene mutation. Proc. Natl. Acad. Sci. USA 97, 13500–13505 (2000).
Jena, K. K. The species of the genus Oryza and transfer of useful genes from wild species into cultivated rice, O. sativa. Breeding Sci. 60, 518–523 (2010).
Kim, S.-M., Suh, J.-P., Qin, Y., Noh, T.-H., Reinke, R. F. & Jena, K. K. Identification and fine-mapping of a new resistance gene, Xa40, conferring resistance to bacterial blight races in rice (Oryza sativa L.). Theor. Appl. Gen. 128, 1933–1943 (2015).
Khush, G. S., Bacalangco, E. & Ogawa, T. A new gene for resistance to bacterial blight from O. longistaminata. Rice Genet. Newsl. 7, 121–122 (1990).
Gong, Y., Borromeo, T. & Lu, B.-R. A biosystematic study of the Oryza meyeriana complex (Poaceae). Plant Syst. Evo. 224, 139–151 (2000).
Aggarwal, R. K., Brar, D. S. & Khush, G. S. Two new genomes in the Oryza complex identified on the basis of molecular divergence analysis using total genomic DNA hybridization. Mol. Gen. Genet. 254, 1–12 (1997).
Zhang, Q. et al. Evaluation of resistance to bacterial blight (Xanthomonas oryzae pv. oryzae) in wild rice species. Sci. Agri. Sin. 27, 1–9 (1994).
Yang, Y. et al. Xylem secondary cell-wall thickening involved in defense responses of Oryza meyeriana to Xanthomonas oryzae pv. oryzae. Acta Phytopathol. Sin. 42, 505–514 (2012).
Cheng, X. Y. et al. Nitric oxide involved in defense responses of Oryza meyeriana to rice bacterial blight. Acta Agri. Zhejiangensis 26, 1–6 (2015).
Yan, C.-Q. et al. Use of asymmetric somatic hybridization for transfer of the bacterial blight resistance trait from Oryza meyeriana L. to O. sativa L. ssp. japonica. Plant Cell Rep. 22, 569–575 (2004).
Yan, C.-Q. et al. Production of the bacterial blight resistant line from somatic hybridization between Oryza sativa L. and Oryza meyeriana L. J. Zhejiang Uni. Sci. 5, 1199–1205 (2004).
Cheng, Z., Tang, K., Yan, H., Fu, J., Ying, F. & Huang, X. Analysis of differentially expressed genes in Oryza meyeriana in response to infection by Xanthomonas oryzae pv. oryzae. Plant Breeding 129, 393–399 (2009).
He, B. et al. Transcriptomic analysis and the expression of disease-resistant genes in Oryza meyeriana under native condition. PLoS ONE 10, e0144518 (2015).
Jones, J. D. & Dangl, J. L. The plant immune system. Nature 444, 323–329 (2006).
Dodds, P. N. & Rathjen, J. P. Plant immunity: towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet. 11, 539–548 (2010).
Heath, M. C. Nonhost resistance and nonspecific plant denfenses. Curr. Opin. Plant Biol. 3, 315–319 (2000).
De Vleesschauwer, D., Xu, J. & Höfte, M. Making sense of hormone-mediated defense networking: from rice to Arabidopsis. Front. Plant Sci. 5, Article 611 (2014).
Wang, Z., Gerstein, M. & Snyder, M. RNA-seq: a a revolutionary tool for transcriptomics. Nat. Rev. Genet. 10, 57–63 (2009).
Zhang, F. et al. Comparative transcriptome profiling of a rice line carring Xa39 and its parents triggered by Xanothomonas oryzae pv. oryzae provides novel insights into the broad-spectrium hypersenstive response. BMC Genom. 16, 111 (2015).
Kim, K. H. et al. RNA-Seq analysis of a soybean near-isogenic line carrying bacterial leaf pustule-resistant and-susceptible alleles. DNA research 18, 483–497 (2011).
Socquet-Juglard, D. et al. Comparative RNA-seq analysis of early-infected peach leaves by the invasive phytopathogen Xanthomonas arboricola pv. pruni. PLOS ONE 8, e54196 (2013).
Cheval, C., Aldon, D., Galaud, J.-P. & Ranty, B. Calcium/calmodulin-mediated regulation of plant immunity. Biochim. Biophys. Acta 1833, 1766–1771 (2013).
Sangster, T. A. & Queitsch, C. The HSP90 chaperone complex, an emerging force in plant development and phenotypic plasticity. Curr. Opin. Plant Biol. 8, 86–92 (2005).
Pieterse, C. M., van der Does, D., Zamioudis, C., Leon-Reyes, A. & van Wees, S. C. Hormonal modulation of plant immunity. Annu. Rev. Cell Dev. Biol. 28, 489–521 (2012).
Trujillo, M. & Shirasu, K. Ubiquitination in plant immunity. Curr. Opin. Plant Biol. 13, 402–408 (2010).
van Loon, L. C., Geraats, B. P. & Linthorst, H. J. Ethylene as a modulator of disease resistance in plants. Trends Plant Sci. 11, 184–91 (2006).
Fu, J. & Wang, S. Insights into auxin signaling in plant-pathogen interactions. Front. Plant Sci. 2, Article 74 (2011).
Marino, D., Peeters, N. & Rivas, S. Ubiquitination during plant immune signaling. Plant Physiol. 160, 15–27 (2012).
Genschik, P., Sumara, I. & Lechner, E. The emerging family of CULLIN3-RING ubiquitin ligases (CRL3s): cellular functions and disease implications. EMBO J. 32, 2307–2320 (2013).
Dixon, R. A., Achnine, L., Kota, P., Liu, C.-J., Srinivasa, M. S. & Wang, L. The phenylpropanoid pathway and plant defence-a genomics perspective. Mol. Plant Pathol. 3, 371–390 (2002).
Liu, J., Liu, X., Dai, L. & Wang, G. Recent progress in elucidating the structure, function and evolution of disease resistance genes in plants. J. Gen. Genom. 34, 765–776 (2007).
Zhang, H. & Wang, S. Rice versus Xanthomonas oryzae pv. oryzae: a unique pathosystem. Curr. Opin. Plant Biol. 16, 188–195 (2013).
Song, W.-Y. et al. A receptor kinase-like protein encoded by the rice disease resistance gene, Xa21. Science 270, 1804–1806 (1995).
Marone, D., Russo, M., Laido, G., De Leonardis, A. D. & Matrangelo, A. M. Plant nucleotide binding site-leucine-rich repeats (NBS-LRR) genes: Active guardians in host defense responses. Int. J. Mol. Sci. 14, 7302–7326 (2013).
van Ooijen, G., van den Burg, H. A., Cornelissen, B. J. & Takken, F. L. Structure and function of resistance proteins in solanaceous plants. Annu. Rev. Phytopathol. 45, 43–72 (2007).
Sekhwal, M. K., Li, P., Lam, I., Wang, X., Cloutier, S. & You, F. M. Disease resistance gene analogs (RGAs) in plants. Int. J. Mol. Sci. 16, 19248–19290 (2015).
Katagiri, F. & Tsuda, K. Understanding the plant immune system. Mol. Plant Mol. Int. 23, 1531–1536 (2010).
Delphine, C. et al. A flagellin-induced complex of the receptor FLS2 and BAK1 initiates plant defence. Nature 448, 497–500 (2007).
Yang, Z. et al. Transcriptome-based analysis of mitogen-activated protein kinase cascades in the rice response to Xanthomonas oryzae infection. Rice 8, 4 (2015).
Boller, T. & He, S. Y. Innate immunity in plants: An arms race between pattern recognition receptors in plants and effectors in microbial pathogens. Science 324, 742–744 (2009).
Narsai, R. et al. Antagonistic, overlapping and distinct responses to biotic stress in rice (Oryza sative) and interactions with abiotic stress. BMC Genom. 14, 93 (2013).
Mur, L. A., Kenton, P., Lloyd, A. J., Ougham, H. & Prats, E. The hypersensitive response; the centenary is up us but how much do we know? J. Exp. Bot. 59, 501–520 (2008).
Robert-Seilaniantz, A., Grant, M. & Jones, J. D. G. Hormone crosstalk in plant disease and defense: more than just jasmonate-salicylate antagonism. Annu. Rev. Phytopathol. 49, 317–343 (2011).
Yuan, Y. X. et al. Functional analysis of rice NPR1-like genes reveals that OsNPR1/NH1 is the rice orthologue conferring disease resistance with enhanced herbivore susceptibility. Plant Biotechnol. J. 5, 313–324 (2007).
Vierstra, R. D. The ubiquitin-26S proteasome system at the nexus of plant biology. Nat. Rev. Mol. Cell Biol. 10, 385–397 (2009)
Kelley, D. R. & Estelle, M. Ubiquitin-mediated control of plant hormone signaling. Plant Physiol. 160, 47–55 (2012).
Thines, B. et al. JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signalling. Nature 448, 661–665 (2007).
Craig, A., Ewan, R., Mesmar, J., Gudipati, V. & Sadandom, A. E3 ubiquitin ligases and plant innate immunity. J. Exp. Bot. 60, 1123–1132 (2009).
Ali, M. B. & McNear, D. H. Induced transcriptional profiling of phenylpropanoid pathway genes increased flavonoid and lignin content in Arabidopsis leaves in response to microbial products. BMC Plant Biol. 14, 1 (2014).
Kauffman, H. E. R., Reddy, A. P. K. Hsieh, S. P. Y. & Merca, S. D. An improved technique for evaluating resistance of rice varieties to Xanthomonas oryzae. Plant Dis. Rep. 57, 537–541 (1973).
Haas, B. J. et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protocols 8, 1494–1512 (2013).
Huang, X. & Madan, A. Cap3: a DNA sequence assembly program. Genome Res. 9, 868–877 (1999).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Conesa, A. & Götz, S. Blast2GO: A comprehensive suite for functional analysis in plant genomics. Intl. J. Plant Genom. 2008, Article ID 619832 (2008).
Grabherr, M. G. et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 29, 644–652 (2011).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Acknowledgements
This work was supported by the Yunnan Province Scientific Projects (2012FA005, 2012HA002, and YBR1201504). We also think Dr. Md. Ashraful Alam so much for critically reading the manuscript.
Author information
Authors and Affiliations
Contributions
Z.-Q.C. and H.F. conceived and designed the research; X.-J.C., L.C., S.-Q.X., J.F., Y.C. and T.-Q.Y. performed the experiments. X.-J.C. and B.H. analyzed the data; X.-J.C., Z.-Q.C. and H.F. wrote and revised the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Cheng, XJ., He, B., Chen, L. et al. Transcriptome analysis confers a complex disease resistance network in wild rice Oryza meyeriana against Xanthomonas oryzae pv. oryzae. Sci Rep 6, 38215 (2016). https://doi.org/10.1038/srep38215
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep38215
This article is cited by
-
Complete genome resource unravels the close relation of an Indian Xanthomonas oryzae pv. oryzae strain IXOBB0003 with Philippines strain causing bacterial blight of rice
3 Biotech (2023)
-
A review of approaches to control bacterial leaf blight in rice
World Journal of Microbiology and Biotechnology (2022)
-
Diversity of resistance to bacterial blight and geographical distribution of 29 populations of wild rice [Oryza meyeriana (Zoll. & Moritzi) Baill.] in Yunnan, China
Genetic Resources and Crop Evolution (2021)
-
Comparative Transcriptome Analysis of Rice Resistant and Susceptible Genotypes to Xanthomonas oryzae pv. oryzae Identifies Novel Genes to Control Bacterial Leaf Blight
Molecular Biotechnology (2021)
-
Transcriptome sequencing revealed the mechanism of promoting floret opening by exogenous methyl jasmonate in sorghum
3 Biotech (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.